* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Introduction 9 INTRODUCTION Circadian rhythms in physiology and
Premovement neuronal activity wikipedia , lookup
Microneurography wikipedia , lookup
Lunar effect wikipedia , lookup
Time perception wikipedia , lookup
Central pattern generator wikipedia , lookup
Optogenetics wikipedia , lookup
Delayed sleep phase disorder wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Neural oscillation wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Metastability in the brain wikipedia , lookup
Difference due to memory wikipedia , lookup
Colin Pittendrigh wikipedia , lookup
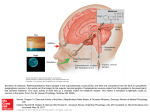
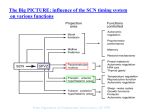
Introduction 9 INTRODUCTION Circadian rhythms in physiology and behavior can be found throughout the plant and animal kingdom and are generated in individual organisms by an endogenous circadian pacemaker. In mammals, the main circadian pacemaker is located in the suprachiasmatic nuclei (SCN) at the base of the hypothalamus. First indications were provided in 1972, when it was shown that lesions of the SCN abolish the adrenal corticosterone rhythm, as well as rhythms in drinking and locomotor activity. In 1990, it was demonstrated that transplantation of the SCN into an arrhythmic SCN-lesioned animal restores rhythmicity with a period that is characteristic of the donor, providing unambiguous evidence for the location of the mammalian circadian pacemaker. Since then, it has become clear that circadian rhythm generation occurs within individual SCN cells. The cell-autonomous mechanism of rhythm generation results in the presence of multiple, single cell oscillators within the SCN. Circadian rhythms need to be correctly phased to the external environment. This is achieved by synchronization to the environmental light-dark cycle, which occurs as a consequence of the phase shifting effects of light. Photic information is transmitted from the retina in the eye to the SCN via a specialized pathway, which terminates predominantly in the ventral SCN region. This suggests that the SCN is a heterogeneous structure and that the multiple, single cell oscillators within the ventral SCN may be functionally different from those in the dorsal SCN. In the past decade, increasing evidence has appeared for the presence of autonomous oscillators outside the SCN in the central nervous system and also in peripheral tissues. These oscillators may or may not act as local pacemakers. For a proper functioning of the circadian system, the multiple oscillators within and outside the SCN need to be mutually synchronized. Interactions are thought to exist within the SCN, between the two SCN nuclei and between the SCN and the periphery. It is now well-established that the generation of circadian rhythmicity in single cells occurs at the molecular level and is based on a transcriptional-translational feedback loop, consisting of several clock genes and their protein products. On the other hand, the presence of a multitude of circadian oscillators, as well as the occurrence of coupling between them, suggests that other characteristics of the circadian timing system may be generated at the neuronal network or tissue level. The aim of this thesis was to obtain insight in the hierarchical organization of the circadian timing system and to determine whether certain attributes of the system arise at the tissue level. The first chapter of this thesis is a review discussing the presence and characteristics of multiple oscillators in several vertebrate and invertebrate animal species. Moreover, the existence and mechanisms of coupling within the circadian timing systems of these species are compared. In the experiments described in chapters 2 through 6, the responses of different components of the system to several types of phase 10 Introduction resetting stimuli are investigated and the interactions between a number of them are characterized. The individual chapters are outlined below. Phase resetting of the circadian timing system is noticeable as a phase advance or delay in the onset of behavioral activity. However, it has been observed that light-induced phase resetting of the activity offset shows different kinetics than that of the onset. This phenomenon is most obvious following light pulses that are administered during the late subjective night. The activity offset responds with a substantial phase advance, the full magnitude of which is reached almost immediately. In contrast, the activity onset shows transient phase resetting with the size of the phase shift increasing in the course of several cycles. As a result, the length of daily activity changes temporarily, but generally returns to baseline values in the course of several days. A model has been proposed suggesting that the mammalian circadian pacemaker consists of two coupled circadian oscillators that control either the activity onset or offset and that respond differently to light. It is not clear whether the systems responsible for the different phase shifting kinetics of the activity onset and offset are located within the SCN or whether the difference is a result of complex downstream responses. The experiments described in chapter 2 were designed to further characterize the systems controlling the behavioral activity onset and offset. In addition to light, several other stimuli, collectively referred to as non-photic stimuli, are able to affect the phase of overt circadian rhythms. In order to investigate the responses of the activity onset and offset to non-photic stimuli, their phase shifts were examined following administration of the opioid agonist fentanyl and the benzodiazepine midazolam. The SCN receives several neuronal projections from the brain, such as a serotonergic projection from the raphe nuclei and a projection from the intergeniculate leaflet that contains, among others, neuropeptide Y and enkephalins, which are endogenous opioids. Many neurotransmitters present in the afferents of the SCN have phase shifting effects on behavioral rhythms and their actions on molecular and electrophysiological processes in the SCN have been relatively well described. A previous study from our lab demonstrated that the opioid agonist fentanyl induces phase shifts in hamster wheel running activity rhythms. These data suggest that the hamster circadian timing system can be modulated by opioidergic input, but do not reveal the site of action, e.g. whether opioids act directly on the SCN in inducing phase shifts. In the experiments described in chapter 3, we investigated whether fentanyl affects the neuronal discharge rate and clock gene expression in the SCN. Moreover, the existence of a putative interaction in the SCN between light input and opioids was investigated at the behavioral and the molecular level. It has become clear that certain types of behavior affect the mammalian circadian system. For example, several hours of wheel running activity during the subjective day may induce phase advances in locomotor activity rhythms. Interestingly, behavioral activity can be accompanied by a suppression of SCN neuronal activity, suggesting that behavioral activity also has acute effects on Introduction 11 the SCN. It has been unresolved whether sleep is one of the behavioral processes that affect the biological clock. Some indications for such an effect have come from the phase shifting effect of sleep deprivation on locomotor activity rhythms. In the experiments described in chapter 4, the presence of a correlation between vigilance state and short-term changes in the discharge rate of SCN neurons was explored through simultaneous recording of the electroencephalogram (EEG) and SCN neuronal activity. When this correlation was established, the presence of a causal relationship between these phenomena was tested using selective sleep deprivation experiments. After having analyzed the effects of several non-photic stimuli on the circadian pacemaker, chapters 5 and 6 focus on the photic input. The effects of short light pulses on the SCN are relatively well described. For example, light pulses induce sustained changes in SCN neuronal activity and, after administration during the subjective night, they result in a change in the expression level of several clock genes in the SCN. In a study by Yamazaki and coworkers (2000), animals were exposed to another photic stimulus, a phase shift of the light-dark cycle, and the effects on the rhythms in clock gene expression in the SCN were investigated. It was demonstrated that the Per1 bioluminescence rhythm phase shifted immediately upon a 6-hour phase advance or a 6-hour phase delay of the light-dark cycle. This was surprising, since the behavior of most laboratory rodents adjusts relatively slowly to a shifted cycle. The findings suggest that the molecular clock in the SCN and overt locomotor rhythmicity can dissociate temporally. The experiments described in chapters 5 and 6 were designed to investigate at which level this dissociation occurs. In chapter 5, the phase resetting characteristics of locomotor rhythms were compared to those of clock gene expression and electrical activity rhythms in the SCN. In chapter 6, the phase resetting kinetics of the electrical activity rhythms in the dorsal and ventral parts of the SCN were studied. These experiments were performed using intact SCN slices and slices that were bisected to physically separate the ventral and dorsal SCN regions. The effect of longterm application of the GABAA antagonist bicuculline on the electrical activity patterns in these regions was investigated, as well as the immediate effects of short-term bicuculline application.