* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 6
Biochemical cascade wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Photosynthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Biochemistry wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Citric acid cycle wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Microbial metabolism wikipedia , lookup
Electron transport chain wikipedia , lookup
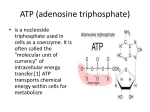
Metabolism: Fueling Cell Growth Chapter 6 Preview • Principles of metabolism – Metabolism, catabolism, anabolism, energy, redox reaction…. • Central metabolic pathway – Glycolysis, TCA • Respiration – Electron transport chain • Fermentation Metabolism • Chemical reactions to keep an organism alive. • Basic needs Principles of Metabolism • Metabolism is broken down into two components – Anabolism – Catabolism • Catabolism – Degradative reactions – Reactions produce energy from the break down of larger molecules • Anabolism – Reactions involved in the synthesis of cell components – Anabolic reactions require energy • Anabolic reactions utilize the energy produced from catabolic reactions Metabolic Pathways Principles of Metabolism Glycolysis TCA Cycle Energy • Definition • Free energy-energy released by breaking chemical bonds – reactants have more free energy • Exergonic reaction – products have more energy • Endergonic reaction Energy source – Compound broken down to release energy – Common energy sources Energy • Oxidizing energy source to release energy Gas +O2 Glucose+O2 CO2+H2O+energy CO2 +H2O +energy Oxidization: gain of oxygen, loss of hydrogen, loss of electron Harvesting Energy •Oxidation/reduction reactions (redox reactions) electron donor electron acceptor LEO - Lose electrons oxidized GER - Gain electrons reduced Protons often follow electrons (i.e. a hydrogen atom is extracted/added; e- + H+ = H ) General rules: •If a compound gains oxygen or loses hydrogen, the reaction is an oxidation •If a compound loses oxygen or gains hydrogen, the reaction is a reduction Harvesting Energy The role of electron carriers “reducing power” In redox reactions, protons often follow electrons Harvesting Energy The role of ATP energy currency Adenosine triphosphate Harvesting Energy The role of ATP energy currency Harvesting Energy Synthesizing ATP •Substrate-level phosphorylation Harvesting Energy Synthesizing ATP •Substrate-level phosphorylation •Oxidative phosphorylation •Photophosphorylation •Other methods involve an electron transport chain and redox reaction Principles of Metabolism Synthesizing ATP •Substrate-level phosphorylation •Oxidative phosphorylation - chemical energy is used to create the proton motive force (involves an electron transport chain); the energy of proton motive force is harvested by making ATP; •Photophosphorylation - radiant energy is used to create the proton motive force (involves an electron transport chain); the energy of proton motive force is harvested by making ATP Central metabolic pathway Central pathways are catabolic and provide • Energy • Reducing power • Precursor metabolites • Central metabolic pathways • Glycolysis • Pentose phosphate pathway • Tricarboxcylic acid cycle Central Metabolic Pathways Glycolysis (aka EmbdenMeyerhoff pathway, glycolytic pathway) glucose 2 pyruvate Central Metabolic Pathways Glycolysis (aka EmbdenMeyerhoff pathway, glycolytic pathway) glucose 2 pyruvate •2 ATP (net gain) •2 spent; 4 made •2 NADH •6 precursor metabolites Central Metabolic Pathways Glycolysis (aka EmbdenMeyerhoff pathway, glycolytic pathway) glucose 2 pyruvate •2 ATP (net gain) •2 spent; 4 made •2 NADH •6 precursor metabolites Central Metabolic Pathways Glycolysis (aka EmbdenMeyerhoff pathway, glycolytic pathway) glucose 2 pyruvate •2 ATP (net gain) •2 spent; 4 made •2 NADH •6 precursor metabolites Central Metabolic Pathways Pentose phosphate pathway glucose intermediate of glycolysis •NADPH (amount varies) •2 precursor metabolites Central Metabolic Pathways Pentose phosphate pathway glucose intermediate of glycolysis •NADPH (amount varies) •2 precursor metabolites Primary role is biosynthesis; ignored in energy-yield calculations; Central Metabolic Pathways Pentose phosphate pathway glucose intermediate of glycolysis •NADPH (amount varies) •2 precursor metabolites Primary role is biosynthesis; ignored in energy-yield calculations; Central Metabolic Pathways Transition step pyruvate (3 C) acetyl CoA (2 C) + CO2 (twice per glucose) Central Metabolic Pathways Transition step pyruvate (3 C) acetyl CoA (2 C) + CO2 (twice per glucose) •NADH •precursor metabolite Central Metabolic Pathways TCA cycle (aka Kreb’s cycle, citric acid cycle) acetyl CoA (2 C) 2 CO2 (twice per glucose) Central Metabolic Pathways TCA cycle (aka Kreb’s cycle, citric acid cycle) acetyl CoA (2 C) 2 CO2 (twice per glucose) •ATP •3 NADH •FADH2 •2 precursor metabolites Central Metabolic Pathways TCA cycle (aka Kreb’s cycle, citric acid cycle) acetyl CoA (2 C) 2 CO2 (twice per glucose) •ATP •3 NADH •FADH2 •2 precursor metabolites Central Metabolic Pathways TCA cycle (aka Kreb’s cycle, citric acid cycle) acetyl CoA (2 C) 2 CO2 (twice per glucose) •ATP •3 NADH •FADH2 •2 precursor metabolites Review of central metabolic pathway Glucose (C6H12O6) Precursor metabolites ATP (substrate-level phosphorylation) Electrons - carried by NADH, FADH2, NADPH (protons often follow, therefore H atoms removed) 6 CO2 Biosynthesis Electron transport chain ATP (oxidative phosphorylation) Glycolysis Pentose phosphate pathway Kreb’s cycle (+ transition step) Oxidation of glucose= Dehydrogenation to CO2+ reducing power (H) Precursor Metabolites Intermediates of catabolism also used in biosynthesis Review Respiration Electron Transport Chain of mitochondria TCA cycle Electron carrier get recycled Electron transport chain Oxidative phosphorylation Part of figure 3.53 Electron Transport Chain of mitochondria Inside of mitochondria FADH2 FAD Terminal electron acceptor Electron Transport Chain of mitochondria Creates the proton motive force FADH2 FAD Electron Transport Chain of mitochondria FADH2 FAD Electron Transport Chain The Mechanics Electron Transport Chain Mitochondrial matrix Intermembrane space (inside) NADH + H+ 2e- (outside) Hydrogen carrier 2H+ Electron carrier Hydrogen carrier Electron carrier 2H+ Hydrogen carrier 2H+ Electron carrier Electron Transport Chain Mitochondrial matrix Intermembrane space (inside) NAD Regenerates NAD (outside) Hydrogen carrier 2e- 2H+ Electron carrier Hydrogen carrier Electron carrier 2H+ Hydrogen carrier 2H+ Electron carrier Electron Transport Chain Mitochondrial matrix Intermembrane space (inside) NAD (outside) Hydrogen carrier 2H+ Electron carrier 2e- Hydrogen carrier Electron carrier 2H+ Hydrogen carrier 2H+ Electron carrier Electron Transport Chain Mitochondrial matrix Intermembrane space (inside) NAD (outside) Hydrogen carrier 2H+ Electron carrier Hydrogen carrier 2e- 2H+ Electron carrier Hydrogen carrier 2H+ Electron carrier Electron Transport Chain Mitochondrial matrix Intermembrane space (inside) NAD (outside) Hydrogen carrier 2H+ Electron carrier Hydrogen carrier 2H+ Electron carrier 2e- Hydrogen carrier 2H+ Electron carrier Electron Transport Chain Mitochondrial matrix Intermembrane space (inside) NAD (outside) Hydrogen carrier 2H+ Electron carrier Hydrogen carrier 2H+ Electron carrier Hydrogen carrier 2e- 2H+ Electron carrier Electron Transport Chain Mitochondrial matrix Intermembrane space (inside) NAD (outside) Hydrogen carrier 2H+ Electron carrier Hydrogen carrier 2H+ Electron carrier Hydrogen carrier Terminal electron acceptor 2H+ Electron carrier 2e- Electron Transport Chain of mitochondria FADH2 FAD Electron Transport Chain of E. coli Aerobic respiration (shown) Anaerobic respiration •NO3 as a TEA (different ubiquinol oxidase) •Quinone used provides humans with vitamin K FADH2 FAD Harvesting Energy The role of electron carriers 12 pairs of electrons (snatched by electron carriers) C6H12O6 + 6 O2 6 CO2 + 6 H2O •Passed to the electron transport chain (used to create the proton motive force); ultimately passed to a terminal electron acceptor (such as O2, making H2O) •Used in biosynthesis (to reduce compounds) e- O2 H 2O Principles of Metabolism Synthesizing ATP •Substrate-level phosphorylation •Oxidative phosphorylation - the energy of proton motive force is harvested; chemical energy is used to create the proton motive force (involves an electron transport chain) ATP synthase e- O2 H 2O ADP + Pi ATP Harvesting Energy Energy source versus terminal electron acceptor Glucose + 6 O2 6 CO2 + 12 H2O Overall Maximum Energy Yield Overall maximum energy yield of aerobic respiration (ignoring the pentose phosphate pathway): Complete oxidation of glucose 4 ATP 10 NADH 2 FADH2 Electron transport chain (oxidative phosphorylation) •3 ATP/NADH •2 ATP/FADH2 Overall Maximum Energy Yield Overall maximum energy yield of aerobic respiration (ignoring the pentose phosphate pathway): Complete oxidation of glucose 4 ATP 10 NADH 2 FADH2 Electron transport chain (oxidative phosphorylation) 38 ATP (maximum theoretical) •3 ATP/NADH •2 ATP/FADH2 Respiration Fermentation •Used when respiration is not an option Lack of TEA •No electron transport chain • •Oxidation of glucose stops at pyruvate •Passes electrons from NADH to pyruvate or a derivative NAD NADH The logic: •Oxidizes NADH, generating NAD for use in further rounds of glucose breakdown •Stops short of the transition step and the TCA cycle, which together generate 5X more reducing power Fermentation Fermentation Review Catabolism of Organic Compounds Other than Glucose (The Elegance of Metabolism) Anabolic Pathways • Synthesis of subunits from precursor metabolites – Pathways consume ATP, reducing power and precursor metabolites – Macromolecules produces once subunits are synthesized Principles of Metabolism • Role of enzymes – Enzymes facilitate each step of metabolic pathway – They are proteins acting as chemical catalysts • Accelerate conversion of substrate to product – Catalyze reactions by lowering activation energy • Energy required to initiate a chemical reaction Enzymes • A specific, unique, enzyme catalyzes each biochemical reaction • Enzyme activity can be controlled by a cell • Enzymes can be exploited medically, industrially • Enzyme names usually reflect the function and end in -ase Enzymes Enzymes Allosteric regulation reversible Enzymes Enzyme inhibition Competitive inhibition - Inhibitor/substrate act at the same site Ex.: PABA folic acid coenzyme Sulfa Enzymes Enzyme inhibition Non-competitive inhibition - Inhibitor/substrate act at different sites •Regulation (allosteric) •Enzyme poisons (example: mercury) Enzymes Environmental factors influence enzyme activity temperature, pH Enzymes Cofactors act in conjunction with certain enzymes Coenzymes are organic cofactors













































































![NAME: Quiz #5: Phys142 1. [4pts] Find the resulting current through](http://s1.studyres.com/store/data/006404813_1-90fcf53f79a7b619eafe061618bfacc1-150x150.png)