* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Organic Chemistry Fifth Edition
Kinetic isotope effect wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Discodermolide wikipedia , lookup
Marcus theory wikipedia , lookup
Aldol reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Petasis reaction wikipedia , lookup
Stille reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
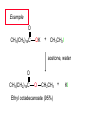
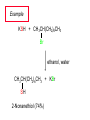
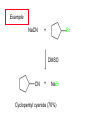
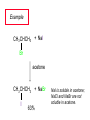
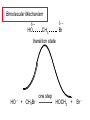
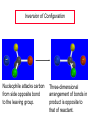
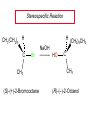
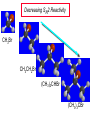
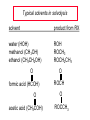
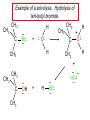
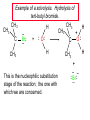
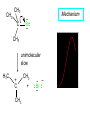
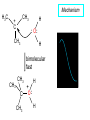
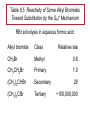
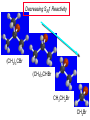
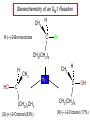
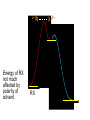
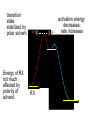
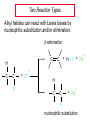
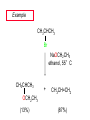
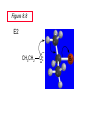
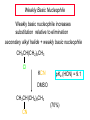
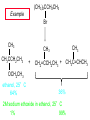
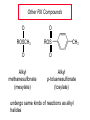
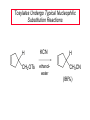
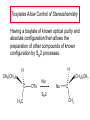
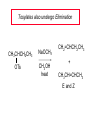
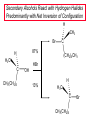
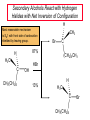
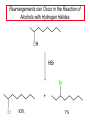
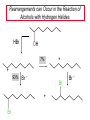
Chapter 8 Nucleophilic Substitution 8.1 Functional Group Transformation By Nucleophilic Substitution Nucleophilic Substitution – Y: + R X Y R + – :X Nucleophile is a Lewis base (electron-pair donor), often negatively charged and used as Na+ or K+ salt. Substrate is usually an alkyl halide. Nucleophilic Substitution Substrate cannot be an a vinylic halide or an aryl halide, except under certain conditions to be discussed in Chapter 12. X C C X Table 8.1 Examples of Nucleophilic Substitution Alkoxide ion as the nucleophile R' ..– O: .. + R X gives an ether R' .. O .. R + :X – Example (CH3)2CHCH2ONa + CH3CH2Br Isobutyl alcohol (CH3)2CHCH2OCH2CH3 + NaBr Ethyl isobutyl ether (66%) Table 8.1 Examples of Nucleophilic Substitution Carboxylate ion as the nucleophile O ..– + R X R'C O: .. gives an ester O R'C .. O .. R + :X – Example O CH3(CH2)16C OK + CH3CH2I acetone, water O CH3(CH2)16C O CH2CH3 + Ethyl octadecanoate (95%) KI Table 8.1 Examples of Nucleophilic Substitution Hydrogen sulfide ion as the nucleophile H ..– S: .. H .. S .. + R X gives a thiol R + :X – Example KSH + CH3CH(CH2)6CH3 Br ethanol, water CH3CH(CH2)6CH3 + KBr SH 2-Nonanethiol (74%) Table 8.1 Examples of Nucleophilic Substitution Cyanide ion as the nucleophile :N – C: + C R R X gives a nitrile :N + :X – Example NaCN + Br DMSO CN + NaBr Cyclopentyl cyanide (70%) Table 8.1 Examples of Nucleophilic Substitution Azide ion as the nucleophile – :N .. – : N .. + gives an alkyl azide + – :N N N .. .. R + N R + X :X – Example NaN3 + CH3CH2CH2CH2CH2I 2-Propanol-water CH3CH2CH2CH2CH2N3 + NaI Pentyl azide (52%) Table 8.1 Examples of Nucleophilic Substitution Iodide ion as the nucleophile ..– : ..I: + R X gives an alkyl iodide .. : ..I R + :X – Example CH3CHCH3 + NaI Br acetone CH3CHCH3 + NaBr I 63% NaI is soluble in acetone; NaCl and NaBr are not soluble in acetone. 8.2 Relative Reactivity of Halide Leaving Groups Generalization Reactivity of halide leaving groups in nucleophilic substitution is the same as for elimination. RI most reactive RBr RCl RF least reactive Problem 8.2 A single organic product was obtained when 1-bromo-3-chloropropane was allowed to react with one molar equivalent of sodium cyanide in aqueous ethanol. What was this product? BrCH2CH2CH2Cl + NaCN Br is a better leaving group than Cl Problem 8.2 A single organic product was obtained when 1-bromo-3-chloropropane was allowed to react with one molar equivalent of sodium cyanide in aqueous ethanol. What was this product? BrCH2CH2CH2Cl + NaCN :N C CH2CH2CH2Cl + NaBr 8.3 The SN2 Mechanism of Nucleophilic Substitution Kinetics Many nucleophilic substitutions follow a second-order rate law. CH3Br + HO – CH3OH + Br – rate = k[CH3Br][HO – ] inference: rate-determining step is bimolecular Bimolecular Mechanism HO Br CH3 transition state HO – + CH3Br one step HOCH3 + Br – Stereochemistry Nucleophilic substitutions that exhibit second-order kinetic behavior are stereospecific and proceed with inversion of configuration. Inversion of Configuration Nucleophile attacks carbon from side opposite bond to the leaving group. Three-dimensional arrangement of bonds in product is opposite to that of reactant. Stereospecific Reaction A stereospecific reaction is one in which stereoisomeric starting materials yield products that are stereoisomers of each other. The reaction of 2-bromooctane with NaOH (in ethanol-water) is stereospecific. (+)-2-Bromooctane (–)-2-Octanol (–)-2-Bromooctane (+)-2-Octanol Stereospecific Reaction H (CH ) CH 2 5 3 CH3(CH2)5 H NaOH C Br CH3 (S)-(+)-2-Bromooctane HO C CH3 (R)-(–)-2-Octanol Problem 8.4 The Fischer projection formula for (+)-2-bromooctane is shown. Write the Fischer projection of the (–)-2-octanol formed from it by nucleophilic substitution with inversion of configuration. Problem 8.4 The Fischer projection formula for (+)-2-bromooctane is shown. Write the Fischer projection of the (–)-2-octanol formed from it by nucleophilic substitution with inversion of configuration. CH3 H CH3 Br CH2(CH2)4CH3 HO H CH2(CH2)4CH3 8.4 Steric Effects and SN2 Reaction Rates Crowding at the Reaction Site The rate of nucleophilic substitution by the SN2 mechanism is governed by steric effects. Crowding at the carbon that bears the leaving group slows the rate of bimolecular nucleophilic substitution. Table 8.2 Reactivity Toward Substitution by the SN2 Mechanism RBr + LiI RI + LiBr Alkyl bromide Class Relative rate CH3Br Methyl 221,000 CH3CH2Br Primary 1,350 (CH3)2CHBr Secondary 1 (CH3)3CBr Tertiary too small to measure Decreasing SN2 Reactivity CH3Br CH3CH2Br (CH3)2CHBr (CH3)3CBr Decreasing SN2 Reactivity CH3Br CH3CH2Br (CH3)2CHBr (CH3)3CBr Crowding Adjacent to the Reaction Site The rate of nucleophilic substitution by the SN2 mechanism is governed by steric effects. Crowding at the carbon adjacent to the one that bears the leaving group also slows the rate of bimolecular nucleophilic substitution, but the effect is smaller. Table 8.3 Effect of Chain Branching on Rate of SN2 Substitution RBr + LiI RI + LiBr Alkyl bromide Structure Relative rate Ethyl CH3CH2Br 1.0 Propyl CH3CH2CH2Br 0.8 Isobutyl (CH3)2CHCH2Br 0.036 Neopentyl (CH3)3CCH2Br 0.00002 8.5 Nucleophiles and Nucleophilicity Nucleophiles The nucleophiles described in Sections 8.1-8.4 have been anions. – .. – .. – .. – : etc. : N C: : : HS HO CH O 3 .. .. .. Not all nucleophiles are anions. Many are neutral. .. .. : NH3 for example CH3OH HOH .. .. All nucleophiles, however, are Lewis bases. Nucleophiles Many of the solvents in which nucleophilic substitutions are carried out are themselves nucleophiles. .. HOH .. .. CH3OH .. for example Solvolysis The term solvolysis refers to a nucleophilic substitution in which the nucleophile is the solvent. Solvolysis substitution by an anionic nucleophile R—X + :Nu— R—Nu + :X— solvolysis R—X + :Nu—H step in which nucleophilic substitution occurs + R—Nu—H + :X— Solvolysis substitution by an anionic nucleophile R—X + :Nu— R—Nu + :X— solvolysis R—X + :Nu—H products of overall reaction + R—Nu—H + :X— R—Nu + HX Example: Methanolysis Methanolysis is a nucleophilic substitution in which methanol acts as both the solvent and the nucleophile. CH3 CH3 CH3 R—X + : O: + R O: H H –H+ R O .. : The product is a methyl ether. Typical solvents in solvolysis solvent product from RX water (HOH) methanol (CH3OH) ethanol (CH3CH2OH) ROH ROCH3 ROCH2CH3 O O formic acid (HCOH) O acetic acid (CH3COH) ROCH O ROCCH3 Nucleophilicity is a measure of the reactivity of a nucleophile Table 8.4 compares the relative rates of nucleophilic substitution of a variety of nucleophiles toward methyl iodide as the substrate. The standard of comparison is methanol, which is assigned a relative rate of 1.0. Table 8.4 Nucleophilicity Rank Nucleophile very good good I-, HS-, RSBr-, HO-, fair weak very weak NH3, Cl-, F-, RCO2H2O, ROH RCO2H Relative rate >105 104 RO-, CN-, N3103 1 10-2 Major factors that control nucleophilicity Basicity Solvation Small negative ions are highly solvated in protic solvents. Large negative ions are less solvated. Table 8.4 Nucleophilicity Rank Nucleophile Relative rate good HO–, RO– 104 RCO2– 103 H2O, ROH 1 fair weak When the attacking atom is the same (oxygen in this case), nucleophilicity increases with increasing basicity. Major factors that control nucleophilicity Basicity Solvation Small negative ions are highly solvated in protic solvents. Large negative ions are less solvated. Figure 8.3 Solvation of a chloride ion by ion-dipole attractive forces with water. The negatively charged chloride ion interacts with the positively polarized hydrogens of water. Table 8.4 Nucleophilicity Rank Nucleophile Relative rate Very good I- >105 good Br- 104 fair Cl-, F- 103 A tight solvent shell around an ion makes it less reactive. Larger ions are less solvated than smaller ones and are more nucleophilic. 8.6 The SN1 Mechanism of Nucleophilic Substitution A question... Tertiary alkyl halides are very unreactive in substitutions that proceed by the SN2 mechanism. Do they undergo nucleophilic substitution at all? Yes. But by a mechanism different from SN2. The most common examples are seen in solvolysis reactions. Example of a solvolysis. Hydrolysis of tert-butyl bromide. CH3 CH3 H H CH3 CH3 .. + + : O: O: C Br : C .. H H CH CH 3 3 + CH3 CH3 C CH3 .. OH .. + H .. Br : .. .. – : Br : .. Example of a solvolysis. Hydrolysis of tert-butyl bromide. CH3 CH3 H H CH3 CH3 .. + + : O: O: C Br : C .. H H CH CH 3 3 + This is the nucleophilic substitution stage of the reaction; the one with which we are concerned. .. – : Br : .. Example of a solvolysis. Hydrolysis of tert-butyl bromide. CH3 CH3 H H CH3 CH3 .. + + : O: O: C Br : C .. H H CH CH 3 3 + The reaction rate is independent of the concentration of the nucleophile and follows a first-order rate law. rate = k[(CH3)3CBr] .. – : Br : .. Example of a solvolysis. Hydrolysis of tert-butyl bromide. CH3 CH3 H H CH3 CH3 .. + + : O: O: C Br : C .. H H CH CH 3 3 + The mechanism of this step is not SN2. It is called SN1 and begins with ionization of (CH3)3CBr. .. – : Br : .. Kinetics and Mechanism rate = k[alkyl halide] First-order kinetics implies a unimolecular rate-determining step. Proposed mechanism is called SN1, which stands for substitution nucleophilic unimolecular CH3 CH3 Mechanism .. Br: .. C CH3 unimolecular slow H3C + C CH3 CH3 + .. – : Br : .. Mechanism H3C CH3 + C H : O: CH3 H bimolecular fast CH3 CH3 H + C CH3 O: H carbocation formation R+ carbocation capture proton transfer RX + ROH2 ROH Characteristics of the SN1 mechanism first order kinetics: rate = k[RX] unimolecular rate-determining step carbocation intermediate ate follows carbocation stability rearrangements sometimes observed reaction is not stereospecific much racemization in reactions of optically active alkyl halides 8.7 Carbocation Stability and SN1 Reaction Rates Electronic Effects Govern SN1 Rates The rate of nucleophilic substitution by the SN1 mechanism is governed by electronic effects. Carbocation formation is rate-determining. The more stable the carbocation, the faster its rate of formation, and the greater the rate of unimolecular nucleophilic substitution. Table 8.5 Reactivity of Some Alkyl Bromides Toward Substitution by the SN1 Mechanism RBr solvolysis in aqueous formic acid Alkyl bromide Class Relative rate CH3Br Methyl 0.6 CH3CH2Br Primary 1.0 (CH3)2CHBr Secondary 26 (CH3)3CBr Tertiary ~100,000,000 Decreasing SN1 Reactivity (CH3)3CBr (CH3)2CHBr CH3CH2Br CH3Br 8.8 Stereochemistry of SN1 Reactions Generalization Nucleophilic substitutions that exhibit first-order kinetic behavior are not stereospecific. Stereochemistry of an SN1 Reaction CH3 H C R-(–)-2-Bromooctane Br CH3(CH2)5 H HO CH3 C (CH2)5CH3 (S)-(+)-2-Octanol (83%) CH3 H2O H C OH CH3(CH2)5 (R)-(–)-2-Octanol (17%) Figure 8.6 Ionization step gives carbocation; three bonds to chirality center become coplanar + Leaving group shields one face of carbocation; nucleophile attacks faster at opposite face. 8.9 Carbocation Rearrangements in SN1 Reactions Because... carbocations are intermediates in SN1 reactions, rearrangements are possible. Example CH3 CH3 CH3 H2O C CHCH3 H Br CH3 C CH2CH3 OH (93%) H2O CH3 CH3 C H CH3 CHCH3 + CH3 C + CHCH3 H 8.10 Effect of Solvent on the Rate of Nucleophilic Substitution In general... SN1 Reaction Rates Increase in Polar Solvents R X R+ Energy of RX not much affected by polarity of solvent. RX transition state stabilized by polar solvent R X R+ Energy of RX not much affected by polarity of solvent. RX activation energy decreases; rate increases In general... SN2 Reaction Rates Increase in Polar Aprotic Solvents An aprotic solvent is one that does not have an —OH group. Table 8.7 Relative Rate of SN2 Reactivity versus Type of Solvent CH3CH2CH2CH2Br + N3– Solvent Type Relative rate CH3OH polar protic 1 H2O polar protic 7 DMSO polar aprotic 1300 DMF polar aprotic 2800 Acetonitrile polar aprotic 5000 Mechanism Summary SN1 and SN2 When... Primary alkyl halides undergo nucleophilic substitution: they always react by the SN2 mechanism. Tertiary alkyl halides undergo nucleophilic substitution: they always react by the SN1 mechanism. Secondary alkyl halides undergo nucleophilic substitution: they react by the SN1 mechanism in the presence of a weak nucleophile (solvolysis). SN2 mechanism in the presence of a good nucleophile. 8.11 Substitution and Elimination as Competing Reactions Two Reaction Types Alkyl halides can react with Lewis bases by nucleophilic substitution and/or elimination. -elimination C H C C X + :Y C + H Y + :X – H C C + :X – Y nucleophilic substitution – Two Reaction Types How can we tell which reaction pathway is followed for a particular alkyl halide? -elimination C H C C X + :Y C + H Y + :X – H C C + :X – Y nucleophilic substitution – Elimination versus Substitution A systematic approach is to choose as a reference point the reaction followed by a typical alkyl halide (secondary) with a typical Lewis base (an alkoxide ion). The major reaction of a secondary alkyl halide with an alkoxide ion is elimination by the E2 mechanism. Example CH3CHCH3 Br NaOCH2CH3 ethanol, 55°C CH3CHCH3 + CH3CH=CH2 OCH2CH3 (13%) (87%) Figure 8.8 E2 CH3CH2 •• – O •• •• Br When is Substitution Favored? Given that the major reaction of a secondary alkyl halide with an alkoxide ion is elimination by the E2 mechanism, we can expect the proportion of substitution to increase with: 1) decreased crowding at the carbon that bears the leaving group Uncrowded Alkyl Halides Decreased crowding at carbon that bears the leaving group increases substitution relative to elimination. primary alkyl halide CH3CH2CH2Br NaOCH2CH3 ethanol, 55°C CH3CH2CH2OCH2CH3 + (91%) CH3CH=CH2 (9%) But a Crowded Alkoxide Base Can Favor Elimination Even with a Primary Alkyl Halide primary alkyl halide + bulky base CH3(CH2)15CH2CH2Br KOC(CH3)3 tert-butyl alcohol, 40°C CH3(CH2)15CH2CH2OC(CH3)3 + CH3(CH2)15CH=CH2 (13%) (87%) When is Substitution Favored? Given that the major reaction of a secondary alkyl halide with an alkoxide ion is elimination by the E2 mechanism, we can expect the proportion of substitution to increase with: 1) decreased crowding at the carbon that bears the leaving group. 2) decreased basicity of the nucleophile. Weakly Basic Nucleophile Weakly basic nucleophile increases substitution relative to elimination secondary alkyl halide + weakly basic nucleophile CH3CH(CH2)5CH3 Cl KCN pKa (HCN) = 9.1 DMSO CH3CH(CH2)5CH3 (70%) CN Weakly Basic Nucleophile Weakly basic nucleophile increases substitution relative to elimination secondary alkyl halide + weakly basic nucleophile I NaN3 N3 (75%) pKa (HN3) = 4.6 Tertiary Alkyl Halides Tertiary alkyl halides are so sterically hindered that elimination is the major reaction with all anionic nucleophiles. Only in solvolysis reactions does substitution predominate over elimination with tertiary alkyl halides. (CH3)2CCH2CH3 Example Br CH3 CH3CCH2CH3 CH3 CH3 + CH2=CCH2CH3 + CH3C=CHCH3 OCH2CH3 ethanol, 25°C 64% 36% 2M sodium ethoxide in ethanol, 25°C 99% 1% 8.12 Nucleophilic Substitution of Alkyl Sulfonates Leaving Groups We have seen numerous examples of nucleophilic substitution in which X in RX is a halogen. Halogen is not the only possible leaving group, though. Other RX Compounds O ROSCH3 O Alkyl methanesulfonate (mesylate) O ROS CH3 O Alkyl p-toluenesulfonate (tosylate) undergo same kinds of reactions as alkyl halides Preparation Tosylates are prepared by the reaction of alcohols with p-toluenesulfonyl chloride (usually in the presence of pyridine). ROH + CH3 SO2Cl pyridine O ROS O CH3 (abbreviated as ROTs) Tosylates Undergo Typical Nucleophilic Substitution Reactions H KCN H CH2OTs ethanolwater CH2CN (86%) The best leaving groups are weakly basic. Table 8.8 Approximate Relative Leaving Group Abilities Leaving Group F– Cl– Br– I– H2O TsO– CF3SO2O– Relative Rate 10-5 1 10 102 101 105 108 Conjugate acid pKa of of leaving group conj. acid HF HCl HBr HI H3O+ TsOH CF3SO2OH 3.5 -7 -9 -10 -1.7 -2.8 -6 Table 8.8 Approximate Relative Leaving Group Abilities Leaving Group F– Cl– Br– I– H2O TsO– CF3SO2O– Relative Rate 10-5 1 10 102 101 105 108 Conjugate acid pKa of of leaving group conj. acid HF HCl HBr HI H3O+ TsOH CF3SO2OH 3.5 -7 -9 -10 -1.7 -2.8 -6 Sulfonate esters are extremely good leaving groups; sulfonate ions are very weak bases. Tosylates can be Converted to Alkyl Halides CH3CHCH2CH3 OTs NaBr DMSO CH3CHCH2CH3 Br (82%) Tosylate is a better leaving group than bromide. Tosylates Allow Control of Stereochemistry Preparation of tosylate does not affect any of the bonds to the chirality center, so configuration and optical purity of tosylate is the same as the alcohol from which it was formed. H H CH3(CH2)5 TsCl C CH3(CH2)5 C OH pyridine H3C H3C OTs Tosylates Allow Control of Stereochemistry Having a tosylate of known optical purity and absolute configuration then allows the preparation of other compounds of known configuration by SN2 processes. H H CH3(CH2)5 C Nu– OTs (CH2)5CH3 Nu C SN2 H3C CH3 Tosylates also undergo Elimination CH3CHCH2CH3 OTs NaOCH3 CH3OH heat CH2=CHCH2CH3 + CH3CH=CHCH3 E and Z Secondary Alcohols React with Hydrogen Halides Predominantly with Net Inversion of Configuration H CH3 Br C 87% H H3C (CH2)5CH3 HBr C CH3(CH2)5 OH H 13% H3C C CH3(CH2)5 Br Secondary Alcohols React with Hydrogen Halides with Net Inversion of Configuration H Most reasonable mechanism is SN1 with front side of carbocation shielded by leaving group. CH3 Br C 87% H H3C (CH2)5CH3 HBr C CH3(CH2)5 OH H 13% H3C C CH3(CH2)5 Br Rearrangements can Occur in the Reaction of Alcohols with Hydrogen Halides OH HBr Br + Br 93% 7% Rearrangements can Occur in the Reaction of Alcohols with Hydrogen Halides HBr OH 7% + + 93% Br – Br + Br Br –