* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download CHAPTER 6
Electron transport chain wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Butyric acid wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Metalloprotein wikipedia , lookup
Biochemical cascade wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Microbial metabolism wikipedia , lookup
Biosynthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Citric acid cycle wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Metabolic network modelling wikipedia , lookup
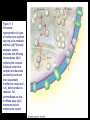
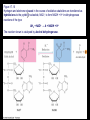
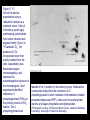
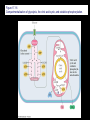
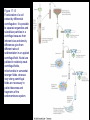
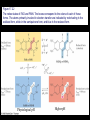
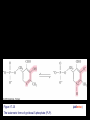
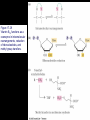
Chapter 17 Metabolism – An Overview Biochemistry by Reginald Garrett and Charles Grisham Metabolism • Metabolism represents the sum of the chemical changes that convert nutrients into energy and the chemically complex products of cells • Metabolism consists of literally hundreds of enzymatic reactions organized into discrete pathways • These pathways proceed in a stepwise fashion, transforming substrates into end products through many specific chemical intermediates • Metabolism is sometimes referred to as intermediary metabolism Metabolism • The metabolism map can be viewed as a set of dots and lines – Intermediate as a black dot – Enzyme as a line – More than 1000 different enzymes and 500 intermediates – About 80% of the intermediates connect to only one or two lines Lines 1 or 2 3 4 5 6 or more Dots 410 71 20 11 8 Outline of Chapter 17 1. Are There Similarities of Metabolism Between Organisms? 2. How Do Anabolic and Catabolic Processes Form the Core of Metabolic Pathways? 3. What Experiments Can Be Used to Elucidate Metabolic Pathways? 4. What Food Substances Form the Basis of Human Nutrition? Special Focus: Vitamins 17.1 – Are There Similarities of Metabolism Between Organisms? • Organisms show a marked similarity in their major metabolic pathways • All life descended from a common ancestral form • For example, Glycolysis, the metabolic pathway by which energy is released from glucose and captured in the form of ATP under anaerobic condition, is common to almost every cell Living things exhibit metabolic diversity • Although most cells have the same basic set of central metabolic pathways, different cells are characterized by the alternative pathways - There is also significant diversity • Classification: – Based on carbon requirement: Autotrophs use CO2; Heterotrophs use organic carbon – Based on energy source: Phototrophs use light; Chemotrophs use Glc, inorganic compounds NH4+ & S Living things exhibit metabolic diversity • Metabolic diversity among the 5 kingdoms • Oxygen is essential to life for aerobes – Aerobes – Anaerobes – Obligate aerobes, facultative anaerobes, and Obligate anaerobes The Sun is Primary Energy for Life • The flow of energy in the biosphere and the carbon and oxygen cycles are intimately related • Phototrophs use light to drive synthesis of organic molecules • Heterotrophs use these organic molecules as building blocks • CO2, O2, and H2O are recycled Figure 17.3 The flow of energy in the biosphere is coupled primarily to the carbon and oxygen cycles. 17.2 – How Do Anabolic and Catabolic Processes Form the Core of Metabolism Pathways? • Metabolism serves two fundamentally different purposes: the generation of energy to drive vital functions and synthesis of biological molecules • Metabolism consists of catabolism and anabolism • Catabolism: degradative pathways – Usually energy-yielding – Oxidative • Anabolism: biosynthetic pathways – Energy-requiring – Reductive Figure 17.4 Energy relationships between the pathways of catabolism and anabolism. Oxidative, exergonic pathways of catabolism release free energy and reducing power that are captured in the form of ATP and NADPH, respectively. Anabolic processes are endergonic, consuming chemical energy in the form of ATP and using NADPH as a source of high energy electrons for reductive purposes. Anabolism and Catabolism Are Not Mutually Exclusive • • Catabolism and anabolism occur simultaneously in the cell The conflicting demands of concomitant catabolism and anabolism are managed by cells in two ways 1. The cell maintains tight and separate regulation of both catabolism and anabolism 2. Competing metabolic pathways are often localized within different cellular compartment Organization of Enzymes in Pathways • • Pathways consist of sequential enzymatic steps The enzymes may be 1. Separate, soluble entities 2. or may form a multienzyme complex 3. or may be a membrane-bound system • New research indicates that multienzyme complexes are more common than once thought - metabolons Figure 17.5 Schematic representation of types of multienzyme systems carrying out a metabolic pathway: (a) Physically separate, soluble enzymes with diffusing intermediates. (b) A multienzyme complex. Substrate enters the complex and becomes covalently bound and then sequentially modified by enzymes E1 to E5 before product is released. No intermediates are free to diffuse away. (c) A membrane-bound multienzyme system. The pathways of catabolism converge to a few end products • Consists of three distinct stages – Stage 1: the nutrient macromolecules are broken down into their respective building blocks – Stage 2: building blocks are further degraded to yield an even more limit set of simpler metabolic intermediates – Stage 3: the oxidation of metabolic intermediates to generate the energy and to produce CO2 and H2O Figure 17.6 The three stages of catabolism. Stage 1: Proteins, polysaccharides, and lipids are broken down into their component building blocks, which are relatively few in number. Stage 2: The various building blocks are degraded into the common product, the acetyl groups of acetyl-CoA. Stage 3: Catabolism converges to three principal end products: water, carbon dioxide, and ammonia. Anabolic pathways diverge to synthesize many biomolecules • The proteins, nucleic acids, lipids, and polysaccharides are constructed from appropriate building blocks via the pathways of anabolism • The building blocks (amino acid, nucleotides, sugars, and fatty acids) can be generated from metabolites • Some pathways serve both in catabolism and anabolism –citric acid cycle- Such pathways are amphibolic Comparing Pathways • Anabolic & catabolic pathways involving the same product are not the same enzymatic reactions • Some steps may be common to both, others must be different - to ensure that each pathway is spontaneous • This also allows regulation mechanisms to turn one pathway on and the other off Figure 17.7 Parallel pathways of catabolism and anabolism must differ in at least one metabolic step in order that they can be regulated independently. Shown here are two possible arrangements of opposing catabolic and anabolic sequenced between A and P. (a) The parallel sequences proceed via independent routes. (b) Only one reaction has two different enzymes, a catabolic one (E3) and it’s anabolic counterpart (E6). These provide sites for regulation. ATP Serves in a Cellular Energy Cycle • ATP is the energy currency of cells • Phototrophs transform light energy into the chemical energy of ATP • In heterotrophs, catabolism produces ATP, which drives activities of cells • Energy released in the hydrolysis of ATP to ADP and Pi • ATP cycle carries energy from photosynthesis or catabolism to the energyrequiring processes of cells Figure 17.8 The ATP cycle in cells. ATP is formed via photosynthesis in phototrophic cells or catabolism in heterotrophic cells. Energy-requiring cellular activities are powered by ATP hydrolysis, liberating ADP and Pi. NAD+ and NADH system in Metabolism • NAD+ collects electrons released from the substrates in oxidative reactions of catabolism • Catabolism is oxidative - substrates lose reducing equivalents, usually H:- ions (hydride ion) • The hydride ions are transferred in enzymatic dehydrogenase reactions from the substrates to NAD+ molecules, reducing them to NADH • The ultimate oxidizing agent is O2, becoming reduced to H2O • Oxidation reaction s are exergonic, and the energy released is coupled with the formation of ATP Figure 17.9 Comparison of the state of reduction of carbon atoms in biomolecules: -CH2- (fats) > CHOH- (carbohydrates) C=O (carbonyls) > -COOH (carboxyls) >CO2 (carbon dioxide, the final products of catabolism). A comparison of state of reduction of carbon atoms in biomolecules. Figure 17.10 Hydrogen and electrons released in the course of oxidative catabolism are transferred as hydride ions to the pyridine nucleotide, NAD+, to form NADH + H+ in dehydrogenase reactions of the type AH2 + NAD+ → A + NADH + H+ The reaction shown is catalyzed by alcohol dehydrogenase. NADPH provides the reducing power for anabolic processes • Anabolism is reductive • The biosynthesis requires the reducing equivalents • NADPH provides the reducing power (electrons) for anabolic processes • In photosynthetic organism, the energy of light is used to pull electrons from water and transfer them to NAPD+; O2 is byproduct of this process Figure 17.11 Transfer of reducing equivalents from catabolism to anabolism via the NADPH cycle. 17.3 – What Experiments Can Be Used to Elucidate Metabolic Pathways? • Eduard Buchner (late 19th century) showed that fermentation of glucose in extract of broken yeast cells yielded ethanol and carbon dioxide. • This led to a search for intermediates of glucose breakdown. • Metabolic inhibitors were important tools for elucidating the pathway steps. • Mutations also were used to create specific metabolic blocks. Figure 17.12 The use of inhibitors to reveal the sequence of reactions in a metabolic pathway. (a) Control: Under normal conditions, the steady-state concentrations of a series of intermediates will be determined by the relative activities of the enzymes in the pathway. (b) Plus inhibitor: In the presence of an inhibitor (in this case, an inhibitor of enzyme 4), intermediates upstream of the metabolic block (B, C, and D) accumulate, revealing themselves as intermediates in the pathway. The concentration of intermediates lying downstream (E and F) will fall. Isotopic Tracers Can Be Used as Metabolic Probes • Substrates labeled with an isotopic form of some element can be fed to cells and used to elucidate metabolic sequences • Radioactive isotopes: 14C, 3H, 32P • Stable ‘heavy’ isotopes: 18O, 15N CO2 + H2O (CH2O) + O2 C16O2 + 2 H218O (CH216O) + H216O + 18O 2 Figure 17.13 One of the earliest experiments using a radioactive isotope as a metabolic tracer. Cells of Chlorella (a green alga) synthesizing carbohydrate from carbon dioxide were exposed briefly (5 sec) to 14C-labeled CO . The 2 products of CO2 incorporation were then quickly isolated from the cells, separated by twodimensional paper chromatography, and observed via autoradiographic exposure of the chromatogram. Such experiments identified radioactive 3phosphoglycerate (PGA) as the primary product of CO2 fixation. The 3phosphoglycerate was labeled in the 1-position (in its carboxyl group). Radioactive compounds arising from the conversion of 3phosphoglycerate to other metabolic intermediates included phosphoenolpyruvate (PEP), malic acid, triose phosphate, alanine, and sugar phosphates and diphosphates. (Photograph courtesy of Professor Melvin Calvin, Lawmann Berkeley Laboratory, University of California, Berkeley.) Figure 17.14 With NMR spectroscopy one can observe the metabolism of a living subject in real time. These NMR spectra show the changes in ATP, creatine-P (phosphocreatine), and Pi levels in the forearm muscle of a human subjected to 19 minutes of exercise. Note that the three P atoms of ATP (a ,b, and g) have different chemical shifts, reflecting their different chemical environments. Metabolic Pathways Are Compartmentalized Within Cells • Eukaryotic cells are extensively compartmentalized by an endomembrane system • The flow of metabolic intermediates in the cell is spatially as well as chemically segregated Figure 17.16 Compartmentalization of glycolysis, the citric acid cycle, and oxidative phosphorylation. Figure 17.15 Fractionation of a cell extract by differential centrifugation. It is possible to separate organelles and subcellular particles in a centrifuge because their inherent size and density differences give them different rates of sedimentation in an applied centrifugal field. Nuclei are pelleted in relatively weak centrifugal fields, mitochondria in somewhat stronger fields, whereas very strong centrifugal fields are necessary to pellet ribosomes and fragments of the endomembrane system. 17.4 – What Food Substances Form the Basis of Human Nutrition? • Protein is a rich source of nitrogen and also provides essential amino acids • Carbohydrates provide metabolic energy and essential components for nucleotides and nucleic acids • Lipids provide essential fatty acids that are key components of membranes and also important signal molecules • Fiber may be soluble or insoluble Special Focus: Vitamins • Many vitamins are "coenzymes" molecules that bring unusual chemistry to the enzyme active site • Vitamins and coenzymes are classified as "water-soluble" and "fat-soluble" • The water-soluble coenzymes exhibit the most interesting chemistry Vitamin B1: Thiamine and Thiamine Pyrophosphate Thiamine pyrophosphate (TPP) • Thiamine - a thiazole ring joined to a substituted pyrimidine by a methylene bridge • Thiamine-PP is the active form • TPP is involved in carbohydrate metabolism in which bonds to carbonyl carbons (aldehyde or ketone) • It catalyzes decarboxylations of a-keto acids and the formation and cleavage of a hydroxyketones Figure 17.17 Thiamine pyrophosphate (TPP), the active form of vitamin B1, is formed by the action of TPP-synthetase. Figure 17.18 Thiamine pyrophosphate participates in (a) the decarboxylation of a-keto acids and (b) the formation and cleavage of a-hydroxyketones. Some Vitamins Contain Adenine Nucleotides • All use the adenine nucleotide group solely for binding to the enzyme • Several classes of coenzymes: 1. pyridine dinucleotides 2. flavin mono- and dinucleotides 3. coenzyme A Nicotinic Acid and the Nicotinamide Coenzymes Two important coenzymes in this class: – Nicotinamide adenine dinucleotide (NAD+) – Nicotinamide adenine dinucleotide phosphate (NADP+) • The reduced forms of these coenzymes are NADH and NADPH • The nicotinamide coenzymes are electron carriers • They transfer hydride anion (H:-) to NAD(P)+ and from NAD(P)H Figure 17.19 The structures and redox states of the nicotinamide coenzymes. Hydride ion (H:-, a proton with two electrons) transfers to NAD+ to produce NADH. Figure 17.20 NAD+ and NADP+ participate exclusively in two-electron transfer reactions. For example, alcohols can be oxidized to ketones or aldehydes via hydride transfer to NAD(P)+. Nicotinamide Coenzymes Structural and mechanistic features • The quaternary nitrogen of the nicotinamide ring acts as an electron sink to facilitate hydride transfer • The C4-position (on the nicotinamide ring) of hydride transfer is a pro-chiral center • Hydride transfer is always stereospecific pro-R, pro-S position Niacin and Pellagra • Nicotinamide was first isolated in 1937 by Elvehjem at the University of Wisconsin • Note similarities between structures of pyridine, nicotinic acid, nicotinamide and nicotine • Tryptophan Riboflavin and the Flavin Coenzymes Riboflavin, or Vitamin B2 • Active forms are flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD) • All these substances contain ribitol and a flavin or isoalloxazine ring • FMN is not a true nucleotide • FAD is not a dinucleotide • But the names are traditional and they persist Figure 17.21 The structures of riboflavin, flavin mononucleotide (FMN), and flavin adenine dinucleotide (FAD). Flavin coenzymes bind tightly to the enzymes that use them, with typical dissociation constants in the range of 10-8 to 10-11 M, so that only very low levels of free flavin coenzymes occur in most cells. Even in organisms that rely on the nicotinamide coenzymes (NADH and NADPH) for many of their oxidationreduction cycles, the flavin coenzymes fill essential roles. Flavins are stronger oxidizing agents than NAD+ and NADP+. They can be reduced by both one-electron and two-electron pathways and can be reoxidized easily by molecular oxygen. Enzymes that use flavins to carry out their reactions— flavoenzymes —are involved in many kinds of oxidation-reduction reactions. Flavin Mechanisms Flavins are one- or two-electron transfer agents • Name "flavin" comes from Latin flavus for "yellow" • The oxidized form is yellow, semiquinones are blue or red and the reduced form is colorless • Flavin coenzymes participate in one-electron transfer and two-electron transfer reactions Figure 17.22 The redox states of FAD and FMN. The boxes correspond to the colors of each of these forms. The atoms primarily involved in electron transfer are indicated by red shading in the oxidized form, white in the semiquinone form, and blue in the reduced form. Physiological pH Higher pH Pantothenic Acid and Coenzyme A Pantothenic acid (vitamin B3) is a component of Coenzyme A (fig. 17.23) • Functions: – Activation of acyl groups for transfer by nucleophilic attack – Activation of the a-hydrogen of the acyl group for abstraction as a proton • Both of these functions are mediated by the reactive -SH group on CoA, which forms thioester linkages with acyl groups Figure 17.23 The structure of coenzyme A. Acyl groups form thioester linkages with the —SH group of the β-mercaptoethylamine moiety. • Acetyl-CoA has a high group-transfer potential Ethyl acetate + H2O → acetate + ethanol + H+ DGo’= - 20KJ/mol Acetyl-CoA + H2O → acetate + CoA-SH + H+ DGo’= - 31KJ/mol – Transfer of the acetyl-group from acetyl-CoA is more spontaneous than from an oxygen ester – The 4-phosphopantetheine group is also in acyl carrier protein in fatty acid biosynthesis Figure 17.24 Acyl transfer from acyl-CoA to a nucleophile is more favorable than transfer of an acyl group from an oxygen ester. Vitamin B6: Pyridoxine and Pyridoxal Phosphate Pyridoxine and pyridoxal-5-phosphate (PLP) • Exists in two tautomeric forms (Fig. 17.25) • Catalyzes reactions involving amino acids Transaminations, decarboxylations, eliminations, racemizations and aldol reactions (Fig. 17.26) • This versatile chemistry is due to: 1. formation of stable Schiff base (aldimine) adducts with a-amino groups of amino acids 2. Act as effective electron sink to stabilize reaction intermediates Figure 17.25 The tautomeric forms of pyridoxal-5-phosphate (PLP). (aldimine) Figure 17.26 The seven classes of reactions catalyzed by pyridoxal-5phosphate. 1. Transamination 2. a-decarboxylation 3. b-decarboxylation 4. b-elimination 5. g-elimination 6. Racemization 7. Aldol reactions Pyridoxal Phosphate Mechanisms • Figure 17.27 is a key figure - relate each intermediate to subsequent mechanisms • A Schiff base linkage with the e-NH2 group of an active site lysine in the absence of substrate • Appreciate the fundamental difference between intermediates 2 through 7 Figure 17.27 Pyriodoxal-5-phosphate forms stable Schiff base adducts with amino acids and acts as an effective electron sink to stabilize a variety of reaction intermediates. Vitamin B12 Contains the Metal Cobalt Cyanocobalamin • B12 is converted into two coenzymes in the body: – 5'-deoxyadenosylcobalamin – methylcobalamin Figure 17.28 The structure of cyanocobalamin (top) and simplified structures showing several coenzyme forms of vitamin B12. The Co—C bond of 5′–deoxyadenosylcobalamin is predominantly covalent (note the short bond length of 0.205 nm) but with some ionic character. Note that the convention of writing the cobalt atom as Co3+ attributes the electrons of the Co—C and Co—N bonds to carbon and nitrogen, respectively. B12 Function & Mechanism • B12 catalyzes 3 kinds of reactions: 1. Intramolecular rearrangements (isomerization; mutase) 2. Reductions of ribonucleotides to deoxyribonucleotides (in certain bacteria) 3. Methyl group transfers (assisted by tetrahydrofolate - which is covered in a later section of this chapter) Figure 17.29 Vitamin B12 functions as a coenzyme in intramolecular rearrangements, reduction of ribonucleotides, and methyl group transfers. Vitamin C: Ascorbic Acid Ascorbic acid • Most plants and animals make ascorbic acid for them it is not a vitamin • Only a few vertebrates - man, primates, guinea pigs, fruit-eating bats and some fish (rainbow trout, carp and Coho salmon) cannot make it • Vitamin C is a reasonably strong reducing agent • It functions as an electron carrier Figure 17.30 The physiological effects of ascorbic acid (vitamin C) are the result of its action as a reducing agent. A twoelectron oxidation of ascorbic acid yields dehydroascorbic acid. Roles of Vitamin C Many functions in the body • Hydroxylations of proline and lysine (essential for collagen) are Vitamin C-dependent • Metabolism of Tyr in brain depends on C • Fe mobilization from spleen depends on C • C may prevent anemia • C ameliorates allergic responses • C can stimulate the immune system Biotin "chemistry on a tether" • Biotin functions as a mobile carboxyl group carrier in a variety of enzymatic carboxylation reactions • Bound covalently to a lysine residue on the protein • The biotin-lysine conjugate is called biocytin • The biotin ring system is thus tethered to the protein by a long, flexible chain Figure 17.31 The structure of biotin Figure 17.32 Biotin is covalently linked to a protein via the ε-amino group of a lysine residue. The biotin ring is thus tethered to the protein by a ten-atom chain. It functions by carrying carboxyl groups between distant sites on biotindependent enzymes. Biotin Carboxylations • • • • Most use bicarbonate and ATP Whenever you see a carboxylation that requires ATP and CO2 or HCO3-, Activation by ATP involves formation of carboxyl phosphate Carboxyl group is transferred to biotin to form N-carboxy-biotin The "tether" allows the carboxyl group to be shuttled from the carboxylase subunit to the transcarboxylase subunit of ACC-carboxylase Lipoic Acid Another example of "chemistry on a tether"! • Lipoic acid, like biotin, is a ring on a chain and is linked to a lysine on its protein • Lipoic acid is an acyl group carrier • Found in pyruvate dehydrogenase and aketoglutarate dehydrogenase • Lipoic acid functions to couple acyl-group transfer and electron transfer during oxidation and decarboxylation of a-keto acids Figure 17.33 The oxidized and reduced forms of lipoic acid and the structure of the lipoic acid-lysine conjugate. Figure 17.34 The enzyme reactions catalyzed by lipoic acid. Folic Acid Folates are donors of 1-C units for all oxidation levels of carbon except that of CO2 • Active form is tetrahydrofolate (THF) • THF is formed by two successive reductions of folate by dihydrofolate reductase • The oxidation states in Table 17.6 page 571 Figure 17.35 Formation of THF from folic acid by the dihydrofolate reductase reaction. The R group on these folate molecules includes the one to seven (or more) glutamate units that folates characteristically contain. All of these glutamates are bound in g-carboxyl amide linkages (as in the folic acid structure shown in the A Deeper Look box on page 571). The one-carbon units carried by THF are bound at N5, or at N10, or as a single carbon attached to both N5 and N10. (DHF) (THF) Vitamin A Group Includes Retinol, Retinal, and Retinoic Acid Retinol, retinyl esters and retinal • Retinol is absorbed from animal source and synthesized from b-carotene from plant source • Retinol is esterified and transported to the liver • Retinol is converted to retinal in the retina of the eye and is linked to opsin to form rhodopsin, a light-sensitive pigment protein in the rods and cones (fig 17.36) • Retinoic acid affects growth, differentiation, and development Figure 17.36 The incorporation of retinal into the light-sensitive protein rhodopsin involves several steps. All-trans-retinol is oxidized by retinol dehydrogenase and then isomerized to 11-cis-retinal, which forms a Schiff base linkage with opsin to form light-sensitive rhodopsin. Vitamin D Is Essential for Proper Calcium Metabolism Ergocalciferol and cholecalciferol • Cholecalciferol is made in the skin by the action of UV light on 7-dehydrocholesterol • Major circulating form is 25-hydroxyvitamin D • 1,25-dihydroxycholecalciferol (1,25dihydroxyvitamin D3) is the most active form • It functions to regulate calcium homeostasis • and plays a role in phosphorus homeostasis Figure 17.37 (a) Vitamin D3 (cholecalciferol) is produced in the skin by the action of sunlight on 7dehydrocholesterol. The successive action of mixed-function oxidases in the liver and kidney produces 1,25dihydroxyvitamin D3, the active form of vitamin D. (b) Ergocalciferol is produced in analogous fashion from ergosterol. Circuratory system Vitamins E and K Less understood vitamins • Vitamin E (a-tocopherol) is a potent antioxidant 1. Molecular details are almost entirely unknown 2. May prevent membrane oxidations (unsaturated fatty acids) • Vitamin K is essential for blood clotting – A post-translational modification of prothrombin is essential to its function – Carboxylation of 10 glutamyl residues on prothrombin (to form g-carboxyglutamyl residues) is catalyzed by a vitamin K-dependent enzyme, liver microsomal glutamyl carboxylase Figure 17.38 The structure of vitamin E (αtocopherol). Figure 17.39 The structures of the K vitamins. Figure 17.40 The glutamyl carboxylase reaction is vitamin K-dependent. This enzyme activity is essential for the formation of gcarboxyglutamyl residues in a variety of proteins, including several proteins of the bloodclotting cascade (Figure 15.4). These latter carboxylations account for the vitamin K dependence of coagulation.