* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Butyric acid wikipedia , lookup
Mitochondrion wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Phosphorylation wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Electron transport chain wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Biochemistry wikipedia , lookup
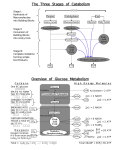
• Nutrition = how an organism obtains – energy and – a carbon source to build the organic molecules of cells. Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings Metabolism & energetics • Metabolism – sum total of all chemical reactions occurring in living organisms. – Anabolic pathways – synthesize compounds, generally endergonic. – Catabolic pathways – break down compounds, usually exergonic. • Many reactions also involve conversion of energy from one form to another. • Energy can exist as potential energy or kinetic energy. There are many kinds of energy that can interconvert from one form to another. 1. How does a cell maintain & regulate its metabolism? 2. How does a cell garner & utilize energy? 3. From where does this energy come? Organisms within the biosphere exchange molecules and energy Energy of sunlight Light (via plants) Useful chemical bond energy complex carbon, Autotrophs: glucose, amino acids Phototrophs & chemotrophs CO2, H2O Chemical oxidations (via iron & sulfur bacteria) Heterotrophs (e.g. some bacteria, animals, humans) Need 9 amino acids & 15 vitamins from outside sources 1st Law of Thermodynamics: In any process, the total energy of the universe remains constant. • Ways to obtain energy: – phototrophs use light energy – chemotrophs get energy from chemicals. • Ways to obtain Carbon – autotrophs only need only CO2 (inorganic C) – Heterotrophs need organic carbon sources • How do we get energy and carbon? Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings Several ways to generate energy! Includes: Animals, bacteria, fungi All organisms Chemotrophs (use chemical compounds as 10 energy source) Chemolithotrophs (use inorganic chem) Includes: plants, bacteria Phototrophs (use light as 10 energy source) Chemoorganotrophs (use organic chem) Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings What is the main role for enzymes? Metabolism 1) All biochemical reactions are integrated. 2) All living organisms have similar metabolic pathways. 3) Energetics and the reactions in the pathways are important. pp. 375 The Tokyo subway system is much like cellular metabolism. Food molecules: complex carbohydrates, etc. Molecules that form the cell: lipids, proteins, etc. Useful forms of energy Catabolic pathways Anabolic (biosynthetic) pathways Building blocks for biosynthesis: sugars, amino acids, etc. Adapted from Molecular Biology of the Cell, 4th ed. Heterotrophic metabolism: Interconversion of material and energy Catabolism (breakdown): Yields energy, precursors coupled Anabolism (synthesis): Requires energy, precursors How are catabolism and anabolism coupled? ATP couples energy between catabolism and anabolism Energy available for work & chemical synthesis (e.g. movement, signal amplification, etc. ADP + Pi ATP Major activities promoted by ATP: -locomotion -membrane transport -signal transduction -keeping materials in the cell -nucleotide synthesis anabolism Energy from food (fuel molecules) or from photosynthesis catabolism ATP is the principal carrier of chemical energy in the cell! pp. 381 ATP: the universal currency of free energy; “high energy” phosphate compound ATP + H2O ADP + Pi + H+ ADP + H2O AMP + Pi + H+ Go’ = -7.3 kcal/mol (G in cells = -12 kcal/mol) Go’ = -7.3 kcal/mol ATP ADP Molecular Biology of the Cell, 3rd ed. Fig. 2-28 ATP is an intermediate “high energy” compound Why ATP? It’s not the highest energy compound… It (and other nucleotide triphosphates) are stable & the high free energy of hydrolysis pp. 380 Another source of energy is the coupling of Oxidation & Reduction reactions NAD+ (and NADP+) carry high-energy electrons and hydrogen atoms. Reduced fuel catabolism NAD+(oxidized) Reduced Products Oxidized Fuel NADH(reduced) anabolism Oxidized Precursors “LEO the lion goes GER.” Losing Electrons (is) Oxidation … Gaining Electrons (is) Reduction. NAD+(oxidized) NADH(reduced) Nicotinamide adenine dinucleotide H: (hydride ion) (PO4) NADP+ NADPH pp. 383 Summary 1. Metabolism consists of many coupled & connecting reactions • • • Major source of energy = oxidation of carbon fuels • ATP = major carrier of energy 2. Few kinds of reactions; many recurring themes 3. Two major activated carrier molecules couple catabolism/anabolism reactions: • • ATP/ADP couples energy (through hydrolysis) NAD+/NADH couples oxidation/reduction (by carrying electrons & hydrogen atom) pg. 373 Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Adapted from MBOC4, fig. 2-70 & pp. 383 Respiration • The 3 types of bacterial respiration – Aerobic - require oxygen for their growth and existence – Anaerobic – do not require oxygen for any respiration – Anaerobes - prefer growing in the presence of oxygen, but can continue to grow without it Catabolism - Respiration, fermentation Respiration: • Glycolysis • Krebs/Tricarboxylic acid (TCA) Cycle • Electron transport chain & oxidative phosphorylation Fermentation: – Glycolysis followed by NAD+ regeneration reactions. Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Adapted from MBOC4, fig. 2-70 & pp. 383 Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Adapted from MBOC4, fig. 2-70 & pp. 383 Glucose catabolism O O O O O O O C6H12O6 glucose (a sugar) oxidation (requires O2) + 6O2 O H O H 6 CO2 + 6 H2O Carbon dioxide reduction water G= -686 kcal/mol Exergonic rxn 3 stages involved: 1) Glycolysis 2) TCA (citric acid) cycle 3) Electron transport/oxidative phosphorylation –Food = electron donor –Oxygen = terminal electron acceptor Regulation of Energy Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits glycolysis ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Adapted from MBOC4, fig. 2-70 & pp. 383 Citric acid cycle 2 CO2 TCA cycle 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O electron transport & ox. phosphorylation O O Glucose catabolism O O O O O O C6H12O6 glucose (a sugar) (requires O2) H O H 6 CO2 + 6 H2O Carbon dioxide water Go’ = -686 kcal/mol 3 stages involved: 1) Glycolysis 2) TCA (citric acid) cycle 3) Electron transport/oxidative phosphorylation no O2 required lactate (muscle) glucose ethanol (yeast) What organisms use glycolysis? 1. Anaerobes (grow without O2) 2. Facultative organisms (grow with & without O2) 3. Aerobes (grow only with O2) Glycolysis: Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Adapted from MBOC4, fig. 2-70 & pp. 383 Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Glycolysis Glycolysis • Splitting of glucose: yield of 2 pyruvate molecules from one glucose molecule. (Also H2O.) • ATP invested in early steps, energy generated in later steps. Net energy yield: 2 ATP, 2 NADH + 2 H+. Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Adapted from MBOC4, fig. 2-70 & pp. 383 Krebs Cycle • Transition step required after pyruvate enters mitochondrion; pyruvate converted to Acetyl CoA. (NAD+ reduced to NADH during this process.) • Krebs cycle doesn’t directly need oxygen, but won’t occur without it. • Krebs cycle involves decarboxylation, oxidation to generate NADH, FADH2, ATP. CO2 is byproduct of these steps. • NADH, FADH2 will relay electrons to electron transport chain. Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Adapted from MBOC4, fig. 2-70 & pp. 383 Electron transport system • Electron transport chain and oxidative phosphorylation produce ATP from products of glycolysis, Krebs. • Electron transport chain = protein complexes with prosthetic groups in/on inner mitochondrial membrane. (Some groups are able to move! E.g. Cyt C) • ETC facilitates series of redox reactions, with oxygen as final electron acceptor. • ATP formation uses proton motive force - voltage across membrane (ion gradient) that results from high [H+] in intermembrane space. Redox reactions • Many energy transfers involve transfer of electrons (or hydrogen atoms). • Oxidation and reduction occur together. – – – – Loss of electrons from one substance = oxidation. Addition of electrons to a substance = reduction. Oxidizing agent - accepts electrons. Reducing agent - gives up electrons. E.g. Na + Cl -> Na+ + Cloxidation reduction Electron transport chain - series of redox reactions • Cells release energy in stages. Electron transport system Development of Proton Motive Force from Chemiosmosis Formation of ATP from Proton Motive Force and ATP Synthase ATP Production during Aerobic Respiration by Oxidative Phosphorylation involving Electron Transport System and Chemiosmosis Bacterial electron transport ASM digital image collection: http://www.asmusa.org Bacterial chemiosmotic ATP generation Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Adapted from MBOC4, fig. 2-70 & pp. 383 Cellular Metabolism Part 2: Breakdown of simple subunits to acetyl CoA accompanied by production of limited amounts of ATP and NADH fats polysaccharides proteins fatty acids and glycerol simple sugars amino acids glucose glycolysis Part 1: Breakdown of large macromolecules to simple subunits ATP NADH pyruvate Acetyl CoA CoA Part 3: Complete oxidation of acetyl CoA to H2O and CO2 accompanied by production of large amounts of NADH and ATP in mitochondrion Citric acid cycle 2 CO2 8 e- (Reducing power as NADH) oxidative phosphorylation ATP O2 H2O Adapted from MBOC4, fig. 2-70 & pp. 383 Fermented … food? • Yogourt – Fermented milk, fermentation carried out by lactic acid bacteria. • Bread – Simple fermentation of sugar to alchohol and CO2 by bread yeast Saccharomyces cerevisiae. CO2 makes bread rise. • Kimchee – Cabbage and other veggies fermented by lactic acid bacteria. • Even some meat & fish products! – E.g. Country-cured ham, Katsuobushi (tuna) Unusual catabolism • Badger Ammunitions Plant - 1942-1976 provided weapons for the military and handled large quantities of explosive nitroglycerin (NG). CONTAMINATION!!!!! How can we clean the NG up? • Organisms capable of degrading NG? – Microorganisms: e.g. Pseudomonas fluorescens, Pseudomonas putida Pseudomonads only convert NG to mononitroglycerin (MNG). Other microbes in soil degrade MNG to glycerol. Glycerol can be converted to glyceraldehyde-3-phosphate and further metabolized. Amazing enzyme • P. fluorescens & P. putida use single enzyme: xenobiotic reductase. • Nonspecific enzyme, recognizes many molecules carrying nitro group (like trinitrotoluene: TNT). Many bacteria important in bioremediation! Two nutritional modes are unique to prokaryotes • Chemoautotrophs – use CO2 as a carbon source, but they obtain energy by oxidizing inorganic substances, – Inorganic energy sources = hydrogen sulfide (H2S), ammonia (NH3), and ferrous ions (Fe2+). – E.g. Nitrobacter - key in N-cycle converts ammonia (NH4) to nitrate (NO3) Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings • Photoheterotrophs – use light to generate ATP but obtain their carbon in organic form. – This mode is restricted to prokaryotes. – E.g. purple bacteria - make salt flats purple & red – E.g. green bacteria – Where does “red herring” come from? • dead herring have salt coating: halophiles grow on salt (red color; smelly); dragged around by animal rights activists to stop fox hunts Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings Carbon cycle • The majority of known prokaryotes are chemoheterotrophs. – parasites, which absorb nutrients from the body fluids of living hosts. – saprobes, decomposers that absorb nutrients from dead organisms, • Almost any organic molecule is food for one of the many chemoheterotrophic bacteria (like oil) • If it can’t be broken down by bacteria its called nonbiodegradable. Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings Nitrogen Cycle – Eukaryotes can only use organic nitrogen, NO3 or NH4. – Diverse prokaryotes can metabolize most nitrogenous compounds. • Prokaryotes are essential to converting N into usable forms for eukaryotes Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings • Prokaryotes are responsible for the key steps in the cycling of nitrogen through ecosystems. – Some chemoautotrophic bacteria convert ammonium (NH4+) to nitrite (NO2-). – Others “denitrify” nitrite or nitrate (NO3-) to N2, returning N2 gas to the atmosphere. – A diverse group of prokaryotes, including cyanobacteria, can use atmospheric N2 directly. – During nitrogen fixation, they convert N2 to NH4+, making atmospheric nitrogen available to other organisms for incorporation into organic molecules. Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings • Cyanobacteria fix N and C (photosynthesis) – = most self-sufficient of all organisms. – Only need: light, CO2, N2, water and some minerals to grow. Fig. 27.11 Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings • Cyanobacteria thought to put 02 in atmosphere. – = Massive change in the world • Great for aerobes (who require O2) • Deadly for anerobes (who are poisoned by o2) – Forced to live in remaining anerobic environments – Prokaryotes can be facultative or obligate aerobes or anerobes • Eukaryotes are all aerobic Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings 2. Photosynthesis evolved early in prokaryotic life • First prokaryotes were probably heterotrophs – Ate the primordial soup of early earth • But photosynthesis (harnessing the sun) shows up early in the fossil record) Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings • Similarity in complex machinery suggests photosynthesis evolved once. – = most parsimonious hypothesis, – Thus:heterotrophic groups represent a loss of photosynthetic ability during evolution. Copyright © 2002 Pearson Education, Inc., publishing as Benjamin Cummings