* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 22 (Part 2)
Survey
Document related concepts
Transcript
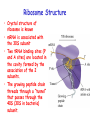
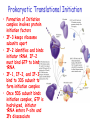
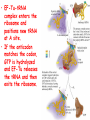
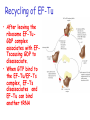
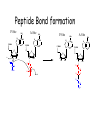
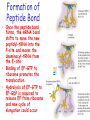
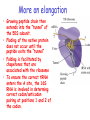
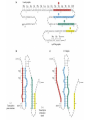
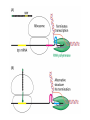
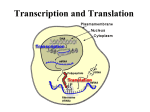
Chapter 22 (Part 2) Protein Synthesis Translation • Slow rate of synthesis (18 amino acids per second) • In bacteria translation and transcription are coupled. As soon as 5’ end of mRNA is synthesized translation begins. • Situation in eukaryotes differs since transcription and translation occur in different cellular compartments. Ribosomes • Protein biosynthetic machinery • Made of 2 subunits (bacterial 30S and 50S, Eukaryotes 40S and 60S) • Intact ribosome referred to as 70S ribosome in Prokaryotes and 80S ribosome in Eukaryotes • In bacteria, 20,000 ribosomes per cell, 20% of cell's mass. • Mass of ribosomes is roughly 2/3 RNA Prokaryotic Ribosome Structure • E. coli ribosome is 25 nm diameter, 2520 kD in mass, and consists of two unequal subunits that dissociate at < 1mM Mg2+ • 30S subunit is 930 kD with 21 proteins and a 16S rRNA • 50S subunit is 1590 kD with 31 proteins and two rRNAs: 23S rRNA and 5S rRNA Eukaryotic Ribosome Structure • Mitochondrial and chloroplast ribosomes are quite similar to prokaryotic ribosomes, reflecting their supposed prokaryotic origin • Cytoplasmic ribosomes are larger and more complex, but many of the structural and functional properties are similar • 40S subunit contains 30 proteins and 18S RNA. • 60S subunit contains 40 proteins and 3 rRNAs. Ribosome Assembly • Assembly is coupled w/ transcription and pre-rRNA processing Ribosome Structure • Crystal structure of ribosome is known • mRNA is associated with the 30S subunit • Two tRNA binding sites (P and A sites) are located in the cavity formed by the association of the 2 subunits. • The growing peptide chain threads through a “tunnel” that passes through the 40S (30S in bacteria) subunit. Mechanics of Protein Synthesis • All protein synthesis involves three phases: initiation, elongation, termination • Initiation involves binding of mRNA and initiator aminoacyl-tRNA to small subunit, followed by binding of large subunit • Elongation: synthesis of all peptide bonds with tRNAs bound to acceptor (A) and peptidyl (P) sites. • Termination occurs when "stop codon" reached Identification of Initiator Codon in Prokaryotes • Involves binding of initiator tRNA (Nformylmethionyl-tRNA) to initiator codon (first AUG) • The 30S subunit scans the mRNA for a specific sequence (Shine-Dalgarno Sequence) which is just upstream of the initiator codon. 16S RNA is involved in recognition of S-D sequence. Prokaryotic Translational Initiation • Formation of Initiation complex involves protein initiation factors • IF-3 keeps ribosome subunits apart • IF-2 identifies and binds initiator tRNA. IF-2 must bind GTP to bind tRNA. • IF-1, IF-2, and IF-3 bind to 30S subunit to form initiation complex • Once 50S subunit binds initiation complex, GTP is hydrolyzed, initiator tRNA enters P-site and IFs disassociate Eukaryotic Initiation of Translation • No S-D sequence. • CAP binding protein (CBP) 5’ end of mRNA by binding to 5’ CAP structure • An initiation complex forms with CBP, initiation factors and the 40S subunit. • The complex then scans the mRNA looking for the first AUG closest to the 5’ end of the mRNA • eIF-2 analogous to IF-2, transfers tRNA to P sight. GTP hydrolysis involed in release Chain Elongation Three step process: 1) Position correct aminoacyl-tRNA at acceptor site 2) Formation of peptide bond between peptidyl-tRNA at P site with aminoacyl-tRNA at A site. 3) Shifting mRNA by one codon relative to ribosome. • Elongation Factor Tu (EF-Tu) binds to aminoacyl-tRNA and delivers it to the A site of the ribosome • When EF-Tu binds GTP a conformational change occurs allowing it to bind to aminoacyl-tRNA. • EF-Tu-tRNA complex enters the ribosome and positions new tRNA at A site. • If the anticodon matches the codon, GTP is hydrolyzed and EF-Tu releases the tRNA and then exits the ribosome. Recycling of EF-Tu • After leaving the ribosome EF-TuGDP complex associates with EFTscausing GDP to disassociate. • When GTP bind to the EF-Tu/EF-Ts complex, EF-Ts disassociates and EF-Tu can bind another tRNA Peptide Bond formation P-Site N 5' tRNA N O H H+ O O C N 5' tRNA H OH H O CH H NH3+ N N N O H 5' tRNA H H O H OH N N H H OH H OH 5' tRNA N O H C N O H H O H OH C CH H H BASE NH2 N N O O H A-Site NH2 N O CH H H P-Site NH2 N N O O H A-Site NH2 H N O C H H CH H NH3+ N N Formation of Peptide Bond • Once the peptide bond forms, the mRNA band shifts to move the new peptidyl-tRNA into the P-site and moves the deaminacyl-tRNA from the E-site • Binding of EF-GTP to ribosome promotes the translocation • Hydrolysis of EF-GTP to EF-GDP is required to release EF from ribosome and new cycle of elongation could occur More on elongation • Growing peptide chain then extends into the “tunnel” of the 50S subunit. • Floding of the native protein does not occur until the peptide exits the “tunnel” • Folding is facilitated by chaperones that are associated with the ribosome • To ensure the correct tRNA enters the A site, the 16S RNA is involved in determing correct codon/anticodon pairing at positions 1 and 2 of the codon. Eukaryotic elongation process • Similar to what occurs in prokaryotes. • Analogous elongation factors. • EF-1a = EF-Tu docks tRNA in Asite • EF-1b = EF-Ts recycles EF-Tu • EF-2 = EF-G involved in translocation process Peptide Chain Termination • Proteins known as "release factors" recognize the stop codon (UGA, UAG, or UAA) at the A site • In E. coli RF-1 recognizes UAA and UAG, RF-2 recognizes UAA and UGA. • RF-3 binds GTP and enhances activities of RF-1 and –2. • Presence of release factors with a nonsense codon at A site transforms the peptidyl transferase into a hydrolase, which cleaves the peptidyl chain from the tRNA carrier • Hydrolysis of GTP is required for disassociation of RFs, ribosome subunit and new peptide Protein Synthesis is Expensive! • For each amino acid added to a polypeptide chain, 1 ATP and 3 GTPs are hydrolyzed. • This is the release of more energy than is needed to form a peptide bond. • Most of the energy is need to over-come entropy losses Regulation of Gene Expression RNA Processing 5’CAP Active enzyme Post-translational modification mRNA AAAAAA RNA Degradation Protein Degradation Regulation of Protein Synthesis Regulation could occur at two levels in translation 1) Initiation – formation of the initiation complex 2) Elongation – elongation could be stalled by if an mRNA contains “rare” codons Regulation of Globin gene translation by heme • When heme is low, HCI kinase phosphorylates eIF-2GDP complex, • GEF binds tightly to phosphorylated eiF-2GDP complex • prevents recycling of eIF-2-GDP and stops translation Regulation of the trp operon • Transcription and translation are tightly coupled in E. coli. • When Trp is aundant, transcription of the trp operon is repressed. • The mechanism of this repression is related to translation of the