* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download No Slide Title
Rosetta@home wikipedia , lookup
Structural alignment wikipedia , lookup
Protein design wikipedia , lookup
Bimolecular fluorescence complementation wikipedia , lookup
Homology modeling wikipedia , lookup
Protein domain wikipedia , lookup
List of types of proteins wikipedia , lookup
Circular dichroism wikipedia , lookup
Protein moonlighting wikipedia , lookup
Protein folding wikipedia , lookup
Protein purification wikipedia , lookup
Western blot wikipedia , lookup
Alpha helix wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Protein–protein interaction wikipedia , lookup
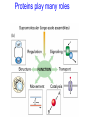
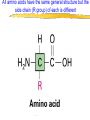
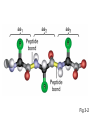
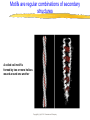
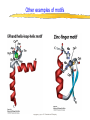
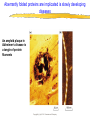
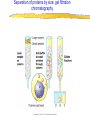
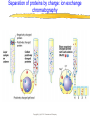
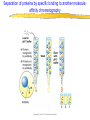
Introduction to Molecular Biology z Molecular biology is interdisciplinary (biochemistry, genetics, cell biology) z Impact of genome projects (human, bacteria, fungi, plants, etc.)….”postgenomics era” z Integration with other fields (e.g. computer science) leading to interdisciplinary career paths (Bioinformatics) Molecular Biology syllabus web site Model Organisms Central Dogma zDNA is transcribed to mRNA zmRNA is translated to protein Proteins play many roles Lecture 1 Protein Structure and Function Reading: Chapters 1-3 Protein Structure & Function - protein structure - protein purification & analysis - protein structure determination Protein structure determines function z Proteins are single, unbranched chains of amino acid monomers z There are 20 different amino acids z A protein’s amino acid sequence determines its three-dimensional structure (conformation) z In turn, a protein’s structure determines the function of that protein All amino acids have the same general structure but the side chain (R group) of each is different Copyright (c) by W. H. Freeman and Company Hydrophilic amino acids Figure 2-13 Copyright (c) by W. H. Freeman and Company Hydrophobic & “special” amino acids Figure 2-13 Copyright (c) by W. H. Freeman and Company Peptide bonds connect amino acids into linear chains Copyright (c) by W. H. Freeman and Company Fig 3-2 Amino acids are the repeating units in proteins, but it is the 3-D protein structure that underlies function. How is 3-D structure obtained? Four levels of structure determine the shape of proteins z Primary: the linear sequence of amino acids z Secondary: the localized organization of parts of a polypeptide chain (e.g., the helix or sheet) z Tertiary: the overall, three-dimensional arrangement of the polypeptide chain z Quaternary: the association of two or more polypeptides into a multi-subunit complex Copyright (c) by W. H. Freeman and Company Secondary structure: the helix The spiral is held by hydrogen bonds between nearly adjucent backbone O and H atoms Figure 3-4 Secondary structure: the beta sheet Hydrogen bonds occur between backbone O and H of separate ajucent strands Copyright (c) by W. H. Freeman and Company Motifs are regular combinations of secondary structures A coiled coil motif is formed by two or more helices wound around one another Copyright (c) by W. H. Freeman and Company Other examples of motifs Copyright (c) by W. H. Freeman and Company Tertiary structure // quaternary structure Regions of proteins form domains: functional, topological or structural (like in case of HA) The structure is stabilized by Interactions between domains Hydrophobic, hydrophylic interactions and disulfide bonds help to keep the structure hemagglutinin Sequence homology suggests functional and evolutionary relationships between proteins Figure 3-10 Folding, modification, & degradation of proteins z A newly synthesized polypeptide chain must undergo folding and often chemical modification to generate the final protein z All molecules of any protein species adopt a single conformation (the native state), which is the most stably folded form of the molecule The information for protein folding is encoded in the sequence Folding of proteins in vivo is promoted by chaperones Aberrantly folded proteins are implicated is slowly developing diseases An amyloid plaque in Alzheimer’s disease is a tangle of protein filaments Copyright (c) by W. H. Freeman and Company Chemical modifications and processing alter the biological activity of proteins Copyright (c) by W. H. Freeman and Company Protein degradation via the ubiquitin-mediated pathway Cells contain several other pathways for protein degradation in addition to this pathway Copyright (c) by W. H. Freeman and Company Functional design of proteins z Protein function generally involves conformational changes z Proteins are designed to bind a range of molecules (ligands) y Binding is characterized by two properties: affinity and specificity z Antibodies exhibit precise ligand-binding specificity z Enzymes are highly efficient and specific catalysts y An enzyme’s active site binds substrates and carries out catalysis Copyright (c) by W. H. Freeman and Company Kinetics of an enzymatic reaction are described by Vmax and Km Copyright (c) by W. H. Freeman and Company Mechanisms that regulate protein function z Allosteric transitions y Release of catalytic subunits, active / inactive states, cooperative binding of ligands z Phosphorylation / dephosphorylation z Proteolytic activation z Compartmentalization Copyright (c) by W. H. Freeman and Company Purifying, detecting, and characterizing proteins z A protein must be purified to determine its structure and mechanism of action z Molecules, including proteins, can be separated from other molecules based on differences in physical and chemical properties Copyright (c) by W. H. Freeman and Company Centrifugation can separate molecules that differ in mass or density Copyright (c) by W. H. Freeman and Company Electrophoresis separates molecules according to their charge:mass ratio SDS-polyacrylamide gel electrophoresis Copyright (c) by W. H. Freeman and Company Two-dimensional electrophoresis separates molecules according to their charge and their mass Copyright (c) by W. H. Freeman and Company Separation of proteins by size: gel filtration chromatography Copyright (c) by W. H. Freeman and Company Separation of proteins by charge: ion exchange chromatography Copyright (c) by W. H. Freeman and Company Separation of proteins by specific binding to another molecule: affinity chromatography Copyright (c) by W. H. Freeman and Company Highly specific enzymes and antibody assays can detect individual proteins Copyright (c) by W. H. Freeman and Company Time-of-flight mass spectrometry measures the mass of proteins and peptides Copyright (c) by W. H. Freeman and Company X-ray crystallography is used to determine protein structure Other techniques such as cryoelectron microscopy and NMR spectroscopy may be used to solve the structures of certain types of proteins Copyright (c) by W. H. Freeman and Company