Chem 101A Exam 4 Concepts Chapter 7 – Modern Atomic Theory
... Notable scientists and their contributions: Rutherford, Bohr, Planc, de Broglie, Heisenberg, Schrödinger. The four Quantum Numbers (n,l,ml,ms), when they are allowed, how they describe the electron state, and how they relate to: Energy levels, orbitals (number allowed, shapes, sizes), elec ...
... Notable scientists and their contributions: Rutherford, Bohr, Planc, de Broglie, Heisenberg, Schrödinger. The four Quantum Numbers (n,l,ml,ms), when they are allowed, how they describe the electron state, and how they relate to: Energy levels, orbitals (number allowed, shapes, sizes), elec ...
periodic table
... 3) element at the top Elements in the same group have: A. similar characteristic properties: bp/fp/sp heat B. same # electrons in the valence shell C. same oxidation #: charge after octet rule applied 4. Zig-zag line between B-Al and Po-At separates metals (80% chart) on the left from non-metals on ...
... 3) element at the top Elements in the same group have: A. similar characteristic properties: bp/fp/sp heat B. same # electrons in the valence shell C. same oxidation #: charge after octet rule applied 4. Zig-zag line between B-Al and Po-At separates metals (80% chart) on the left from non-metals on ...
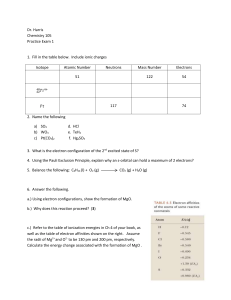
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... Why do atoms exhibit discontinuous (line) spectra when they emit light? Why can’t an atom emit any wavelength of light? ...
... Why do atoms exhibit discontinuous (line) spectra when they emit light? Why can’t an atom emit any wavelength of light? ...
File
... 32. Give an example of a compound. H2O 33. What is a molecule? An element with more than one atom attached to it 34. Give an example of a molecule. O₂- air we breathe O₃- ozone layer 35. As you go from left to right on the periodic table, describe the changes that occur to element's atomic structure ...
... 32. Give an example of a compound. H2O 33. What is a molecule? An element with more than one atom attached to it 34. Give an example of a molecule. O₂- air we breathe O₃- ozone layer 35. As you go from left to right on the periodic table, describe the changes that occur to element's atomic structure ...
Chem 400 Chem 150 REVIEW SHEET Amanda R
... Trends in Periodic Table – trends of elements to predict formation of bonds o Counting valence electrons, electron configuration o Atomic radii increases to the left and down o Electron Affinity/Ionization Energy and electronegativity increases going up and to the right Types of Bonds – must know wh ...
... Trends in Periodic Table – trends of elements to predict formation of bonds o Counting valence electrons, electron configuration o Atomic radii increases to the left and down o Electron Affinity/Ionization Energy and electronegativity increases going up and to the right Types of Bonds – must know wh ...
Trends in the periodic table - Brigham Young University
... M + H2O MOH (M = Li, Na, K, Rb, Cs) ...
... M + H2O MOH (M = Li, Na, K, Rb, Cs) ...
NOTES: 2.1 - Intro to Chemistry
... Isotopes: atoms of an element that have different # of neutrons ● in nature, elements occur as mixtures of isotopes ● some are radioactive: unstable isotope where nucleus decays emitting subatomic particles and/or energy as radioactivity causing one element to transform into another element ...
... Isotopes: atoms of an element that have different # of neutrons ● in nature, elements occur as mixtures of isotopes ● some are radioactive: unstable isotope where nucleus decays emitting subatomic particles and/or energy as radioactivity causing one element to transform into another element ...
17 review for test - Blair Community Schools
... What is the atomic mass? What determines that identity of an atom? What happens to metallic properties as one goes across the table? ...
... What is the atomic mass? What determines that identity of an atom? What happens to metallic properties as one goes across the table? ...
Learning Objectives
... of atomic structure. 4. Distinguish between each of the following pairs of terms: a. neutron and proton b. atomic number and mass number c. atomic weight and mass number 5. Explain how the atomic number and mass number of an atom can be used to determine the number of neutrons. 6. Explain how two is ...
... of atomic structure. 4. Distinguish between each of the following pairs of terms: a. neutron and proton b. atomic number and mass number c. atomic weight and mass number 5. Explain how the atomic number and mass number of an atom can be used to determine the number of neutrons. 6. Explain how two is ...
Atoms and Elements Notes
... • Electrons in the outer most shell • Can be determined by the column # (group) that the element exists in. • All atoms trying to reach the magic 8 number of valence electrons. (Octet Rule) • Number of valence electrons determines how reactive an element is and what type of bonds they form. ...
... • Electrons in the outer most shell • Can be determined by the column # (group) that the element exists in. • All atoms trying to reach the magic 8 number of valence electrons. (Octet Rule) • Number of valence electrons determines how reactive an element is and what type of bonds they form. ...
Chapter 6 Vocabulary crossword puzzle
... 3. Elements in which the highest occupied s and p sublevels are partially filled 6. Measures the ability of an atom to attract electrons when the atom is in a compound; the element named Cesium has the lowest amount, while the element named Fluorine has the highest amount 7. Term that refers to a se ...
... 3. Elements in which the highest occupied s and p sublevels are partially filled 6. Measures the ability of an atom to attract electrons when the atom is in a compound; the element named Cesium has the lowest amount, while the element named Fluorine has the highest amount 7. Term that refers to a se ...
ATOMS AND THE PERIODIC TABLE chapter three
... (METALOIDS). The elements that are between the metals and nonmetals are known as: SEMICONDUCTORS (METALOIDS) They may exhibit metallic and nonmetallic properties. B Si Ge ...
... (METALOIDS). The elements that are between the metals and nonmetals are known as: SEMICONDUCTORS (METALOIDS) They may exhibit metallic and nonmetallic properties. B Si Ge ...
Chemical Bonding
... • Elements are pure substances that are made of only one type of atom. • Isotopes are elements with different numbers of neutrons. • Because isotopes have the same number electrons, all isotopes of an element have the same chemical properties. ...
... • Elements are pure substances that are made of only one type of atom. • Isotopes are elements with different numbers of neutrons. • Because isotopes have the same number electrons, all isotopes of an element have the same chemical properties. ...
UNIT 5 REVIEW PROBLEMS
... 8. Determine which element would have characteristics of both a metal and a nonmetal: a. b. c. d. ...
... 8. Determine which element would have characteristics of both a metal and a nonmetal: a. b. c. d. ...
Page 233 - ClassZone
... negative ion, how many electrons would bromine (Br) gain to form a negative ion? a. 0 c. 2 b. 1 d. 3 ...
... negative ion, how many electrons would bromine (Br) gain to form a negative ion? a. 0 c. 2 b. 1 d. 3 ...
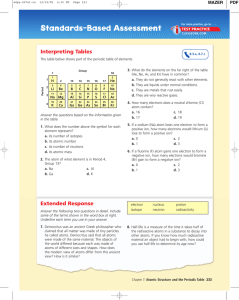
C2- Topic 1: Atomic structure and the periodic table. Assessable
... Explain how Mendeleev: - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
... Explain how Mendeleev: - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
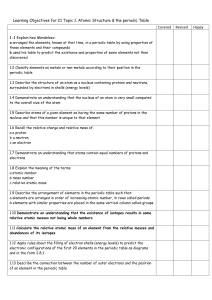
C2 Topic 1 Can Do Sheet
... Learning Objectives for C1 Topic 1. Atomic Structure & the periodic Table Covered 1.1 Explain how Mendeleev: a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some ele ...
... Learning Objectives for C1 Topic 1. Atomic Structure & the periodic Table Covered 1.1 Explain how Mendeleev: a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some ele ...
Chemistry Test Review – 8th Science Vocabulary: Element atom
... The difference between mass number and atomic mass The charge and location of each type of subatomic particle (proton, neutron, electron) Electrons Which ones have the most energy Which ones have the least energy Trends in the Periodic Table Atomic Radius Valence electrons Characteristics of Metals ...
... The difference between mass number and atomic mass The charge and location of each type of subatomic particle (proton, neutron, electron) Electrons Which ones have the most energy Which ones have the least energy Trends in the Periodic Table Atomic Radius Valence electrons Characteristics of Metals ...
Chemistry Overview
... Elements • Element: pure substance that can’t be broken down into simpler substances • Element Symbols – 1-3 letters – Begin with 1 capital letter – Some based on Latin names • Ex/ gold = Au for Aurum iron = Fe for Ferros ...
... Elements • Element: pure substance that can’t be broken down into simpler substances • Element Symbols – 1-3 letters – Begin with 1 capital letter – Some based on Latin names • Ex/ gold = Au for Aurum iron = Fe for Ferros ...
Periodic Table Vocabulary Periodic Table – a chart that organizes
... chemical reaction, matter cannot be created or destroyed but can be changed into a different form. Period law- The chemical properties of elements tends to repeat over specific atomic number intervals Law of definite proportions – elements within a compound have specific mass ratios Isotopes Atoms o ...
... chemical reaction, matter cannot be created or destroyed but can be changed into a different form. Period law- The chemical properties of elements tends to repeat over specific atomic number intervals Law of definite proportions – elements within a compound have specific mass ratios Isotopes Atoms o ...
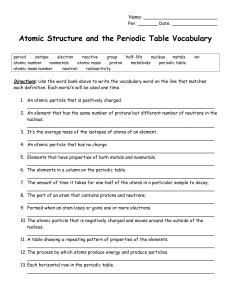
Atomic Structure and the Periodic Table Vocabulary
... nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. An atomic particle that has no charge. __________________________________________________ ...
... nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. An atomic particle that has no charge. __________________________________________________ ...
Summative Assessment Study Guide Name: Due date: SPS1
... SPS3. Students will distinguish the characteristics and components of radioactivity. a. Differentiate among alpha and beta particles and gamma radiation. b. Differentiate between fission and fusion. c. Explain the process half-life as related to radioactive decay. d. Describe nuclear energy, its pra ...
... SPS3. Students will distinguish the characteristics and components of radioactivity. a. Differentiate among alpha and beta particles and gamma radiation. b. Differentiate between fission and fusion. c. Explain the process half-life as related to radioactive decay. d. Describe nuclear energy, its pra ...