* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Neuro Objectives 17
Ligand binding assay wikipedia , lookup
Vesicular monoamine transporter wikipedia , lookup
Peptide synthesis wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Proteolysis wikipedia , lookup
Biochemical cascade wikipedia , lookup
Lipid signaling wikipedia , lookup
Paracrine signalling wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Signal transduction wikipedia , lookup
NMDA receptor wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Chemical synapse wikipedia , lookup
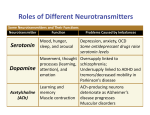
Neuro Objectives 17 1. Chemical vs. electrical synapses: a. Time course: Chemical: 1-5 msec or more (30-50 nm gap) Electrical: instantaneous (3.5 nm gap) b. Localization: Chemical: Can diverge/converge Electrical: Confined to space of transmission c. Directionality of transmission: Chemical: Unidirectional Electrical: Bidirectional 2. 4 steps of chemical synaptic transmission: 1. Synthesis of neurotransmitter within presynaptic neuron 2. Storage and release of the neurotransmitter from synaptic vesicles 3. Interaction of the neurotransmitter with specific post-synaptic receptors 4. Termination of neurotransmitter synaptic action 3. Synthesis of major small molecule neurotransmitters: a. Acetylcholine: choline acetyltransferase acetyl CoA + choline acetylcholine (metabolism) (diet) b. Dopamine: tyrosine hydroxylase tyrosine DOPA (essential aa) DOPA decarboxylase DOPA dopamine c. Norepinephrine: dopamine β-hydroxylase dopamine norepinephrine d. Serotonin: tryptophan hydroxylase tryptophan (essential aa) 5-OH-tryptophan aromatic amino acid decarboxylase 5-OH-tryptophan serotonin 4. Chemical synaptic transmission events: a. Action potential reaches pre-synaptic bouton b. Voltage-gated Ca2+ channels open to increase intracellular Ca2+ c. Ca2+ causes vesicles to fuse with bouton membrane, releasing neurotransmitters in quantal release (all units in one vesicle at a time) d. Neurotransmitter interacts with post-synaptic receptors e. Post-synaptic receptors cause change in post-synaptic cell 5. Mechanistic bases for EPSPs and IPSPs: EPSP is a result of a depolarizing (excitatory) potential, usually axodendritic and caused by an influx of Na+ through a ligand-gated Na+ channel. An IPSP is a result of a hyperpolarizing (inhibitory) potential, usually axosomatic and caused by an influx of Cl- through a ligand-gated Cl- channel or outflux of K+ through a ligand-gated K+ channel. Integration at the axon hillock: Integration of EPSPs and IPSPs occurs throughout the cell, but at the axon hillock (where the threshold to fire an action potential is the lowest) the integration is especially important due to its capability to fire an action potential. 6. Termination of: a. small molecule neurotransmitters: a. reuptake: glutamate, aspartate, GABA, glycine, norepinephrine, dopamine, and serotonin are all pumped back into the presynaptic cell against a concentration gradient b. metabolism: acetylcholine (acetylcholinesterase), norepinephrine (MAO, COMT to form MHPG), dopamine (MAO, COMT to form HVA), and serotonin (MAO to form 5-HIAA) are metabolized by the body and levels of their metabolites can quantitate activity of the neurotransmitter b. peptide neurotransmitters: all metabolized either by endopeptidases (cleaving the middle of the protein) or exopeptidases (cleaving the ends of the protein) 7. Metabolism of biogenic amine neurotransmitters: a. Dopamine: MAO, COMT to form HVA b. Norepinephrine: MAO, COMT to form MHPG c. Serotonin: MAO to form 5-HIAA 8. Small molecule vs. peptide neurotransmitters: a. synthesis: Small molecule: enzymes in presynaptic nerve terminal Peptide: cell body from specific mRNAs b. processing: Small molecule: enzymatic buildup of precursors Peptide: enzymatic cleavage of large polypeptides c. d. storage: Small molecule: small vesicles Peptide: large vesicles termination: Small molecule: reuptake, diffusion, enzymatic catabolism Peptide: enzymatic hydrolysis 9. Time courses of action for: a. neurotransmitter-gated ion channels: milliseconds (short-lived) b. receptors linked to second messengers: seconds to hours (long-lived) 10. Neurotransmitter-gated ion channels: a. Nicotinic cholinergic receptor (activated by acetylcholine): Nicotine acts as an agonist, primary excitatory neurotransmitter of the PNS, increase in 1.5 Na+ to 1 K+ permeability for depolarization (muscle contraction) b. NMDA receptor (activated by glutamate): NMDA acts as an agonist, primary excitatory neurotransmitter of the CNS, increase in Na+ and Ca2+ influx for depolarization (learning and memory) c. GABA receptor (activated by GABA): primary inhibitory neurotransmitter of the CNS, increase in Cl- influx for hyperpolarization 11. Role of G-proteins in receptor activation: Intracellular G-protein binds to GTP causing dissociation into 3 subunits. These subunits activate PLC or adenylate cyclase. GTP is hydrolyzed back to GDP by intrinsic GTPases and the G-proteins bind back to their receptors. cAMP formation: when G-proteins bind adenylate cyclase, it forms cAMP from ATP which activates enzymes including PKA. cAMP is terminated by phosphodiesterase. o Positive coupling: noradrenergic beta-adrenergic receptor, dopamine D1 receptor, histamine H2 receptor o Negative coupling: noradrenergic alpha2-adrenergic receptor, dopamine D2 receptor, muscarinic M2 receptor PI turnover: PI is a phospholipid in the bilayer that can be doubly phosphorylated to make PIP2. When G-proteins activate PLC, PLC cleaves PIP2 to make IP3 and DAG. o IP3 increases Ca2+ concentration which feeds back to degrade IP3 via Ca2+ dependent enzymes o DAG activates PKC to phosphorylate downstream proteins. Coupling: noradrenergic alpha1-adrenergic receptor, muscarinic M1 receptor, serotonin 5HT2 receptor