* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cellular Respiration
Lactate dehydrogenase wikipedia , lookup
Photosynthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Mitochondrion wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Phosphorylation wikipedia , lookup
Electron transport chain wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Microbial metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Biochemistry wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
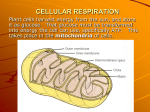
Chapter 9 p. 160-170 Principles of Energy Harvest Cells require energy to perform many types of work By breaking down complex organic molecules into simpler products Some energy used for work; the rest is released as heat Catabolic Pathway: releases stored energy Cell Respiration & Fermentation are Catabolic Fermentation: does NOT need O2 to break down sugars Cell Respiration: uses O2 to break down organic molecules (glucose) Yields most amt ATP Occurs mostly in mitochondria ∆G = -686 kcal/mole Cells Recycle ATP ATP releases energy when phosphate group is removed P group transferred to another molecule (“phosphorylated”) Causes molecule to perform work Redox Reactions Release Energy Rearrangement of e-’s during chemical rxns releases energy stored in food Redox rxn: “reduction-oxidation”; involves transfer of e-’s from one reactant to another Oxidation: loss of e- by reducing agent Reduction: gain of e- by oxidizing agent May also involve change in e- sharing As e- moves toward more electronegative atom, energy is released Electrons “fall” from Organic Molecules to O2 Cell respir. is a redox rxn; glucose is oxidized & O2 is reduced C6H12O6 + 6O2 → 6CO2 + 6H2O +energy H+ transferred from glucose → O2 H+ will lose its e-’s easily to O2 Carbs & fats (high in H+) act as reservoirs of e-’s for cell respiration “Fall” of Electrons Occurs in Steps Glucose (org mol.) is broken down in a series of steps, each w/ a catalyst H+ atoms from glucose are transferred to NAD+ (coenzyme) before O2 Dehydrogenase: enzyme that removes pair of H atoms from glucose (org. mol.) Delivers 2 e- & 1 proton to NAD+ e-’s lose very little energy in this process To get e-’s from NADH to O2 requires the use of the Electron Transport Chain (ETC) Passes e-’s along in series of steps Involves proteins embedded in inner membrane of mitochondria e-’s in NADH at “top”; O2 at “bottom” Each molecule along chain more electroneg. than one before During cell respir., e-’s move from food→NADH→e- transport chain→O2 Overview of Cellular Respiration Occurs in 3 stages: 1) Glycolysis (glucose → 2 pyruvate) 2) Krebs Cycle (pyruvate derivitive →CO2) 3) Electron Transport Chain Glycolysis & Krebs break down glucose (org. mol.) ETC accepts e-’s from first 2 stages (via NADH) ATP Synthesis during Cellular Respiration Oxidative Phosphorylation: energy released in ETC used for ATP synthesis in mitochondrion Substrate-Level Phosphorylation: produces small amounts ATP during glycolysis & Krebs An enzyme transfers P group from organic molecule to ADP Each molecule glucose yields ~38 ATP Glycolysis Harvests Chemical Energy Glycolysis = “splitting of sugar” 10-step process Steps 1-5 = spends 2 ATP Steps 6-10 = produces 4 ATP + 2 NADH Net Yield = 2 ATP, 2 NADH NADH creates more ATP in ETC Energy in pyruvate extracted in Krebs Pyruvate → Acetyl CoA in Mitochondria In presence of O2, pyruvate enters mitochondria In mitoch.: pyruvate → acetyl CoA → Krebs Krebs Cycle 8-step cycle that takes in acetyl CoA and expels CO2 Acetate + oxaloacetate → citrate Citrate → oxaloacetate Energy gets stored in 3 NADH & 1 FADH2, which give e-s to ETC 2 ATP produced (2 acetates enter cycle) Chapter 9 p. 170-178 ETC Coupled to ATP Synthesis So far, most energy is stored in NADH & FADH2, linking Krebs to ETC ETC does not make ATP directly; it allows e-s to fall gradually to O2 Mostly composed of proteins with non-protein (prosthetic) groups attached Groups undergo redox rxns as they move e-s Cytochrome: protein w/ a heme group FADH2 adds e-s further down; creates 1/3 less energy than NADH ATP Synthesized by Chemiosmosis ATP Synthase: protein in inner membrane of mitochondria; converts ADP + P → ATP Uses energy from H+ conc. gradient across mitoch. membrane (“Proton-Motive Force”) Created by redox rxns in ETC (pump H+ out) As H+ diffuses back in through ATP synthase, force “churns” synthase, connecting ADP + P → ATP Chemiosmosis: coupling of chemical rxn to osmosis of H+ back into mitochondria Also occurs in chloroplasts for photosynthesis and in cell membrane of prokaryotes Cell Respiration: a review Energy flow: glucose → NADH → ETC → proton-motive force → ATP ATP synthesis: Glycolysis: 2 ATP Krebs: 2 ATP ETC: 34ATP Total: ~38 ATP # ATP estimated because: 1) depends on whether NADH/FADH2 used 2) some energy used for other work Usable energy yield ~40%; rest lost as heat, sweat, etc. Related Metabolic Processes ATP yield from cell respiration depends on presence of O2 (aerobic) When O2 absent, ATP generated by Fermentation (anaerobic) Glycolysis produces 2 ATP regardless NAD+ is oxidizing agent Fermentation Produces ATP w/out O2 Fermentation: generates 2 ATP by substrate-level phosphorylation w/ NAD+ (glycolysis) e-’s from NADH → pyruvate (or derivitive) and NAD+ gets recycled Many types; differ in waste products made Alcoholic Fermentation Pyruvate → acetylaldehyde (releases CO2) Acetylaldehyde → ethanol (NADH is oxidized) Performed by yeast (brewing) and bacteria Lactic Acid Fermentation Pyruvate → lactate (ionized lactic acid) NO CO2 released NADH is oxidized Performed by fungi & bacteria to make cheese & yogurt Also occurs in muscles early in exercise, when O2 is low As lactate accumulates, muscle fatigue & burning result Fermentation/Respiration Compared Both use glycolysis to break down glucose (2 ATP) Differ in how NADH is oxidized to NAD+ Pyruvate leads to next step – depends on presence of O2 Cell respiration includes Krebs and ETC, producing ~19x’s more ATP Faculative Anaerobes: can survive using either process Evolutionary Significance of Glycolysis Ancient prokaryotes probably generated ATP through glycolysis due to low levels atmospheric O2 Determined to have evolved early because: Very widespread pathway Occurs in cytosol (not in membrane-bound organelle) Versatility of Catabolism Glycolysis can accept many types of organic molecules Carbs: starch, glycogen, disacch. break down to glucose → glycolysis Proteins: excess amino acids remove amine group → glycolysis Fats: glycerol → glyceraldehyde phosphate → glycolysis Beta Oxidation: breaks down fatty acids into pieces that enter Krebs Produces >2x’s ATP as carbs, but must work harder