* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cell Processes
Survey
Document related concepts
Transcript
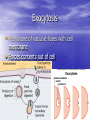
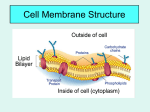
Cell Transport S. Dickinson HHS Biology How do “smells” get out of the balloon? • Diffusion • Selectively permeable membrane • Homeostasis • Active transport • Hypertonic/hypotonic/isotonic • Osmosis Concentration • The mass of a solute in a given volume of solution • If you dissolve 12 grams of salt in 3 liters of water, what is the concentration of salt in the solution? • 12g/3L = 4g/L • Suppose you add 12 more grams of salt in to the solution. What would be the resulting concentration? • 12g + 12g = 24 g • 24g/3L = 8g/L • What if you then added another 3 liters of water to that solution. What would be the resulting concentration? • 3L + 3L = 6L • 24g/6L = 4g/L 4g/L; 8g/L; 4g/L • Which solution would be called the most concentrated? • 8g/L • Why? – 8g in 1L is more than 4g in 1L Selectively Permeable • A membrane through which some substances can pass through, but some cannot Diffusion • Movement of particles from an area of high concentration to an area of low concentration • Particles try to reach equilibrium/homeostasis • Equilibrium/homeostasis is reached when the concentration of the solute is the same throughout the system Diffusion • Substances diffuse across membranes without requiring the cell to use energy • Particles of a solution continue to move across the membrane even when equilibrium is reached, there is just no further change in concentration Passive Transport • Does not require energy Active Transport (Fig. 7-19) • Movement of materials against the concentration gradient • Requires energy • Generally carried out by transport proteins in cell membrane • Can be used to transport calcium, potassium, and sodium ions across the membrane Endocytosis • Membrane infolds to take in materials • Pocket breaks loose to form vacuole in the cytoplasm – Phagocytosis: taking in food; amoeba's use this to take in food – Pinocytosis: membranes form pockets that fill with water and break off as vacuoles in the cytoplasm Exocytosis • Membrane of vacuole fuses with cell membrane • Forces contents out of cell Facilitated Diffusion • Does not require energy but does require a protein channel • Movement from high [] to low [] Osmosis • Diffusion of water through a selectively permeable membrane • Water moves from an area of high water concentration to an area of low water concentration Osmosis Cartoon Project • Draw an Osmosis Cartoon Project on the left side Biomolecule Foldable • Make a foldable like the Organelle Foldable only this time you need 4 flaps • You will write the Polymer/Biomolecule on the outside with a picture representation. On the inside, you will have the function on the side not cut and the monomer with a picture on the side you cut. Carbohydrates Made up on carbon, hydrogen, and oxygen Body’s main source of energy Can also be used for structural purpose Sugars Monosaccharides Single sugars Ex: glucose, galactose, fructose Disaccharides Two monosaccharides bound together (Monomer = monosaccharide; polymer = monosaccharides bound together. Ex: lactose (glucose and galactose), sucrose (glucose and fructose), and maltose (two glucose molecules) Monomer – building block of a molecule Polymer – the “building” or actual molecule Polysaccharide Thousands of monosaccharides linked together Ex: glycogen, starch cellulose Lipids Made mostly of carbon and hydrogen Not soluble in water Consists of fats, oils, and waxes Can be used to store energy Many lipids are formed with glycerol and a fatty acid tail Steroids are lipids Saturated Lipid with only single bonds “Saturated with hydrogens” Solid at room temperature Ex: animal fat (lard), fatty meats Unsaturated Has at least one double bond Liquid at room temperature Ex: vegetable oil, olive oil, canola oil Polyunsaturated Many double bonds (more than one can be considered polyunsaturated) Nucleic Acids Nucleotides joined covalently Contain carbon, hydrogen, oxygen, nitrogen, and phosphorous Polymers from monomers called nucleotides Nucleotide has 5-carbon sugar, phosphate group, and a nitrogenous baase RNA – ribose sugar; DNA – deoxyribose sugar – Store genetic information Proteins Contain carbon, nitrogen, hydrogen, and oxygen Monomer of protein (polymer) is amino acid – Have amino group (-NH2) at one end and a carboxyl group (-COOH) at the other end More than 20 different amino acids found in nature When two or more amino acids are bound, still has amino group on one end and carboxyl group on the other end Polypeptide chain has three or more amino acids bound together Polypeptide bond forms as a condensation reaction (rxn) joins the amino group of one amino acid with the carboxyl group of the next in line H2O released Peptides Short polymers Hypertonic • Solution has higher solute concentration than the cell • Water will move from inside the cell to outside the cell • Figure 7-16 (pg. 186) Hypotonic • Solution has a lower solute concentration than the cell • Water moves into the cell Isotonic • Concentration of solutes is the same in and out of the cell Utilize Fig. 7-16 • A homeowner contracts a lawn company to add fertilizer to the lawn in order to make the grass grow better. This process is normally done by spraying a mixture of fertilizer and water onto the lawn • What would happen if too much fertilizer and too little water were sprayed onto the lawn? • Can you suggest what happened to the cells of the grass? • In that case, was the fertilizer-water mixture hypotonic or hypertonic compared to the grass cells? Figure 7-15 • In the beaker on the left, which solution is hypertonic and which is hypotonic? – The solution on the left side of the membrane is hypertonic and the solution on the right is hypotonic • In this model, to which material is the membrane permeable, water or sugar? – Water!! This makes the membrane selectively permeable