* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cell Respiration
Biosynthesis wikipedia , lookup
Butyric acid wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Mitochondrion wikipedia , lookup
Metalloprotein wikipedia , lookup
Phosphorylation wikipedia , lookup
Photosynthesis wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Electron transport chain wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Microbial metabolism wikipedia , lookup
Citric acid cycle wikipedia , lookup
Biochemistry wikipedia , lookup
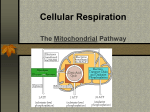
Cell Respiration Mrs: Jackie Respiration defined Respiration- series of chemical reactions that occur inside a living cell. Involves breakdown of large substances into smaller substances Substance most commonly used is glucose Stages of respiration 1. Glycolysis Occurs in the cytosol (semifluid part of the cytoplasm) Involves the phosphorylation of glucose into glucose-6-phosphate Follow by many reactions resulting in the splitting of glucose in half to form 2 pyruvic acid molecules ( each with 3 Carbons) Other important products 2NADH + H+ and 2 ATP Glycolisis continues 1. Glucose 6-phosphate is formed when the 6th carbon on the glucose molecule is phosphorylated by an ATP molecule. 2. Glucose 6-phosphate is converted into a 5-carbon ring isomer, fructose 6phosphate. 3. Fructose 6-phosphate is phosphorylated by another ATP to form fructose 1, 6diphosphate. 4. Fructose 1, 6-diphosphate is processed by an enzyme into two glyceraldehyde 3phosphate molecules. 5. Two molecules of glyceraldehyde 3phosphate are oxidized, losing hydrogen atoms and gaining phosphate groups to form 1, 3-diphosphoglycerate. Two molecules of NAD+ are converted into NADH2+ in the process. Glycolysis continues 6. Two 1,3-diphosphoglycerate molecules phosphorylate ADP (adenine diphosphate) to yield two molecules of 3-phosphoglycerate and two ATPs are produced. 7. The phosphate groups on 3phosphoglycerate move to the 2nd carbon, forming 2-phosphoglycerate. 8. The two 2-phosphoglycerate molecules are dehydrated and forms two high-energy phosphoenolpyruvate molecules. 9. The two phospoenolpyruvate phosphorylates two ADPs and produces two more ATPs and two molecules of pyruvate. Glycolysis summary In the next sequence of reactions 4 molecules of ATP are produced and the total energy harvest of ATP can be calculated as shown on the table Number of ATP molecules used 2 Number of ATP molecules produced 4 Net number of ATP 2 molecules gained in glycolysis Glycolysis NAD+ is a coenzyme Hydrogen carrier NADH + H formed by removing hydrogen from glucose (oxidation of glucose), and giving this H to NAD+ (reduction) NAD+ +2H NADH+ H+ Glucose is reduced (removal of H) and NADH is oxidized Glycolysis NAD+ NADH + H+ Glucose Pyruvic oxidation pyruvate acid made in glycolysis can now enter into aerobic or anaerobic pathways of respiration Anaerobic respiration If respiration is anaerobic Pyruvate is reduced into either lactic acid releasing NAD+ or Alcohol is produced with the release of NAD+ and carbon dioxide The reduction of pyruvate into lactic acid or ethanol does not release energy. Only serves in the release of NAD+ Anaerobic respiration These NADs can be used again in glycolysis This step produced ethanol or lactic acid which is less toxic as a waste product in the surroundings of these organisms Anaerobic respiration Occurs in mostly small animals which do not need much energy for the way of life The two ATP formed are enough for providing energy to these simple organisms such as bacteria and yeast Also occurs during strenuous exercise in mammals Anaerobic respiration Glycolysis Phosphorilation Oxidation Lysis 2 pyruvate + 2 ATP +2NADH + H+ Fermentation Fermentation 2 pyruvate either 2NADH + H 2NADH + H 2NAD 2NAD + CO2 LACTIC ACID Ethanol Oxidation and reduction Oxidation involves the loss of electrons from an element Reduction involves a gain of electron Phosphorylation is a process in which ATP is made. Industrial uses of anaerobic respiration Alcoholic fermentation Manufacture of alcoholic drinks • Wine – make by incubating crushed grapes in a sealed container at a temperature of 40 degrees celsius. • Need the correct bacterias and yeast that carry out anaerobic respiration and breakdown of sugar in grapes into ethanol and CO2 Industrial uses of anaerobic respiration Manufacture of Yogurt Milk is incubated over night with bacteria at around 40 degrees Lactic acid makes the milk to clot into yogurt Aerobic respiration After glycolysis the pyruvic acid enters into the mitochondria In the matrix of the mitochondria, pyruvate is decarboxylated (CO2 mol is removed) Decarboxylase remove CO2 Dehydrogenase oxidised (hydrogen removed) Aerobic respiration Resulting molecule is called acetyl group (2 carbons) Results from the removal of Carbon dioxide and combination with coenzyme A= Acetyl Co A This reactions are called link reactions since they link pyruvate with the Krebs cycle This compound Acetyl Co A then enters into the matrix of the mitochondria. The Krebs Cycle Starting molecule is 2 carbon compound called oxaloacetic acid It reacts with acetyl CoA and forms a 6 carbon compound called Citric acid The Krebs Cycle Coenzyme A comes out unaffected Citric acid loses CO2 and becomes a 5 carbon compound A different stages in the cycle compounds loses Hydrogen which comes out in the form of NADH+H and FADH2 The Krebs Cycle Final output 2ATP, 4CO2, 6NADH +H and 2FADH2 Chemiosmotic oxidative phosphorylation and the electron transport chain Chemiosmotic oxidative phosphorylation and the electron transport chain NADH + H and FADH2 coming out of all the reaction of respiration are processed in the mitochondria to produce ATP This is carry out by electron carriers that are situated in the inner membrane of the mitochondria and cristae Electron transport chain NADH +H gives its electron to the first electron carrier in the ETC, thus NADH +H becomes NAD+, in other terms NADH+H is oxidized and the first carrier in the chain is reduce The first carrier passes the electron to the next carrier and so on electrons are passed from one carrier to the next in the electron transport chain As the electrons are passed from one carrier to the next they go down to a lower and lower energy level. Electron transport chain These electrons are finally taken by oxygen to form water. Oxygen is the final recipient of the electrons passing down the electron transport chain. In this process oxygen becomes reduced into water. Going down the energy gradient, the electrons released energy Electron transport chain The energy released from the electrons is used to pump protons (H+) from the matrix to the intermembranal space. Due to the small volume of this space, it become highly concentrated with protons very quickly Electron transport chain This created two compartments in the mitochondria with different proton concentrations. The matrix with a low concentration and the intermembranal space with a high concentration. This results in the protons moving down their concentration gradient from the intermembranal space to the matrix. However the only path they can pass is through channels provided by the enzyme ATP synthetase. Electron transport chain Protons diffuse quickly through the ATPase channels, thus activating this enzyme. When ATPase becomes active, it catalyses the phosphorilation of ADP into ATP, and so ATP is formed in this way from NADH+H All the above steps are called oxidative phosphorilation. Products NAD+ and FAD take hydrogen stripped off the glucose molecule into the ETC where they are processed into ATP. Carbons of the glucose are released in the form of CO2.