* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lecture Resource ()
Physical organic chemistry wikipedia , lookup
Bottromycin wikipedia , lookup
George S. Hammond wikipedia , lookup
Aromatization wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Ene reaction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Hydroformylation wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Stille reaction wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Asymmetric induction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
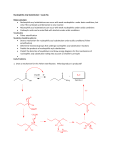
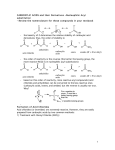
Organic Chemistry 4th Edition Paula Yurkanis Bruice Chapter 17 Carbonyl Compounds I Nucleophilic Acyl Substitution Irene Lee Case Western Reserve University Cleveland, OH ©2004, Prentice Hall Carbonyl Compounds with Groups that Can Be Replaced by a Nucleophile Nomenclature of Carboxylic Acids Acyl Halides O O O C CH3 C H3C Cl C Cl ethanoyl chloride acetyl chloride CH3CH2CHCH2 Br 3-methylpentanoyl bromide b-methylvalerylbromide cyclopentanecarbonyl chloride Acid Anhydrides R O O C C O R Esters Salts of Carboxylic Acids Cyclic Esters Are Known as Lactones Amides Amides with Substituent(s) Bonded to the Nitrogen O O C C CH3CH2 CH3CH2CH2CH2 NH NCH2CH3 CH3 N-cyclohexylpropanamide N-ethyl-N-methylpentanamide Cyclic Amides Are Known as Lactams O a O NH a NH b d g 2-azacyclohexanone d-valerolactam O b NH g 2-azacyclopentanone g-butyrolactam 2-azacyclobutanone b-propiolactam Nitriles CH3 CH3C C N ethanenitrile acetonitrile methylcyanide N benzenecarbonitrile benzonitrile phenyl cyanide CH2 CHC propenenitrile acrylonitrile CH3CHCH2CH2CH2C N 5-methylhexanenitrile d-methylcapronitrile isohexyl cyanide N Two major resonance contributors in esters, carboxylic acids, and amides Carboxylic acids have relatively high boiling points because Amides have the highest boiling points The reactivity of carbonyl compounds resides in the polarity of the carbonyl group in a nucleophilic acyl substitution reaction Z– will be expelled if it is a much weaker base than Y– (k–1 >> k2) Y– will be expelled if it is a much weaker base (k2 >> k–1) Reaction Coordinate Diagrams for Nucleophilic Acyl Substitution Reactions (a) the Nu– is a weaker base (b) the Nu– is a stronger base (c) the Nu– and the leaving group have similar basicities A Molecular Orbital Description of How Carbonyl Compounds React All carboxylic acid derivatives react by the same general mechanism The tetrahedral intermediate eliminates the weakest base If the nucleophile is neutral … Reactions of Acyl Halides Formation of Amides from Acyl Halides Tertiary amines cannot form amides Reactions of Acid Anhydrides Acid anhydrides do not react with sodium chloride or with sodium bromide Reactions of Esters a hydrolysis reaction a transesterification an aminolysis reaction There are no negatively charged species in the reaction Excess water will force the equilibrium to the right Alcohols that have low boiling points can be removed by distillation as they are formed Acid catalyzes the reaction by … Esters with tertiary alkyl groups undergo hydrolysis much more rapidly than do others H R O CH3 C C CH3 R O OH CH3 C C CH3 O CH3 CH3 H HO OH2 CH3 C CH3 CH3 CH3 HO C CH3 CH3 Hydroxide ion increases the rate of formation of the tetrahedral intermediate This reaction is not reversible Elucidating the reaction mechanism of nucleophilic acyl substitution Fats and Oils Are Triesters of Glycerol Nucleophilic Acyl Substitution of Carboxylic Acids Carboxylic acids do not undergo nucleophilic acyl substitution reactions with amines Hydrolysis of Amides Dehydration of an Amide Dehydration reagents commonly used are SOCl2, P2O5, or POCl3 An amide cannot be hydrolyzed without a catalyst An acid catalyst can make a group a better leaving group Hydrolysis of an Imide The Gabriel Synthesis Nitriles are harder to hydrolyze than amides Designing the Synthesis of Cyclic Compounds Formation of lactones Preparation of a compound with a ketone group attached to a benzene ring Preparation of a cyclic ester Activation of Carboxylic Acids The goal is to convert the OH group into a better leaving group such that the acyl chloride can be prepared The acyl halide can be used to prepare other carboxylic acid derivatives Activated Carboxylic Acid Derivatives in Living Organisms Dicarboxylic acids readily lose water when heated if they can form a cyclic anhydride with a five- or a six-membered ring