* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Dynamics of Protein Metabolism in the Ruminant
Clinical neurochemistry wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Paracrine signalling wikipedia , lookup
Gene expression wikipedia , lookup
Biosynthesis wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Genetic code wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Point mutation wikipedia , lookup
Expression vector wikipedia , lookup
Ancestral sequence reconstruction wikipedia , lookup
Magnesium transporter wikipedia , lookup
Biochemistry wikipedia , lookup
Metalloprotein wikipedia , lookup
Interactome wikipedia , lookup
Bimolecular fluorescence complementation wikipedia , lookup
Western blot wikipedia , lookup
Protein purification wikipedia , lookup
Protein–protein interaction wikipedia , lookup
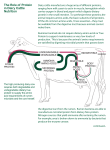
Dynamics of Protein Metabolism in the Ruminant Microbial Protein Ruminally synthesized microbial protein supplies 50% OR MORE of absorbable AA’s when rations are balanced properly. Digestibility of microbial protein is about 85% and has an EAA profile similar to that of lean body tissue and milk. Microbial protein EAA is constant and not influenced by diet change. 11/12/02 7 2.2 2.3 Analysis of Dietary Protein Crude protein (CP %) = total N (%) 6.25 Factor is based on 16% N in protein. True protein varies between 13 to 19% N. Source oilseed proteins cereal proteins meat or fish alfalfa true microbial protein %N in protein 18.5 17.0 16.0 15.8 15.0 Conversion factor 5.40 5.90 6.25 6.33 6.67 Not all N in protein is present as true protein. Research shows... Researchers at Cornell University reported that in 67 lactation trials evaluating the UIP approach, milk production significantly increased in 19%, decreased in 9% and did not change in 73%. Emphasized the need to consider the quality of UIP sources and AA balance when utilizing the protein partitioning system. 11/12/02 14 Cornell Protein Partitioning System This system, introduced in the late 1980’s differentiated between the various N forms by breaking the protein content of feed down into fractions based on: – availability – site of digestion – degree of rumen solubility 11/12/02 10 Protein Categories (Based on the Cornell system) CRUDE PROTEIN AVAILABLE PROTEIN DEGRADEABLE PROTEIN UNAVAILABLE PROTEIN (ADF-N) UNDEGRADEABLE PROTEIN METABOLIZABLE PROTEIN RAPIDLY SOLUBLE PROTEIN 11/12/02 UNUSED PROTEIN SLOWLY SOLUBLE PROTEIN 11 Classification of protein and nitrogen fractions in feedstuffs Total Borate Buffer Sol A B1 Insol B2 B3 C Neutral Detergent Sol A1 B1 B2 Insol B3 C Acid Detergent Sol A1 B1 B2 B3 Insol C Crude Protein True protein (60 to 80%) Essential amino acids Arginine (Arg) Histidine (His) Isoleucine (Ile) Leucine (Leu) Lysine (Lys) Methionine (Met) Phenylalanine (Phe) Threonine (Thr) Tryptophan (Trp) Valine (Val) Non-essential amino acids Alanine (Ala) Asparagine (Asn) Aspartic acid (Asp) Cysteine (Cys) Glutamic acid (Glu) Glutamine (Gln) Glycine (Gly) Proline (Pro) Serine (Ser) Tyrosine (Tyr) Non-protein nitrogen Amides Amines Amino acids Peptides Nucleic acids Nitrates Ammonia Urea Lignified nitrogen Classification of Protein and Nitrogen in Feedstuffs Fraction A1 Composition NH3, NO3, AA, peptides B1 True soluble protein Globulins and some albumins Most albumins and glutelins Prolamines, extensins B2 B3 C Denatured proteins Heat damaged protein and N bound to lignin Ruminal Degradation (%/h) Instantaneous 200 to 300 Intestinal Digestion(%) None reaches intestine 100 5 to 15 100 0.1 to 1.5 80 0 0 Log of % nutrient remaining Calculations Protein Fraction A, B1 B2 B3 C Hours Calculate slope (change per hour) of each line. Slope = kd, has units of % of pool remaining that is lost per hour. Terms for describing nitrogen components of feedstuffs Degradable Intake Protein (DIP): dietary crude protein degraded in the rumen. Undegraded intake protein (UIP): dietary crude protein that is not degraded in the rumen and escapes or bypasses the rumen to the intestine. It is largely true protein but also contains ADFIP. Soluble protein (SolP): Contains non-protein nitrogen, amino acids and peptides. Soluble protein is degraded instantaneously in the rumen. Terms for describing nitrogen components of feedstuffs Non-protein nitrogen (NPN): Includes amides, amines, amino acids, some peptides, nucleic acids, nitrates, urea, ammonia. Degraded instantaneously in the rumen. Acid detergent fiber insoluble protein (ADFIP): Consists of heat damaged protein and nitrogen associated with lignin. Resists ruminal fermentation and is indigestible in the small intestine. Protein content of common feedstuffs Feedstuff CP %DM DIP %CP UIP %CP Alfalfa silage Barley silage Corn silage Alfalfa hay Timothy hay Barley straw Barley grain 19.5 11.9 8.6 22 10.8 4.4 13.2 92 86 77 84 73 30 67 8 14 23 16 27 70 33 SolP %CP 50 70 50 28 25 20 17 NPN ADFIP %SolP %CP 100 100 100 93 96 95 29 15 6.1 9 14 5.7 65 5 Protein content of protein supplements Plant sources CP DIP %DM %CP UIP %CP SolP NPN ADFIP %CP %SolP %CP Canola meal Soybean meal Soypass* Brewer’s grains Corn distiller’s gr. Corn gluten meal 40.9 52.9 52.6 29.2 30.4 66.3 32.2 20 66 65.9 73.7 59 32.4 33 6.8 4 6 4 67.9 80 34 34.1 26.6 41 *Commercial product: LignoTech USA, Inc. 65 27 50 75 67 75 6.4 1 1 12 18 2 Protein content of protein supplements CP DIP %DM %CP UIP %CP SolP NPN ADFIP %CP %SolP %CP 93.8 85.8 67.9 50 25 30 40 47 75 70 60 53 5 9 21 16.1 0 89 0 93.8 1 32 1 4.9 Non-protein nitrogen sources Urea 291 100 0 100 100 0 Animal sources Blood meal Feather meal Fishmeal Meat and bone Ruminally Protected Protein A nutrient(s) fed in such a form that provides an increase in the flow of that nutrient(s), unchanged, to the abomasum, yet is available to the animal in the intestine Methods to decrease the rate and extent of ruminal degradation involved the use of heat, chemical agents, or combination of both Heat Processing Heat processing decrease rumen protein degradation by denaturation of proteins and by the formation of protein-CHO (Millard reactions) and protein cross-links. Commercial methods that rely solely on heat include: cooker-expeller, roasting, extrusion, pressure toasting, and micronization. Heat processing reduced fraction A, increases fraction B, and C, and decreases in the fractional rates of degradation of the fraction B Heat Processing cont. Over heating also causes significant losses of lysine, cysine, and arginine. Among those AA, lysine is the most sensitive to heat damage and undergoes both destruction and decreased availability Chemistry of the Maillard reaction between reducing sugars and lysine residues during heat treatment of proteins Heat Processing Careful control of heating conditions is required to optimize the content of digestible RUP. Under heating results in only small increase in digestible RUP. . Over heating reduces the intestinal digestibility of RUP through the formation of indigestible Millard products and protein complexes. Chemical Treatment Chemical treatment of feed proteins can be divided into three categories: 1) chemicals that combine with and introduce cross-links in proteins, (2) chemicals that alter protein structure by denaturation (e.g., acids, alkalis, and ethanol), and (3) chemicals that bind to proteins but with little or no alteration of protein structure (e.g., tannins). Chemical Treatment cont. For a variety of reasons, often including less than desired levels of effectiveness, use of chemical agents as the sole treatment for increasing the RUP content of feed proteins has not received commercial acceptance. A more effective approach involving “chemical” agents has been to combine chemical and heat treatments. An example of this approach is the addition of lignosulfonate, a byproduct of the wool pulp industry that contains a variety of sugars (mainly xylose), to oilseed meals before heat treatment. Chemical Treatment cont. The combined treatments enhance nonenzymatic browning (Millard reactions) because of the enhanced availability of sugar aldehydes that can react with protein. Characterization of Protein Sources Common protein supplements that are high in RUP are: Fish meal Meat and bone meal (MBM) Feather meal (FtM) Blood meal (BM) Corn gluten meal (CGM) Distillers dried grains (DDG) DDG with solubles (DDGS) Brewers dried grains (BDG) Brewers wet grains (BWG) Nitrogen transactions in the rumen Sources of nitrogen in the rumen Dietary crude protein (true protein and NPN). Recycled microbial protein (bacteria and protozoa). Endogenous N (urea, abraded epithelial cells, salivary proteins). Degradation of nitrogenous compounds by ruminal microorganisms Bacteria 30 to 50% of the bacteria are proteolytic. Most species have some activity with the exception of the main cellulolytic bacteria (Fibrobacter succinogenes, Ruminococcus flavefacians, R. albus). Major proteolytic bacteria: Ruminobacter amylophilus, Butyrivibrio Fibrisolvens and Prevotella ruminicola. P. ruminicola is the most numerous proteolytic bacteria (> 60% of ruminal bacteria) with strains that occur on both roughage and mixed roughageconcentrate diets. Bacteria cont’d R. amylophilus is the most active proteolytic bacteria. Important on starch-based diets. Breakdown of both soluble and insoluble protein in the rumen. Protozoa Minor involvement in soluble protein breakdown. Engulf and hydrolyze particulate proteins and bacteria. Predatory activity of protozoa against rumen bacteria contributes to bacterial protein degradation and turnover in the rumen. Fungi Minor role in protein degradation. PROTEIN D. ruminantium, B. fibrisolvens, E. caudatum Clostridium spp, E. simplex, E. budayi E. caudatum ecaudatum, E. ruminantium, E. maggii Fusobacterium spp., E. medium L. multipara O. caudatus, P. ruminicola P. multivesiculatum, R. amylophilus, S. ruminantium O. joyonii, N. frontalis, S. bovis, P. communis OLIGOPEPTIDES Dipeptidyl peptidase S. bovis, R. amylophilus, P. ruminicola DIPEPTIDES D. ruminantium, E. caudatum F. succinogenes, M. elsdenii, P. ruminicola Isotricha spp., L. multipara, S. ruminantium Dipeptidase AMINO ACIDS C. aminophilum, C. sticklandii P. anerobius, B. fibrisolvens, P. ruminicola M. elsdenii, S. ruminantium, E. caudatum Isotricha spp. AMMONIA Properties of ammonia producing bacteria High Numbers Low Activity Low Numbers High Activity Butyrivibrio fibrisolvens Megasphaera elsdenii Prevotella ruminicola Selenomonas ruminantium Streptococcus bovis Clostridium aminophilum Clostridium sticklandii Peptostreptococcus anaerobius > 109 per ml 107 per ml 10 to 20 nmol NH3 min-1 (mg protein) 300 nmol NH3 min-1 (mg protein) Breakdown of NPN in the rumen Major sources of NPN include: dietary NPN, and recycled urea. Extremely rapid and releases ammonia. Major end product of protein degradation in the rumen Ammonia Influence of diet on proteolysis Concentrate Increase in total microbial population, including several of the more active protein degrading bacteria which are also amylolytic (Prevotella rumincola, Ruminobacter amylophilus and Streptococcus bovis). Fresh forage Increase in the proportion of proteolytic bacteria relative to total microbial population. Microbial protein synthesis in the rumen Factors Influencing Microbial Protein Synthesis Ammonia Most important source of N for bacterial protein synthesis. 50 to 80% of bacterial N is derived from ammonia. Bacteria hydrolyzing structural carbohydrates utilize ammonia as N source. Several mechanisms for the uptake of ammonia: high affinity, low Km (ammonia concentration) enzyme system glutamate synthetase - glutamate synthase (GS-GOGAT) lower affinity, higher Km system NADP-glutamate dehydrogenase (NADP-GDH), NAD-GDH and alanine dehydrogenase. Minimum level of ammonia is necessary for maximum growth and efficiency (5 mg/100 ml of rumen fluid). Peptides and amino acids 20 to 50% of ruminal microbial N is derived from this pool. Supplying preformed peptides and amino acids spares the cost associated with synthesizing amino acids. Rapidly fermenting organisms, bacteria hydrolyzing non-structural carbohydrates (starch, pectin, sugars), utilize peptides, amino acids and ammonia. Availability of peptides improves microbial growth. Synchronization of protein and carbohydrate degradation Microbial protein synthesis is maximized when the release of N from protein occurs with the release of energy from the degradation of carbohydrates. Fractional Outflow Rates Increasing the rate of passage removes the more mature organisms, reducing the median age of the microbes. Reduces the amount of energy expended on maintenance so more energy can be used for growth. Efficiency of Microbial Growth 14 BCP/100 gm TDN 12 10 8 6 Rate of passage pH 4 2 0 55 Diet % of TDN (DOM) 70 Effect of dilution rate on YATP. Reduces the amount of intraruminal N recycling (microbial protein turnover). Intraruminal nitrogen recycling Turnover of bacteria and protozoa. 30 to 55% of bacterial N 75 to 90% of protozoal N Causes of microbial N recycling Engulfment and subsequent digestion of bacterial cells by protozoa Lysis due to autolytic enzymes, bacteriocins, or other soluble compounds in response to nutrient deprivation or interspecies competition Activity of bacteriophages and mycoplasmas. Ammonia accumulation in the rumen Ammonia concentration exceeds the capacity of the ruminal bacteria to utilize it. Absorbed across the ruminal wall into the blood where it is transported to the liver and metabolized to urea. Urea is filtered by the kidney and excreted in urine as waste N. In addition to poor N retention, the synthesis of urea from ammonia also has an energetic cost (12 kcal/g N) to the animal. Urea recycling Blood urea originates from the endogenous metabolism of tissue protein, the deamination of excess absorbed amino acids and the absorption of ruminal ammonia. Recycled to the rumen primarily through the rumen wall and to a lesser extent via saliva (approx 15% of urea recycled to the rumen is via saliva) Facultative microorganisms located on the rumen epithelium wall have urease activity Factors involved in increasing the permeability of the rumen wall to urea Composition of microbial protein reaching the intestine Hay sheep 1 sheep 2 Hay and Conc sheep 1 sheep2 N in rumen digesta, g Fungi .21 .60 .42 Protozoa 8.0 5.86 18.3 Bacteria 11.5 10.4 9.03 N in rumen digesta, % of total microbial N Fungi 1.1 3.6 1.5 Protozoa 40.7 34.7 65.9 Bacteria 58.3 61.7 32.6 N in duodenal digesta, g Fungi .10 .22 .21 Protozoa .55 1.08 1.83 Bacteria 13.1 14.6 10.1 N in duodenal digesta, % of total microbial N flow Fungi .73 1.4 1.7 Protozoa 4.0 6.8 15.1 Bacteria 95.3 91.8 83.2 .69 11.5 8.07 3.4 56.7 39.8 .49 1.63 15.9 2.7 9.0 88.2 Undegraded dietary protein Protein that escapes microbial degradation passes to the lower digestive tract where it will be largely degraded. Only the very refractive N component such as N bound to lignin or products of the Maillard reaction will not be degraded. Benefit to the animal of supplying UIP will depend on the provision of essential amino acids that are required in excess of what is supplied by microbial protein. Protein digestion in the abomasum HCl Denatured protein disruption of non-covalent bonds uncoiling of protein Protein HCl Pepsinogen (inactive) Denatured protein Pepsin (hydrolysis bonds at carboxylic end of aromatic AA and Leu) Pepsin pH 1.6 to 3.2 Small polypeptides few amino acid Secretion and activation of pancreatic and intestinal proteolytic enzymes Polypeptides Short peptides AA Intestinal endocrine cell CCK and Secretin Pancreatic acinar cell Cholecystokinin (CCK) Intestinal mucosal cell Enterokinase Trypsinogen Chymotrypsinogen Proelastase Procarboxypeptidase Trypsin Chymotrypsin Elastase Carboxypeptidase Sites of hydrolysis of proteolytic enzymes Pancreas Trypsin Chymotrypsin Elastase Carboxypeptidase Dibasic AA (Arg, Lys), C-terminal end Aromatic C terminal peptides Neutral C terminal peptides C-terminal end Intestine Enteropeptidase N-terminal end Digestion in the small intestine Pancreatic and intestinal proteases Polypeptides Oligopeptides Dipeptides Tripeptides Amino acids Dipeptides Tripeptides Intestinal di- and tripeptidases (cell membrane and cytosol) Amino acids Protein absorption Small intestine Major site of absorption Amino acids absorbed in the ileum Dipeptides and tripeptides absorbed in the jejunum Active transport (energy dependent) 9 carrier systems for amino acids specific for certain amino acids Protein metabolism Intestinal cell Glu, Asp, Gln metabolized by intestinal cell provides 40% of energy requirements Liver protein synthesis synthesis of non-essential amino acids C-skeletons catabolized for energy and the amine group metabolized to urea Nitrogen metabolism in the large intestine N supplied to the lower tract comes from the recycling of urea and other endogenous protein (sloughed epithelial cells, enzymes and glycoproteins of mucus). Energy substrates come from the residual fermentable fibre, the glycocalyx of rumen microorganisms, starch and other polysaccharides that have resisted rumen and enteric digestion. As the amount of fermentable energy from the diet reaching the lower tract increases, microbial synthesis increases and fecal N excretion increases. Routes of nitrogen excretion Urine (urea) Endogenous urinary N from the catabolism of tissue proteins Absorption and metabolism of excess ruminal ammonia. Catabolism of excess absorbed amino acids Feces Microbial N synthesized in and passed from the large intestine. Sloughed cells and secretions of the GI tract. Undigested unabsorbed dietary protein. Feed Undegradable in rumen Indigestible Rumen Digestible Peptides amino acids Degradable in rumen NPN NH3 Energy Plasma urea Microbial N Small intestine Peptides amino acids NH3 Endog N Energy Tissues Maintenance Growth Conceptus Lactation Wool NH3 Large intestine Microbial N Feces Endog N Urine Meeting the protein requirements of ruminant animals Degradable intake protein in the rumen for ruminal microorganisms to maximize digestibility of the diet and feed intake. Absorbable essential amino acids at the intestine from the digestion of microbial protein produced in the rumen and dietary intake protein that escapes rumen fermentation. This is important to the dairy producer because... You are looking for rations that will support higher milk production levels. You are being forced to place more emphasis on milk protein production. With high feeding costs and low milk prices, you are trying to meet the N needs of ruminal fermentation and the AA requirements with a minimum concentration of CP in the diet. 11/12/02 17 The goal should be... To provide the appropriate quantities and balance of AA’s to the intestine for utilization keeping in mind both the needs of the animal and the economics of the situation. i.e. Increase UIP or increase microbial protein. Which is more economical? 11/12/02 18 Optimizing Protein and AA Nutrition Maximize dry matter intake (DMI) – positive correlation between DMI, microbial protein synthesis and AA flow to the gut – as DMI’s increase, the rate of passage of digesta also increases 11/12/02 19 Optimizing Protein and AA Nutrition Maximize Microbial Protein Synthesis – a combination of high rumen available carbohydrate with high rumen available protein will maximize bacterial protein production and hence maximize rumen microbial protein delivery to the lower gut. 11/12/02 20 Defaunation (protozoal removal) Removal of protozoal predation of bacteria. Increases substrates (starch) available for fermentation and growth by bacteria. Increases amount of bacterial protein synthesized in the rumen. Increases the flow of microbial protein from the rumen. Reduction in ammonia concentration. Chemistry of the Maillard reaction between reducing sugars and lysine residues during heat treatment of proteins Rumen Protected Amino Acids (RPAA) Commercial products now exist which allow nutritionist to raise intestinal levels of methionine and lysine. Basically “by-pass” AA’s – Some RPAA’s are more resilient than others and will not degrade in the rumen – Some RPAA’s will be more resistant to mechanical and thermal stresses at the mill 11/12/02 15