* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Phytopathologische Zeitschrift
Cytoplasmic streaming wikipedia , lookup
Tissue engineering wikipedia , lookup
Endomembrane system wikipedia , lookup
Cell encapsulation wikipedia , lookup
Extracellular matrix wikipedia , lookup
Cell growth wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell culture wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
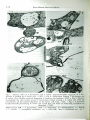
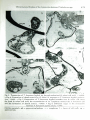
Ph yrop3th . Z ., 107,168- 175 (1983) © 1983 Vcrbg Paul Parcy, Berlin und H amburg l SSN 0031 -948 1 I lntcrCodc: PHYZA3 The Hebrew University of Jerusalem, Department of Plant Pathology and Microbiology, Faculty of Agriculture, Rehovot, Israel Ultrastructural Studies of the Interaction Between Trichoderma spp. and Plant Pathogenic Fungi By Y. ELAD, RINA BARAK, I. CHET and Y. HENIS With 10 figures Received October 18, 1982 Abstract The ultrastructural changes during parasitism of the biocontrol agents Trichoderma harzianum and T. hamatum, were observed under a uansmission electron microscope. Electron micrographs show that during the interaction of TridJoderma spp. with either Sclerotium rolfsii or Rhizoctonia solani the hyphae of the paras ites contact their host, and then enzymatically digest their cell walls. Extracellular fibrillar material is deposited between the interacting cells. P arasite organelles, e.g. mitochondria, ves icles and dark osmiophilic inclusions, accumula te in the parasitizing cells. In response to the invasion, the hoStt produces a sheath matrix which encapasulates the penetrating hyph a and <the h ost cells become empty of cy.taplasm. Z usammenfassung Ultrastrukturelle Untersuchungen der Wechselwirkung zwischen Trichoderma-Arten und pflanzenpathogenen Pilzen Die ultrastrukturellen .Anderungen wahrend der parasirtischen Phase der BiokontrolleAgen z ien Trichoderma harzianum und T. hamatum wurden unter einem Transmissionselektronenmikroskop beobachtet. Die Elektronenmikrogramme zeigen, daB wahrend der Wechselwirkung der Trichoderma-Arten entweder mit Sclerotium rolfsii oder mit Rhizoctonia solani U.S . Copyright Clearance Center Code Statement: 0031-9481 /83/0702-0168$02.50/0 Ultra.S1tructural Studies of the Interaction between Trichoderma spp. 169 die Hyphen der Parasiten mit ihrem Win in Konukt kommen und dann seine Zell wand e enzymatisch vcrzehren. Ein exuazelluHirer fibrillenartiger Stoff wird zwischen den sich gegenseitig beeinflussenden Zellen eingelager.t. Parasi tische Organelle, z. B. Mitochondrien, Vesike l und dunkle osmiumliebende EinschluBkorper, we rden in den parasiticrten Zellen angeha uft. Gegen diese Invasion erzeugt der Win eine umhiillende Gewebeschicht, die die eindrangcnden Hyphen einkapselt und die Wirtszellen sind Zytoplasma leer. Several fungi are mycoparas1t1c on soil-borne plant pathogens. Among these are Gliocladium spp. (Tu 1980), Trichoderma spp. (DENNIS and WEBSTER 1971 ), Pytbium acanthicum (HocH and FuLLER 1977) and S phaerotheca spp. (HocH and PROVVIDENTI 1979). Work dealing with biological control of pathogenic fungi demonstrates that T. harzianum and T. hamatum are effective biocontrol agents (ELAD et al. 1980, 1981 a, 1981 b) HENIS and CHET 1975, WELLS et al. 1972 and HARMAN et al. 1980). Its mode of action in parasitism was recently studied (ELAD et al. 1982 a, 1982 b) . When a parasitic fungus reaches the host hyphae it usually coils around it or produces appressorium-like structures and hook-shaped contact branches. This was shown with Trichoderma hamatum vs. Rhizoctonia solani (CHET et al. 1981); T. harzianum vs. Sclerotium rolfsii (ELAD et al. 1982 a) and Stephanorna phaeospora vs. Fusarium sp. (HocH 1978). Mechanical and enzymatic penetration of the host fungi by the parasite are the next step of interaction (HocH and FuLLER 1977, ELAD et al. 1982 a). Trichoderma harzianum produced P-(1,3)-glucanase and chitinase when grown on cell walls of its host - Sclerotium rolfsii or Rhiz octonia solani (ELAD et al. 1982 b). The enzymatic activity detected in dual cultures of both the mycoparasite and the soil-borne pathogens was suggested as the mechanism by means of which this parasite attacks its host. However, no ultrastructural studies by means of transmission electron microscopy of the sequence of events, occurring during the interaction of Trichoderma spp. with its host, have yet been made. In the present work we used TEM to study the ultrastructural changes during the process of parasitism in this fungal-fungal interaction. Materials and Methods Rhiz octonia solani Kuhn a nd Sclerotium rolfsii Sacc. were used as host fun gi to the mycoparasires Trichoderma harzianum Rifai (ELAD et al. 1980, 1981a, 1981 b, l982a, 1982b) and T. hamatum (Bon.) Bain (CHET et al. 1981 , ELAD et al. 1982a). Fungi were maintained on a synthetic medium (SM) (OKON et al. 1973) at 30 ± 1 ° C . The following system was used in orde r to obse rve •t he interaction sites: A cellophane membrane, well washed in boiling, distilled water, was placed on t he surface of water agar (CHET et al. 1981). Agar discs (SM) covered with mycelium of Tricho derma spp. were placed on one end of the cellophane membrane and discs wi.t h one of the pathogenic fungi were placed on the other. The cellophane plates were then incubaud a t 28 ± 1 °C for 4 days. The mycoparasitc and its host grew towards each other and the hyphae in termin gled on th e cellophane plates. The cultures were fir st observed by a phase-conxrast microscope. 170 ELAD, B.ARAK, CHET and HENIS Preparation for TEM microscopy The mycelium fr om the in.tcraction site was removed and the organisms were fixed in 3 % glut::traldchyde (Sigma Chemicals Co., St. Louis, Mo. 63178, USA) buffered with 0.2 M phosphate buffer, pH 7.0. After refri ge ration for 12 h, the specimens, in the same buffer post-fi~ed in :tqueous 1 % O sO 4 (Sigma), a nd dehydrated via a graded e,t hanol series (and l ate ~ wah a graded propylcneoxide seri es), were embedded in an Epon 812 medium (Polaron Equtpmcnt Ltd., Watford, England, CHET and HENIS 1969). Thin sections, prepared wi.th an LKB ultr ot oma III microtome, using glass knives, were stained with uranyl acetate and lead citra t e and examined with a jeol 100 CX tra nsmiss ion electron microscope, at 80 kV. Results and Discussion Mycelial san1ples, taken from the interaction sites of dual cultures, of either R. so/ani or S. rolfsii and either T. hamatum or T. harzianum, were observed in a transmission electron microscope. In general, no significant differences were observed between the interaction of either of the mycoparasites with either host. Ultrastructurally, the process of penetration and appressorium development m ay b e associated with a variety of morphological alterations at or near the host-parasite interface. A Trichoderma hypha first contacts the host mycelium (Fig. 1). During this time a significant accumulation and deposition of intercellular fibrill ar material can be observed outside the host-parasi te interacting cells (Fig. 2). Our observations support the idea that there is an increase of a mucilaginous substance, apparently polysaccharides, originating from either one of the interacting fungi. This was also found with Stephanorna phaeospora which parasitizes several species of Fusarium (HocH 1978) and is simila r to the description of the nematode-capturing organs of Arthrobotrys ofigospora (NORDBRING-HERTZ and STALHAMMAR-CARLEMALM 1978). Basically, there are two main types of interaction between Trichoderma and either R. solani or S. rolfsii. The mycoparasite may produce and appressorium-like body (Fig. 1) or it may coil around the host hyphae (Fig. 3). That Trichoderma spp. coil around the host hyphae, has been observed in earlier works using light (DENNIS and WEBSTER 1971 )., Nomarski interference (CHET et al. 1981) and scanning electron microscopy (ELAD et al. 1982 a). This, however_, is the first study of T richoderma-pathogenic fungi interactions, using transmission electron microscopy. This method enabled us to detect the ultrastructural changes occurring during the process of parasitism, inside the cells an d not only on their surface. After the meeting of the two hyphal cells, the coiling hyphal branch of Trichoderma constricts and partially digests the S. rolfsii cell wall in the interaction si te (Fig. 3 ). Trichoderma then begins to penetrate the host cell (Fig. 4). Direct penetration of the host cell wall has been previously found with T. hamatum using SEM (ELAD et al. 1982 a). Penetration occurs at different locations of the host cell walls even when these are thick, melanized walls (Figs. 4 to 6 ). Melanin is known for its ability to confer resistance to biological and Ultrastructural S.tudies of the Interaction between Trichoderma spp. 171 chetnical degradation in fungi (CHET and HEN IS 1969, HEN IS and CHET 1975) . The digestion of the host cell walls indicates the activity of extracellular enzyn1es, e.g. P-(1 ,3 )-glucanases and chitinases, recently reported by ELAD et al. (1982 b), with the same antagonistic fungus. The enzymatic decomposition of the host cell wall was shown and discussed with other mycoparasites, such as Piptocephalis virginiana (MANOCHA 1981 ), Gliocladium virens which attacks Sclerotinia sclerotiorum (Tu 1980), Pythium acanthicum (HocH and FuLLER 1977) and Verticillium lecanii which parasitize uredospores of Puccinia graminis var. tritici (HANSSLER et al. 1982). On the other hand, our observations also reveal that a physical pressure on the host hypha is exerted before penetration (Fig. 1). After penetration, the host forms a layer associated with the penetrating parasitic hyphae. This electronlucent sheath encapsulates the penetrating hyphae in a relatively narrow layer (Figs. 6 and 7). It can be seen that the host cell walls are partially dissolved (Figs. 4, 6 and 7). Similarly, JEFFERIES and YouNG (1978), working with the mycoparasite Piptocephalis unispora which attacks fungi from the Mucorales_, showed the formation of a sheath matrix in the host cells, resulting from the invasion of the parasite. This effect was also found by HocH (1978) and RAKVIDHYASASTRA and BUTLER (1973 ). The Trichoderma isolates tested were found to be ''destructive mycoparasites" (HASHIOKA 1973, Tu 1980). Mycoparasitism finally causes the host cells to be empty of cytoplasmic contents. The remaining cytopslasm_, mainly surrounding the invading hyphae, also show disintegration of the remaining host cytoplasm. Decomposition of the host cytoplasm seems to proceed rapidly after the wall is pierced by the mycoparasite (Figs. 2 and 4-1 0). In general, the hyphal cells of the host, R. solani, are large enough to contain a few parasitic cells (Fig. 8). The parasitic cells also invade from cell to cell, via the host's septum (Fig. 10). In some cases, the invading hypha! tips of T. hamatum expand inside the host (Fig. 9). Invaded R. solani cells were found to be empty or with little cytoplasmic residue. This emptiness results from the disintegration of the cell wall and leakage of the cytoplasm. The leakage could, however, occur during specimen preparation. Lack of cell contents may result from translocation into intact cells and autolysis (OKON et al. 1973 ). The host cytoplasm is, apparently, utilized by the parasite, which is capable of degrading proteins and lipids (ELAD et al. 1982 b) . Cytoplasm of the parasite accumulates in the cells associated with the host mycelium (Figs. 1 and 3). An accumulation of vesicles and mitochondria can be observed in the penetrating hyphae (Figs. 4, 7_, 8 and 9). This may indicate a high metabolic activity in the infecting tip. The presence of vesicles in metabolically active tip growing cells has often been connected with wall synthesis and softening activity (BARTNICKI-GARCIA 1973 ), at the tips of which, layers of newly formed cell wall material can be observed (Figs. 6 and 7). 172 ELAD, BARAK , CHET and HENIS .·c . . . .. Fig. 1. Adjacent cells of T. harzianum and S. rolfsii. Appressorium-like structure of T. harzianum is attached to S. rolfsii cell, Y 7875. • Fig. 2. A ssociation between T. harzianum and R. solani. Note fibrillar materi al deposited in between the hyphae, X 5250. • Fig. 3. Electron micrograph of :1 thin section across T. harzianum coiling around an S. rolfsii cell. Note the constriction an d the partial digestion of the host's cell wall, X 6825. • fjg. 4. T. hamatum hypha l branches penetrating R . solani cell. Lysed host cell walls and remaining cytoplasm are observed, X 7500 Abbrevia.rions: HR = T. harzianum; HM = T. hamatum; m = mi-tochondria; s = sheath; v = vesicles; = osmiophilic inclusion; cw = cell wall; mew = melanized cw; fm = UltraSitructural Studies of the Interaction between Trichoderma spp. 173 7 0 • 6 , ... ....-. .. .) . ." ~ Fig. 5. Penetration of T. hamatum hyphal tip through melanized R. solani cell wall, X 11 ,250. • Fig. 6. Formation of shea.th matrix by R . solani in response to T. hamatum hypha l penet ration, X 8625. • Fig. 7. P enetration of T. hamatum hypha! branches into R . so /ani cell. Note the lysed R. solani cell wall, the accumulation of i.ts cytoplasm around the T . hamatum cell and ·the formation of sheMh matrix, X 6450. • Fig. 8. Different stages in t he interact ion between T. hamatum and R. so/ani, X 3975 fibrillar material; ph = penetrating hyph ae; c appressori urn. = cytoplasm; 1 = layers of cell wall ; ap = 174 ELAD, BARAK, CHET and HENIS Fig. 9. Penetration of T. hamatum into R. so/ani hyphae. Note the accumulation of mucilaginous material o utside the host cell and widening of the parasi.te's tip inside the R. solani cell, X 4875. • Fig. 10. Penetration of T. hamatum through R. solani septum. Note the aggregation of host cytoplasm around the parasitic tip, X 3825 In several cases osmiophilic inclusions were observed in Trichoderma mycelium which interacted with its host. These are more abundant in the infecting tips than in normal cells. Similar organelles were found in A. oligospora during nematode trapping (NoRDBRING-HERTZ and STALHAMMARCARLEMALM 1978) . Apparently, these organelles have a function in the parasitic activity of Trichoderma . Mycoparasitism, as evidenced by the interactions described herewith, is the mode of action by which the potential biocontrol agents, Trichoderma spp., attack the soil-borne plant pathogens, R. solani and S. rolfsii. This r esearch was supported by a grant from the United Sta.te s-Israel Binational Agricultural Research and Development Fund (BARD). We thank E. HATAV, E. ABRAHAM, N. BAHAT and Z. SADOVSKI of the E lectron Microscopy and Photography Department in the Hebrew University of Jerusalem for their technical assistance. Literature BARTIN ICKI-GARCIA, S., 1973: Fundamental aspects of hyphal morphogenesis. In: J. M. AsHWORTH a nd ]. E. SMITH (Eds.), Microbia l differentiation, 245- 267. Cambridge University Press, London. CHET, I., G . E. HARMAN, and R. BAKER, 1981: T}·ichoderma hamatum: its hyphal interactions with Rhizoctonia solani and Py thium spp. Microb. Ecol. 7, 29-38. - - , and Y. HEN IS, 1969: Effect of catechol a nd disodium EDTA on melanin content of hyphal and sclerotial walls of Sclerotium rolfsii Sacc. and the role of melanin in the susceptibility of these walls to ,8-(1,3)-glucanase a nd chitinase. Soil Bioi. Biochem. 1, 131-138. DENN IS, L., and J. WEBSTER, 1971: Antagonism properties of species groups of Trichoderma . III. Hyphal interaction. Trans. Brit. mycol. Soc. 57, 363-369. Ultrasuuctural Studies of the Interaction between Trichod erma spp. 175 ELAD, Y., I. CHET, P. BoYLE, and Y. H ENIS, 1982a: The p arasitism of Trichoderma spp. on plant pathogens - Ultrastructural s.tudies an d detection by FITC lec tins. Phy.topathology (in press). - - , - -, and Y. HENIS, 1981 a: Biological control of Rhiz octonia solani in st raw berry fields by Trichoderma harzianum . Plant and Soil 60, 245-254. - - , - - , and ~ - , 1982b): Degreda.tion of plant pathogenic fungi by Trichoderma harzianum. Ca~ad. J. Microbial. 28, 719-725 . - - , --,and J .KATAN, 1980: Trichoderma harzianum: a biocontrol agent of Sclerotium rolfsii and R,hizoctonia solani. Phytopa.thology 70, 119-1 21. - - , Y. HADAR, I. CHET, and Y. HENIS, 1981b: Biological cont rol of Rhiz octonia so /ani by Trichoderma harzianum in carnation. Plant Dis. 65, 675-677. HANSSLER, G., M. HERMANNS, und H . J. REISENER, 1982: Elektronenmikroskopische Beobachtungen der Interaktion zwischen Uredosporen von Pu ccinia graminis var. tritici und Verticillium Jecanii. Phytopa·t h. Z. 103, 139- 148. fuRMAN, G. E ., I. CHET, and R. BAKER, 1980: Trichoderma hamatum effects on seed and seedling disease induced in radi·sh and p ea by Pythium or by Rhizoctonia so/ani. Phytopathology 70, 1167-1172. HASHIOKA, Y., 1973: Mycoparasitism in relation to phytopathogens. Shokubutsu Byogai Kenkyu Kyoto 8, 179-190. HENIS, Y., and I. CHET, 1975: Microbial control of plant pathogens. Adv. appl. Microbial. 19, 85-111. HocH, H . C ., 1978 : M ycoparasitic relationships. IV. Stephanoma phaeospora parasitic in a species of Fusarium. Mycologia 70, 370-379. - - , and M . S. FuLLER, 1977 : Mycoparasitic rela.tionships. I. Morphological feat ures of interactions between Pythium acanthicum and several fungal h osts. Arch. Microbial. 11, 207-224. - - , and R. PROVVIDENTI, 1979: Mycoparasitic relationships : cytology of the S phaerotheca fulig inea -Tilletiop sis sp. interaction. Phytopa.t hology 69, 359-36L. JEFFERIES, P., and T. W. K. YouNG, 1978 : Mycoparasitism by Piptochephalis unispora (Mu corales) : host range and reaction with Ph ascolomy ces articulosus . Canad. J. Bot. 56, 2449-2459. MANOCHA, M. S., 1981: Host specificity and mechanism of resistance m a mycoparasltlc system. Physiol. Plant Pathol. 18, 257-265. NoRBRING-HERTZ, B., and M. STALHAMMER-CARLEMALM, 1978: Capture of nematodes by Arthrabotrys oligospora, an electron microscope s.t udy. Canad. J. Bot. 56, 1297-1307. 0KON, Y., I. CHET, and Y. H ENIS, 1973: Effect of lactose, ethanol and cycloheximide on translocation pattern of radioactive compounds and on sclerotia! fo rmation in Sclerotium rolfsii . ]. gen. Microbial. 74, 251-258. RAXVIDHYASASTRA, V., and E. E. BuTLER, 1973 : Mycoparasitism by Stephanoma phaeospora. Mycologia 65, 580-593. Tu, J . C., 1980: Gliocladium virens, a destructive mycoparasite of Sclerotinia sclerotio rum. Phytopathology 70, 670-674. WELLS, H. D., D. K . BELL, and C . A. ]AWARSKI, 1972: Efficacy of T richoderma harzianum as a biological control for Sclerotium rolfsii. Phytopathology 62, 442-447. Authors' address: Depafltment of Plant Pathology and Micr obiology, The H ebrew University of Jerusalem, Faculty of Agriculture, R ehovot 76100 (Israel). This document is a scanned copy of a printed document. No warranty is given about the accuracy of the copy. Users should refer to the original published version of the material.








![Transcriptome analysis of [i]Solanum lycopersicum[i] roots](http://s1.studyres.com/store/data/015306049_1-3e3651d660a3d3f0659f11cb0728af36-150x150.png)









