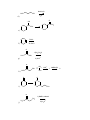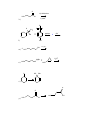* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Mock Exam One
Physical organic chemistry wikipedia , lookup
Metal carbonyl wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Kinetic resolution wikipedia , lookup
Ene reaction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Elias James Corey wikipedia , lookup
Discodermolide wikipedia , lookup
Stille reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Aldol reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Asymmetric induction wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
1.) Which of the following best accounts for the following statement? Alcohols have higher boiling points and higher water solubility than alkanes of similar molecular weight. a.) The bond angle around the oxygen of an alcohol is < 109.5°. b.) Alcohols contain a polar covalent bond that an alkane does not have. c.) The strongest intermolecular force present in an alcohol is hydrogen bonding while the strongest intermolecular force present in alkanes is London dispersion. d.) Alcohols always have a higher molecular weight than alkanes. 2.) Which of the following would yield a secondary alcohol after the indicated reaction, following hydrolysis if necessary? a.) LiAlH4 and a Ketone b.) CH3CH2MgBr and an Aldehyde c.) 2-butene and Hg(OAc)2, H2O followed by NaBH4 d.) All of these. 3.) Which of the following terms best describes the reactive nature of the Grignard reagent? a.) Carbocation d.) Free Radical b.) Electrophile e.) Nucleophile c.) Carbene 4.) The compound shown below is best classified as: N OH a.) Carbinolamine c.) Enamine b.) Hydrazone d.) Oxime 5.) Through what mechanism is a tertiary alcohol converted to a tertiary alkyl halide? a.) SN2 b.) SN1 c.) E1 d.) E2 e.) This reaction is not possible. 6.) Which of the following could be used in the presence of a Grignard reagent? NH2 S b.) H3C a.) H c.) H3C CH f.) none of the above CH3 OH e.) H3C d.) 7.) A carbene carbon is in an hybridized and the lone pair of electrons resides orbital. a.) sp3: sp3 b.) sp3: p c.) sp2: sp2 d.) sp2: p e.) sp: p 8.) Which of the following does not undergo oxidation with CrO3 and H3O+? a.) Butylalcohol c.) sec-butylalcohol b.) Isobutylalcohol d.) tert-butylalcohol 9.) What is the best choice of reagents to achieve the following transformation? CH3 CH3 OH H3C a.) HCl/ether b.) Cl2/hv Cl H3C c.) SOCl2/pyridine d.) Cl2/H2O 10.) Which of the following statements is not true? a.) The carbon atom of a carbonyl group is electrophilic. b.) In general, aldehydes are more reactive than ketones. c.) Nucleophilic addition to carbonyl groups can be catalyzed by acid or base. d.) Addition of a nucleophile to a carbonyl group changes the hybridization of the carbonyl carbon from sp3 to sp2. 11.) What type of reaction takes place upon treatment of a ketone with HCN to form a cyanohydrin? a.) nucleophilic addition c.) electrophilic addition b.) nucleophilic substitution d.) electrophilic substitution 12.) Acid catalyzed dehydration of an alcohol proceeds through what mechanism? a.) SN1 b.) SN2 c.) E1 d.) E2 e.) This reaction is not possible. 13.) Which of the following statements about imine and enamine formation is false? (choose all that apply) a.) Reacting a ketone or an aldehyde with a primary amine will yield an imine. b.) Reacting a ketone or an aldehyde with a secondary amine will yield an enamine. c.) Imine and enamine formation should be carried out in highly acidic conditions. d.) Imines can be reduced to primary amines. e.) Enamines can be reduced to secondary amines. 14.) During a Tollen’s test, the is oxidized and the a.) silver: aldehyde is reduced. b.) aldehyde: silver 15.) 2-methyl-3-pentanol is classified as a . a.) primary alcohol c.) secondary alcohol b.) tertiary alcohol d.) none of the above 16.) The substance formed on addition of water to an aldehyde or ketone is called a hydrate or a: a.) vicinal diol c.) geminal diol e.) acetal b.) ketal d.) none of the above 17.) Which of the following compounds could serve as the reagents X and Z in the following sequence? (C6H5)3P X CH3CH2CH2CH2Li CH3 Z THF CH3 H3C H3C H3C H3C CH3 Br 1 X Z A 1 5 B 1 4 C 2 4 D 2 5 E 3 4 Br Br H3C CH3 CH3 2 3 H3C O H3C H 4 O H3C CH3 5 18.) When the carbonyl group of a neutral ketone is protonated: a.) the resulting species becomes more electrophilic. b.) the resulting species is activated toward nucleophilic attack. c.) subsequent nucleophilic attack on the resulting species is said to occur under acid-catalyzed conditions. d.) the resulting species has a positive charge. e.) All of the above. 19.) What is the IUPAC name of the following compound? CH3 H3C OH H3C a.) (E)-2-methyl-3-penten-2-ol c.) (Z)-2-methyl-3-penten-2-ol b.) (E)-4-methyl-2-penten-4-ol d.) (Z)-2-methyl-4-penten-2-ol 20.) Which of the following is/are secondary alcohols? H3C OH OH H3C H3C a.) OH CH3 CH3 1 OH CH3 H3C 3 2 only one b.) only three 4 c.) two and four d.) one, two, three, and four 21.) Secondary alcohols can be made by the reaction of Grignard reagents with: a.) formaldehyde c.) ethylene oxide b.) ketones d.) aldehydes other than formaldehyde 22.) The lower the pKa, the the acid. 23.) The nucleophile in the reduction of carbonyl compounds by LiAlH4 and NaBH4 is . 24.) Aldehydes are generally more reactive toward nucleophiles than ketones. Provide two reasons that explain this observation. 25.) Predict the product for the following reaction. Show the mechanism including electron movement with arrows and all intermediates that accounts for the formation of the product. H3C H3C HBr OH 26.) Predict the products for the following reaction. Show the mechanism including electron movement with arrows and all intermediates that accounts for the formation of each possible product. Determine which products are major and minor and explain your choices. CH3 OH CH3 H3 O + Heat 27.) Outline a Wittig synthesis of the following molecule starting with a ketone and an alkyl halide. List all necessary reagents and show the complete mechanism. 28.) Give the reagents that are responsible for the following transformation. Show the mechanism that accounts for the product. O H3C OCH 2CH 2CH 3 CH3 H3C CH3 OCH 2CH 2CH 3 30.) Show what amines and carbonyl compounds combine to give the following compounds. CH3 NH N NH2 N O a.) CH3 b.) OH N N c.) NH2 d.) 31.) For each of the following reactions, provide the structure of the missing reagents, reactants, or products. 1.)C 6H5MgBr OH ? 2.)H 3O a.) ? b.) + H2NNH 2 KOH PCC OH CH 2Cl 2 c.) Na 2Cr 2O7 H3C OH H3 O d.) + O OH CH3 CH3 e.) CH2I2 Zn,CuCl f.) O 1.)CH 3CH 2Li H3C CH3 2.)H 3O g.) + O CH3 h.) H3C + NH2 H3O + 1 CH3 O O CH3 i.) O 1.)NaBH 4,ethanol CH3 H3C j.) 2.)H 3O + NaBH 3CN 2 O 1.)LiAlH4,ether H3C H 2.)H3O k.) + Cl H O OH pyridine A CN - CH3 OH 2 HC 3 MgBr n.) O + l.) m.) S CH3 H3C O Li (s) + H3O + O H3C HO CH3 o.) O O H3C p.) H3C H OH PBr3 OH q.) ether Br NaSH r.) H3C O s.) H3C + H H3C O HNO 3 heat t.) O H H u.) 1.) NaBH 4,ethanol 2.) H 3O + CH3 H3C N v.) 1.) LiAlH 4, ether 2.) H 3O OH O Br2 H3C w.) CH3 - OH + - C H3 O + O Br2 H CH3 x.) H3 O + O H CH3 Ag2O NH3 y.) H3C H CHCl 3 KOH H z.) CH3 32.) Determine the products of the following reactions. Show the mechanism. O CH3 H3C O a.) O 2 b.) CH3 H3C NaOH NaOH 33.) Identify the missing products in the synthesis shown below. O CH3 (C 6H5)3P=CH 2 1.)BH 3,THF THF 1.)CH 3MgBr PCC 1 3 2 2.)H 2O2,OH - 4 CH 2Cl 2 2.)H 3O + 34.) Complete the following synthesis problems. Show all reagents, reaction conditions, and intermediate products. O a.) OH O H3C H O H3C H O H H O b.) CH2 O H c.) N