* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Consortium for Educational Communication
Fatty acid synthesis wikipedia , lookup
Butyric acid wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
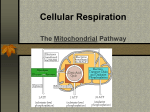
Mitochondrion wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Metalloprotein wikipedia , lookup
Electron transport chain wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Photosynthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Biochemistry wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Citric acid cycle wikipedia , lookup
Consortium for Educational Communication FAQ’S 1. What do you mean by respiration? Ans: In physiology, respiration (often confused with breathing) is defined as the transport of oxygen from the outside air to the cells within tissues, and the transport of carbon dioxide in the opposite direction. This is in contrast to the biochemical definition of respiration which refers to cellular respiration: Cellular respiration is the set of the metabolic reactions and processes that take place in the cells of organisms to convert biochemical energy from nutrients into adenosine triphosphate (ATP), and then release waste products. The reactions involved in respiration are catabolic reactions, which break large molecules into smaller ones, releasing energy in the process as they break high-energy bonds. Respiration is one of the key ways a cell gains useful energy to fuel cellular activity. Chemically, cellular respiration is considered an exothermic redox reaction. The overall reaction is broken into many smaller ones when it occurs in the body, most of which are redox reactions themselves. 2. What are the basic types of respiration? Ans: Cellular respiration may be divided into two categories depending on the availability of oxygen. aerobic respiration: Aerobic respiration requires oxygen in order to generate energy (ATP). Although carbohydrates, fats, and proteins can all be processed and consumed as reactant, it is the preferred method of pyruvate breakdown in glycolysis and requires that pyruvate enter the mitochondrion in order to be fully oxidized by the Krebs cycle. The product of this process is energy in the form of ATP (adenosine triphosphate), by substrate-level phosphorylation, NADH and FADH2. b) Anaerobic respiration is used by some microorganisms in which neither oxygen (aerobic respiration) nor pyruvate or a pyruvate derivative (fermentation) is the final electron acceptor. Rather, an inorganic acceptor such as sulfur is used. Yeast and muscle cells are examples which they can respire anaerobically for a short time. This is also called as oxygen debt. 3. What are respiratory substrates? Name the most common respiratory substrate. Consortium for Educational Communication Ans: Like animals, the plants also require energy for growth and development, metabolism, and other life activities. This energy mainly comes from respiratory breakdown of various substrates. These substrates are called organic nutrients and synthesized inside the plants by anabolic processes (viz., photosynthesis, fat synthesis, protein synthesis, etc.) these organic substances, which are present in large amounts in plant cells and respired completely to CO2 and H2O, are called respiratory substrates. The most commonly used respiratory substrates are carbohydrates. Differentiate between Respiration and Combustion. 4. Ans: Respiration It takes place in air oxygen. It is a single-step process or Enzymes are not involved Energy release is vigorous and not under control energy is released in form of heat and light (a flame can result) Combustion Takes place in cells. While a series of chemical reactions. It is controlled by enzymes. Energy release is slower and under control energy is released in form of heat and an energy-rich compound Differentiate between Glycolysis and Krebs cycle. Ans: Glycolysis is a series of reactions carried out by a group of soluble enzymes located in the cytosol. Chemically, glucose is partly oxidized to produce two molecules of pyruvate (a three carbon compound), a little ATP, and stored reducing power in the form of a reduced pyridine nucleotide, NADH. Both glycolysis and TCA cycle takes place in the inner membrane of mitochondrion. Tricarboxylic acid cycle: When oxygen is present, acetyl-CoA is produced from the pyruvate molecules created from glycolysis. Once acetyl-CoA is formed, two processes can occur, aerobic or anaerobic respiration. When oxygen is present, the mitochondria will undergo aerobic respiration which leads to the Krebs cycle. However, if oxygen is not present, fermentation of the pyruvate molecule will occur. 5. Differentiate between Aerobic respiration and Fermentation Ans: If we compare both anaerobic respiration and anaerobic fermentation, we can say that both of them are completely Consortium for Educational Communication separate from each other as both follow different pathways. In anaerobic respiration, the process involves the transfer of electrons through a system of chain system in the membrane of the cell. Fermentation takes place when a co-enzyme, NADH reduces the pyruvate to form the organic compound. It is the process of getting energy by the oxidation of some compounds like carbohydrates, and by using an endogenous electron-acceptor that is usually an organic compound. The common products of fermentation are lactic acid, ethanol and hydrogen etc. But respiration is the process where electrons are given to an exogenous electron-acceptor, such as oxygen, through the electron transport chain. Another difference between fermentation and respiration is that it is not necessary for fermentation to occur in environments suitable for anaerobic respiration. A good example of this is yeast, which ferments even if oxygen is present, or if sugar is present. Thus, we can easily say that fermentation can occur when the electrons that are present in the coenzymes (NADH derived after glycolysis) are turned back partly to pyruvate. It should also be noted that the electrons are donated to things that have come from the pyruvate cells. There is yet another difference between anaerobic respiration and anaerobic fermentation is the electron acceptor, which is also known as the final electron acceptor. For fermentation, pyruvate is the final electron acceptor. This can be seen in yeast fermentation, which gives alcohol as the final acceptor and does not break further for releasing energy. However, the main purpose of anaerobic respiration is to produce ATP (Adenosine Triphosphate), which is used by a cell for energy purposes. 6. What are the main steps in aerobic respiration? Where does it take place? Ans: The main steos of aerobic respiration are: a) Glycolytic breakdown of glucose to pyruvic acid b) Oxidative decarboxylation of pyruvic acid to acety coA c) Krebs cycle and d) Terminal oxidation and phosphorylation in respiratory Consortium for Educational Communication chain. 8. Explain the role of ETS. Ans: The electron transport chain consists of a collection of electron transport proteins bound to the inner of the two mitochondrial membranes. This system transfers electrons from NADH, produced during glycolysis and the TCA cycle, to oxygen. This electron transfer releases a large amount of free energy in the form of ATP. 9. Distinguish between Glycolysis and Fermentation. Ans: Glycolysis is a series of reactions carried out by a group of soluble enzymes located in the cytosol. Chemically, glucose is partly oxidized to produce two molecules of pyruvate (a three carbon compound), a little ATP, and stored reducing power in the form of a reduced pyridine nucleotide, NADH. Both glycolysis and TCA cycle takes place in the inner membrane of mitochondrion. Without oxygen, pyruvate (pyruvic acid) is not metabolized by cellular respiration but undergoes a process of fermentation. The pyruvate is not transported into the mitochondrion, but remains in the cytoplasm, where it is converted to waste products that may be removed from the cell. This serves the purpose of oxidizing the electron carriers so that they can perform glycolysis again and removing the excess pyruvate. Fermentation oxidizes NADH to NAD+ so it can be re-used in glycolysis. 10. Distinguish between Aerobic respiration and Anaerobic respiration. Ans: oxygen requirement oxidation of sugar energy released end products Occurrence aerobic respiration anaerobic respiration Essential complete oxidation large amount inorganic: CO2 and H2O Nill incomplete oxidation Small amount organic:ethanol or lactic acid in lower organisms (e.g. bacteria and yeast) and vertebrate muscles. in most living Consortium for Educational Communication Distinguish between Glycolysis and Citric acid Cycle. Ans: Glycolysis is a series of reactions carried out by a group of soluble enzymes located in the cytosol. Chemically, glucose is partly oxidized to produce two molecules of pyruvate (a three carbon compound), a little ATP, and stored reducing power in the form of a reduced pyridine nucleotide, NADH. Both glycolysis and TCA cycle takes place in the inner membrane of mitochondrion. Tricarboxylic acid cycle: When oxygen is present, acetyl-CoA is produced from the pyruvate molecules created from glycolysis. Once acetyl-CoA is formed, two processes can occur, aerobic or anaerobic respiration. When oxygen is present, the mitochondria will undergo aerobic respiration which leads to the Krebs cycle. However, if oxygen is not present, fermentation of the pyruvate molecule will occur. In the presence of oxygen, when acetyl-CoA is produced, the molecule then enters the citric acid cycle (Krebs cycle) inside the mitochondrial matrix, and gets oxidized to CO2 while at the same time reducing NAD to NADH. NADH can be used by the electron transport chain to create further ATP as part of oxidative phosphorylation. To fully oxidize the equivalent of one glucose molecule, two acetyl-CoA must be metabolized by the Krebs cycle. Two waste products, H2O and CO2, are created during this cycle. 1. Define RQ. What is its value for fats? Ans: Respiratory quotient (R.Q) id the ratio of the volume of carbon dioxide released to the volume of oxygen taken in respiration in the given period of time at standard temperature and pressure. R. Q. = Volume of CO2 evolved Volume of O2 absorbed Its value for fats is less than unity 2. Give briefly the structure of ATP? ATP contains the purine base adenine and the sugar ribose which together form the nucleoside adenosine. The basic building blocks used to construct ATP are carbon, hydrogen, nitrogen, oxygen, and phosphorus which are assembled in a complex that contains the number of subatomic parts equivalent to over 500 hydrogen atoms. One phosphate ester bond and two phosphate anhydride Consortium for Educational Communication bonds hold the three phosphates (PO4) and the ribose together. The construction also contains a b-N glycoside bond holding the ribose and the adenine together. 14. What are the main constituents of ATP? Ans: ATP consists of the purine base adenine and the sugar ribose which together form the nucleoside adenosine and three phosphates. 15. What do you mean by compensation point? The compensation point is the amount of light intensity on the light curve where the rate of photosynthesis exactly matches the rate of respiration. At this point, the uptake of CO2 through photosynthetic pathways is exactly matched to the respiratory release of carbon dioxide, and the uptake of O2 by respiration is exactly matched to the photosynthetic release of oxygen. 16. What are redox reaction? Any oxidation-reduction (redox) reaction can be divided into two half reactions: one in which a chemical species undergoes oxidation and one in which another chemical species undergoes reduction. Although oxidation and reduction must occur together, it is convenient when describing electron transfers to consider the two halves of an oxidation-reduction reaction separately. 17. Define redox potential? Reduction potential (also known as redox potential, oxidation / reduction potential, ORP or ) is a measure of the tendency of a chemical species to acquire electrons and thereby be reduced. Reduction potential is measured in volts (V), or millivolts (mV). Each species has its own intrinsic reduction potential; the more positive the potential, the greater the species’ affinity for electrons and tendency to be reduced.