* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download CH - UTH e
Discodermolide wikipedia , lookup
Aromaticity wikipedia , lookup
Homoaromaticity wikipedia , lookup
Elias James Corey wikipedia , lookup
Petasis reaction wikipedia , lookup
Asymmetric induction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Hydroformylation wikipedia , lookup
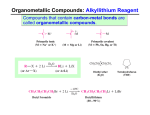
Organometallic O lli Compounds p Organometallic g Compounds p • Organometallic compound: a compound that contains a carbon-metal carbon metal bond • In this chapter, we focus on organometallic compounds of Mg, Li, and Cu – these classes illustrate the usefulness of organometallics in modern synthetic organic chemistry – they illustrate how the use of organometallics can bring about transformations that cannot be accomplished li h d in i any other h way Organometallic Reagents The Key Concepts: y p Make a carbon negatively charged/polarlized so it is nucleophilic. h d/ l li d it i l hili Reaction with electrophilic carbons can p make carbon‐carbon bonds. This is a Big g Deal! The First Organometallic Reagents… Grignard Reagents • Discovered by Victor Grignard in 1900 – Key factors are ethereal solvent and water‐free conditions t f diti • Awarded Nobel Prize in 1912 Victor Grignard Grignard, Victor , 1871–1935, French chemist. He shared the 1912 Nobel Prize in Chemistry for his work in organic synthesis based on his discovery (1900) of the Grignard Reagent. He taught at the Univ. of Nancy (1909 Grignard Reagent. He taught at the Univ. of Nancy (1909–19) 19) and at the Univ. of and at the Univ. of Lyons (from 1919 until the end of his career). Grignard Reagents • Grignard reagent: an organomagnesium compound – prepared by addition of an alkyl, alkyl aryl, aryl or alkenyl (vinylic) halide to Mg metal in diethyl ether or THF Br 1B 1‐Bromobutane b t + Mg B + Mg Br M Bromobenzene ether MgBr B t l Butylmagnesium bromide i b id (an alkyl Grignard reagent) ether et e M B MgBr Phenylmagnesium bromide (an aryl Grignard reagent) An Alternative to Grignard g Reagents g are Alkyl y Lithiums Both are prepared from alkyl, vinyl, and aryl halides under anhydrous conditions dry ether + B Br Mgo n-butyl bromide M B MgBr or tetrahydrofuran (THF) recall: THF = Grignard Reagent O dry ether Br n-butyl bromide + 2 Lio or tetrahydrofuran (THF) Li alkyl lithium + LiBr Grignard and Organolithium Reagents • Given the difference in electronegativity between carbon and magnesium (lithium), the δ‐ and Mg (C‐ g ( ‐Li) bond is polar covalent, with C ) p C‐Mg (C Mgδ+ ( Liδ+ ) – Grignard Grignard and organolithium reagents behave like and organolithium reagents behave like carbanions • Carbanion: an anion in which carbon has an unshared pair of electrons and bears a negative charge δ- δ- δ+ Li δ+ MgBr Grignard Reagent alkyl lithium Grignard and Organolithium Reagents • Carbanion: an anion in which carbon has an unshared pair of electrons and bears a negative g charge – Carbanions are strong bases Carbanions are strong bases‐‐ ‐‐they are easily they are easily q quenched by even very weak acids (water, alcohols, y y ( amines, amides, carboxylic acids, even terminal alkynes). A limitation to utility! alkynes). A limitation to utility! pKa = ca. 50 δδ+ MgBr Grignard g Reagent g + δ- δ+ OH pKa = 16 alkane corresponding to original alkyl halide + O- MgBr+ Grignard and Organolithium Reagents • Carbanion: an anion in which carbon has an unshared pair of electrons and bears a negative g charge – Carbanions are strong bases Carbanions are strong bases‐‐ ‐‐they are easily they are easily q quenched by even very weak acids (water, alcohols, y y ( amines, carboxylic acids, amides, even terminal alkynes). A limitation to utility! alkynes). A limitation to utility! pKa = ca. 50 δδ+ Li δ- δ+ NH2 + alkane corresponding to original alkyl halide + alkyl lithium pKa = 40 NH- Li+ Limitations • Can’t make Grignards with acidic or electro‐philic f functional groups present in the molecule: i l i h l l • R2NH pKa 38‐40 Terminal Alkynes Terminal Alkynes pKa 25 ROH pKa 16‐18 Carbonyls & Nitros pKa 11‐27 Grignard and Organolithium Reagents • Carbanion: an anion in which carbon has an unshared pair of electrons and bears a g charge g negative – Carbanions are also great nucleophiles. This is the reason for their g great utility! y • Key Point: Grignard and Organolithium Reagents • Great nucleophiles that add efficiently to electrophilic carbons, such as epoxides and carbonyl group of aldehydes, ketones and esters However their basicity can be a ketones and esters. However, their basicity can be a limitation! • Epoxides illustrate how many common organic illustrate how many common organic functional groups contain electrophilic carbons Grignard and Organolithium Reagents • Carbanions (nucleophiles) can react with electrophilic carbon centers in favorable cases. The net result is a carbon‐‐carbon bond carbon carbon bond‐‐ ‐‐a big deal! a big deal! • Grignards and organolithium reagents react with many oxygen‐ oxygen o yge ‐co containing electrophiles, but not with alkyl ta g e ect op es, but ot t a y halides. We’llll illustrate this with epoxides. illustrate this with epoxides • We • Recall, acidic protons will “kill” our reagents and/or won’t allow them to be generated in the first place won’t allow them to be generated in the first place Grignard reagents react productively with: formaldehyde to give primary alcohols aldehydes ld h d to give i secondary d alcohols l h l ketones to give tertiary alcohols esters to ggive tertiary y alcohols CO2 to give acids epoxides to give primary alcohols The one we are choosing for the sake of initial illustration Epoxides: p The Example p We Want to Stress New C-C bond δ- MgBr δ+ - O Considered R t Retrosynthetically: + OH O MgBr H O+ 3 Dilute THF OH MgBr + O Detailed Mechanism Highlighting Retention of Stereochemistry New C-C bond Key Points to Note: Attack at least hindered carbon M h i Mechanism iis SN2-like lik iin iinitial iti l step t Single enantiomeric product Chiral center not affected byy reaction Relief of ring strain helps drive reaction H3O+ breaks up initial salt Two More Examples of Additions to Epoxides 1. δδ+ MgBr O δ- New CC C-C bond δ+ 2. HCl, H2O OH Grignard Reagent 1. δδ+ Li Organolithium Reagent O New C-C bond δ- OH δ+ H H H H 2. HCl, H2O Note: Stereochemistry at epoxide retained in product Gilman Reagents • Lithium diorganocopper reagents, known more g commonlyy as Gilman reagents – prepared by treating an alkyl, aryl, or alkenyl lithium compound with Cu(I) iodide diethyl ether 2 CH3 CH2 CH2 CH2 Li + CuI or THF Butyllithium Copper(I) iodide ( CH3 CH2 CH2 CH2 ) 2 Cu - Li Lithium dibutylcopper Lithi dib t l (a Gilman reagent) + + LiI Gilman Reagents • Coupling with organohalogen compounds – form new carbon-carbon bonds by coupling with alkyl and alkenyl chlorides, bromides, and iodides. (Note that this doesn’t work with Grignard or organolithium reagents. THEY ARE TOO BASIC AND DO E2 ELIMINATIONS.) ELIMINATIONS ) R'Br R Br + R2 CuLiBr d iethyl eth er or THF R'-R R R + RCu + LiBr •Example I 1 . Li, Li pent ane 2 . CuI 2 1-Iod od ecane d ie i th h yll ethe h r Br or THF 2-Methyl-1-dodecen e CuLi New C-C bond Gilman Reagents – coupling with a vinylic halide is stereospecific; the configuration of the carbon-carbon double bond is retained I + t rans-1-Iodo-1-nonene rans 1 Iodo 1 nonene 2 CuLi Lithiu m dib utylcopper d iethyl eth er or THF New C-C C C bond trans -5-Tridecen e Gilman Reagents • A variation on the preparation of a Gilman g is to use a Grignard g reagent g with a reagent catalytic amount of a copper(I) salt CH3 (CH2 ) 7 (CH2 ) 7 CH2 Br C C H H (Z)-1-Bromo-9-octadecene Cu+ THF + CH3 ( CH 2 ) 4 MgBr CH3 (CH2 ) 7 (CH2 ) 1 2 CH3 C C H H (Z)-9-Tricos ene (Muscalu re) Gilman Reagents • Reaction with epoxides – regioselective g ring g opening p g ((attack at least New hindered carbon) C-C O OH bond 1 . ( CH 2 =CH) 2 CuLi 2 . H2 O,, HCl S tyrene oxide (racemic) 1-Ph enyl-3-buten-1-ol (racemic) Interim Summary of Introduction to y Organometallic Reagents… Organolithium reagents and Grignard reagents are very basic but also great nucleophiles. They react with basic but also great nucleophiles. They react with epoxides at the less hindered site to give a two‐carbon chain extended alcohol. They do not couple with alkyl‐, aryl‐, or vinyl halides. Gilman reagents react with epoxides as do organolithium reagents and Grignard reagents. H However, they also add to alkyl‐, aryl‐, and vinyl halides th l dd t lk l l d i l h lid to make new C‐C bonds. Feeling Lost? Fortunately, Dr. Iverson will i be back on Monday! Meanwhile…. Back to Grignard Reagents… • Addition of a Grignard reagent to formaldehyde followed by H3O+ gives a 1° alcohol H H - + CH3CH2 MgBr C O H - THF + g CH3CH2 C O MgBr H H3O dil. + H +2 CH3CH2 C OH + Mgg H • This sequence (mechanism) is general and important! Gi Grignard dR Reactions ti - + CH3CH2 MgBr O C O - THF + CH3CH2 C O MgBr O H3O + dil dil. These are valuable and important reactions… Please add to your card stock! +2 CH3CH2 C OH + Mg O Grignard reagents react with esters δ– R R' •• δ+ OCH 3 •• C MgX O •• •• R' diethyl ether R C •• OCH3 •• •• O •• + MgX •• – but species formed is unstable p and dissociates under the reaction conditions to form a ketone Grignard reagents react with esters δ– R R' •• δ+ OCH 3 •• C R' diethyl ether R OCH3 •• •• O •• + MgX •• – MgX O •• •• this ketone then goes on hi k h to react with a second mole of the Grignard mole of the Grignard reagent to give a tertiary alcohol C •• –CH3OMgX g R R' C O •• •• Example O CHCOCH OCH3 2 CH3MgBr + (CH3)2CHC 1. diethyl y ether 2. H3O+ OH (CH3)2CHC CHCCH CH3 CH3 (73 73%) %) Two of the groups attached to the h d h tertiary carbon come from the Grignard from the Grignard reagent Practice OH MgBr O + H H Practice OH MgBr O + O MgBr + H H The Same Chemistry is seen With Organolithium Reagents O H2C CHLi + CH 1. diethyl ether 2. H3O+ CHCH OH CH2 (76 76%) %) Practice O OH MgBr + O C O