* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 12_Lecture
Butyric acid wikipedia , lookup
Mitochondrion wikipedia , lookup
Metalloprotein wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Biosynthesis wikipedia , lookup
Electron transport chain wikipedia , lookup
Microbial metabolism wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Biochemistry wikipedia , lookup
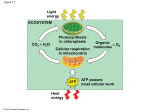
Lecture Presentation Chapter 12 Food as Fuel—A Metabolic Overview Julie Klare Fortis College Smyrna, GA © 2014 Pearson Education, Inc. Outline • 12.1 How Metabolism Works • 12.2 Metabolically Relevant Nucleotides • 12.3 Digestion—From Food Molecules to Hydrolysis Products • 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites • 12.5 The Citric Acid Cycle—Central Processing • 12.6 Electron Transport and Oxidative Phosphorylation • 12.7 ATP Production • 12.8 Other Fuel Choices © 2014 Pearson Education, Inc. 12.1 How Metabolism Works • Animals get energy from the covalent bonds contained in carbohydrates, fats, and proteins. • In the first stage of metabolism, biomolecules in food are digested into smaller units through hydrolysis reactions. • Polysaccharides are hydrolyzed into monosaccharide units. • Triglycerides are broken down to glycerol and fatty acids. • Proteins are hydrolyzed into their amino acid units. © 2014 Pearson Education, Inc. 12.1 How Metabolism Works • The molecules produced by the breakdown are absorbed through the intestinal wall into the bloodstream and transported to different tissues for use by the cells. • In the cells, the hydrolysis products are broken down into a few common metabolites containing two or three carbons. • Metabolites are chemical intermediates formed by enzyme-catalyzed reactions in the body. © 2014 Pearson Education, Inc. 12.1 How Metabolism Works • As long as cells have oxygen and are producing energy, two-carbon acetyl groups can be broken down further to carbon dioxide through the citric acid cycle. • This cycle works to produce the molecules ATP, nicotinamide adenine dinucleotide (NADH), and flavin adenine dinucleotide (FADH2). © 2014 Pearson Education, Inc. 12.1 How Metabolism Works • Chemical reactions that occur in living systems are biochemical reactions. • Chemical reactions occur in a series called a metabolic pathway. • The sugar molecule glucose (containing six carbons) is broken down to two molecules of pyruvate (three carbons each) through a series of chemical reactions referred to as glycolysis. • Metabolism can be considered in two parts, catabolism and anabolism. © 2014 Pearson Education, Inc. 12.1 How Metabolism Works • Catabolism refers to chemical reactions in which larger molecules are broken down into a few common metabolites. These reactions tend to be exergonic (-G). • Anabolism refers to chemical reactions in which metabolites combine to form larger molecules. These reactions tend to be endergonic (+G). • The energy released during catabolic reactions is captured in ATP and used to drive anabolic reactions. © 2014 Pearson Education, Inc. 12.1 How Metabolism Works © 2014 Pearson Education, Inc. 12.1 How Metabolism Works • In animals, a cell membrane separates the materials inside the cell from the exterior aqueous environment. • The nucleus contains DNA that controls cell replication and protein synthesis for the cell. • The cytoplasm consists of all the material between the nucleus and the cell membrane. • The cytosol is the fluid part of the cytoplasm. It is the aqueous solution of electrolytes and enzymes that catalyzes many of the cell’s chemical reactions. © 2014 Pearson Education, Inc. 12.1 How Metabolism Works • Within the cytoplasm are organelles. • Ribosomes are the sites of protein synthesis. • Mitochondria are the energy-producing factories of the cells. • A mitochondrion consists of an outer membrane, an inner membrane, and an intermembrane matrix. • Enzymes in the matrix and inner membrane catalyze the oxidation of carbohydrates, fats, and amino acids. © 2014 Pearson Education, Inc. 12.1 How Metabolism Works © 2014 Pearson Education, Inc. 12.1 How Metabolism Works © 2014 Pearson Education, Inc. 12.2 Metabolically Relevant Nucleotides • Nucleotides act as energy exchangers and can also be coenzymes. • All of these nucleotides have two forms: a high-energy form and a low-energy form. • They consist of some basic components: the nucleoside adenosine, a phosphate, and a five-carbon sugar. Many of these molecules also have a vitamin within their structure. © 2014 Pearson Education, Inc. 12.2 Metabolically Relevant Nucleotides © 2014 Pearson Education, Inc. 12.2 Metabolically Relevant Nucleotides © 2014 Pearson Education, Inc. 12.2 Metabolically Relevant Nucleotides © 2014 Pearson Education, Inc. 12.2 Metabolically Relevant Nucleotides • ATP is often referred to as the energy currency of the cell. • ATP can undergo hydrolysis: during hydrolysis, energy is released as a product, so in this case, ATP is the high-energy form and ADP is the low-energy form. • The energy given off during the hydrolysis of ATP can be coupled to drive a chemical reaction that requires energy. © 2014 Pearson Education, Inc. 12.2 Metabolically Relevant Nucleotides NADH/NAD+ and FADH2/FAD • Nicotinamide adenine dinucleotide (NAD+) and flavin adenine dinucleotide (FAD) are energy-transferring compounds with a high-energy form that is reduced (hydrogen added) and a low-energy form that is oxidized (hydrogen removed). • The abbreviations for these forms are NADH (reduced form) and NAD+ (oxidized form) and FADH2 (reduced form) and FAD (oxidized form). • The active end of each molecule contains a vitamin component. • Nicotinamide is derived from the vitamin niacin (B3), and riboflavin (B2) is found in FAD. © 2014 Pearson Education, Inc. 12.2 Metabolically Relevant Nucleotides Acetyl Coenzyme A and Coenzyme A • Another important energy exchanger is coenzyme A (CoA). • The two forms of this compound are acetyl coenzyme A (high energy) and coenzyme A (low energy). • Energy is released from acetyl coenzyme A when the C—S bond in the thioester functional group is hydrolyzed, producing an acetyl group and coenzyme A. • CoA contains adenosine, three phosphates, and a pantothenic acid (vitamin B5)-derived portion. © 2014 Pearson Education, Inc. 12.3 Digestion—From Food Molecules to Hydrolysis Products Carbohydrates • Starch (amylose and amylopectin) begins to be digested in your mouth by alpha-amylase in saliva. • This salivary amylase hydrolyzes some of the α-glycosidic bonds in the starch molecules, producing glucose, the disaccharide maltose, and oligosaccharides. • Only monosaccharides are small enough to be transported into the bloodstream. • To complete the digestion of starch, enzymes in the small intestine hydrolyze starch and disaccharides into monosaccharides. • Cellulose cannot be digested because we lack the enzyme cellulase that hydrolyzes its β-glycosidic bonds. © 2014 Pearson Education, Inc. 12.3 Digestion—From Food Molecules to Hydrolysis Products © 2014 Pearson Education, Inc. 12.3 Digestion—From Food Molecules to Hydrolysis Products Fats • Dietary fats are nonpolar molecules, so to assist in digestion, bile is excreted from the gall bladder into the stomach during digestion. • Bile contains bile salts, which are amphipathic: they place their nonpolar face toward the dietary fats and their polar face toward the water, forming micelles. • Breaking up larger nonpolar globules into smaller droplets (micelles) is called emulsification. • The micelles move the dietary fats closer to the intestinal cell wall so cholesterol can be absorbed and triglycerides hydrolyzed. • Once across the intestinal wall, free fatty acids and monoglycerides are reassembled as triglycerides while the cholesterol is linked to another free fatty acid forming a cholesterol ester. • These are repackaged as a lipoprotein called a chylomicron. • Chylomicrons transport triglycerides to the tissues, where they are used for energy production or stored. © 2014 Pearson Education, Inc. 12.3 Digestion—From Food Molecules to Hydrolysis Products © 2014 Pearson Education, Inc. 12.3 Digestion—From Food Molecules to Hydrolysis Products Proteins • • • Protein digestion begins in the stomach, where proteins are denatured (unfolded) by the acidic digestive juices. Digestive enzymes like pepsin, trypsin, and chymotrypsin hydrolyze peptide bonds. Amino acids are absorbed into the bloodstream for delivery to the tissues. © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites The Chemical Reactions in Glycolysis • In the body, energy must be transferred in small amounts to minimize the heat released in the process. • Reactions that produce energy are coupled with reactions that require energy, thereby helping to maintain a constant body temperature. • In glycolysis, energy is transferred through phosphate groups undergoing condensation and hydrolysis reactions. There are 10 chemical reactions in glycolysis that result in the formation of two molecules of pyruvate from one molecule of glucose. © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites The Chemical Reactions in Glycolysis • The first five reactions require an energy investment of two molecules of ATP, which are used to add two phosphate groups to the sugar molecule. • This molecule is split into two sugar phosphates. • Reactions 6 through 10 generate two high-energy NADH molecules during the addition of two more phosphates and four ATP molecules when the four phosphates are removed from the sugar phosphates. © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites Regulation of Glycolysis • The main step of regulation in glycolysis is step 3. • The enzyme phosphofructokinase, which catalyzes the phosphorylation of fructose-6phosphate to fructose-1,6-bisphosphate, is heavily regulated by the cells. • ATP acts as an inhibitor of phosphofructokinase. • If cells have plenty of ATP, glycolysis slows down. © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites The Fates of Pyruvate • Aerobic conditions: pyruvate produces more energy for the cell when the carboxylate functional group of pyruvate is liberated as CO2 during oxidative decarboxylation. • The acetyl group binds to coenzyme A during the oxidation through a sulfur atom, creating a thioester functional group and acetyl CoA. • This reaction occurs in the mitochondria. © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites The Fates of Pyruvate • Anaerobic conditions: the middle carbonyl in pyruvate is reduced (hydrogen added) to an alcohol group, and lactate is formed. • The hydrogen (and energy) required for this reaction is supplied by NADH and H+, producing NAD+. • The NAD+ produced funnels back into glycolysis to oxidize more glyceraldehyde-3-phosphate (step 6), providing a small amount of ATP. This reaction occurs in the cytosol. © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites • Yeast converts pyruvate to ethanol under anaerobic conditions. • This process is called fermentation. • In the preparation of alcoholic beverages, yeast produces pyruvate from glucose in grape juices and under low-oxygen conditions transforms pyruvate into ethanol. © 2014 Pearson Education, Inc. 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites Fructose and Glycolysis • Fructose is readily taken up in the muscle and liver. • In the muscles, it is converted to fructose-6-phosphate, entering glycolysis at step 3. In the liver, it is converted to the trioses used in step 5. • Fructose that enters a cell flows from reaction 5 to 10. • Fructose uptake by the cells is not regulated by insulin: all fructose in the bloodstream is forced into catabolism. • Glycolysis is regulated at step 3. The triose products created in the liver provide an excess of reactants that create excess pyruvate and acetyl CoA that, if not required for energy by the cells, is converted to fat. © 2014 Pearson Education, Inc. 12.5 The Citric Acid Cycle—Central Processing • During aerobic catabolism, glucose, amino acids, and fatty acids funnel into and out of the citric acid cycle. • The citric acid cycle degrades two-carbon acetyl groups from acetyl CoA into CO2 and generates the high-energy molecules NADH and FADH2. • The initial reaction is a condensation reaction between acetyl CoA and the four-carbon molecule oxaloacetate. • The six-carbon citrate loses first one and then a second carbon as CO2, forming the four-carbon succinyl CoA. • These carbon–carbon bond-breaking reactions transfer energy and produce NADH from the coenzyme NAD+. • Succinyl CoA then runs through a set of reactions regenerating oxaloacetate, and the cycle begins again. © 2014 Pearson Education, Inc. 12.5 The Citric Acid Cycle—Central Processing Reactions of the Citric Acid Cycle • Reaction 1, Formation of Citrate: The acetyl group from acetyl CoA (two carbons) combines with oxaloacetate (four carbons), forming citrate (six carbons) and CoA. • Reaction 2, Isomerization to Isocitrate: The –OH and one of the –H atoms are swapped in citrate to form isocitrate. This rearrangement is necessary because isocitrate is oxidized in the next reaction. • Reaction 3, First Oxidative Decarboxylation (Release of CO2): An alcohol undergoes oxidation (two hydrogens removed) to a ketone called -ketoglutarate, and NAD+ is reduced to NADH, accepting the proton and electrons removed during the oxidation. The six-carbon isocitrate is decarboxylated to the five-carbon -ketoglutarate. © 2014 Pearson Education, Inc. 12.5 The Citric Acid Cycle—Central Processing Reactions of the Citric Acid Cycle • Reaction 4, Second Oxidative Decarboxylation: The thiol group of CoA is oxidized (loses a hydrogen), and another NAD+ is reduced to NADH. Alpha-ketoglutarate (five carbons) is decarboxylated into a succinyl group (four carbons). The CoA is bonded to the succinyl group, thus producing succinyl CoA. • Reaction 5, Hydrolysis of Succinyl CoA: Succinyl CoA undergoes hydrolysis to succinate and coenzyme A. The energy produced produces the high-energy nucleotide guanosine triphosphate or GTP from GDP and Pi. GTP is converted to ATP in the cell. © 2014 Pearson Education, Inc. 12.5 The Citric Acid Cycle—Central Processing Reactions of the Citric Acid Cycle • Reaction 6, Dehydrogenation of Succinate: One hydrogen is eliminated from each of the two central carbons of succinate, forming a trans C=C bond, thus producing fumarate. These two hydrogens reduce the coenzyme FAD to FADH2. • Reaction 7, Hydration of Fumarate: Water adds to the trans double bond of fumarate as –H and –OH forming malate. • Reaction 8, Oxidation of Malate: As in reaction 3, the secondary alcohol of malate is oxidized to a ketone forming oxaloacetate, providing protons and electrons to reduce the coenzyme NAD+ to NADH. © 2014 Pearson Education, Inc. 12.5 The Citric Acid Cycle—Central Processing • One turn of the citric acid cycle produces a net energy output of three NADH, one FADH2, and one GTP (which forms ATP). • Two CO2 and one CoA also are produced. • The net reaction for one turn of this eight-step cycle is © 2014 Pearson Education, Inc. 12.5 The Citric Acid Cycle—Central Processing © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation • Two ATP are produced in glycolysis and two ATP in the citric acid cycle. Where is all the energy? • NADH and FADH2 are produced in glycolysis (two NADH per glucose), from pyruvate oxidation to acetyl CoA (two NADH per glucose), and in the citric acid cycle (six NADH and two FADH2 per glucose). • High-energy reduced forms of the nucleotides transfer electrons and hydrogens through the inner mitochondrial membrane and to form H2O. • The energy generated as a result of this process is used to drive the reaction of ADP to form ATP. • This is called oxidative phosphorylation. © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation • Mitochondria are the ATP factories of the cell. • Reduced nucleotides from the citric acid cycle are produced here, and their energy upon oxidation is used to generate ATP. • The reactions of the citric acid cycle occur in the matrix of the mitochondria. The reduced nucleotides, NADH and FADH2, begin their journey through the inner membrane here. • Enzyme complexes I through V are embedded in the inner membrane of the mitochondria and electron carriers that transport the electrons and protons of NADH and FADH2 through the inner mitochondrial membrane. • Two of the electron carriers, coenzyme Q and cytochrome c, are not firmly attached to any one complex and shuttle electrons between the complexes. © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation • Complex I, NADH Dehydrogenase: At complex I, NADH enters electron transport. During its oxidation, two electrons and two protons are transferred to the electron transporter coenzyme Q, reducing its two ketone groups to alcohols (see figure at left). NAD+ is regenerated and returns to a catabolic pathway as in the citric acid cycle. The overall reaction at complex I is NADH + H+ + Q → NAD + + QH2 © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation • Complex II, Succinate Dehydrogenase: FADH2 enters electron transport after the reduced nucleotide is produced in the conversion of succinate to fumarate in the citric acid cycle. Two electrons and two protons from FADH2 are also transferred to coenzyme Q to yield QH2. FADH2 + Q → FAD + QH2 • Complex III, Coenzyme Q—Cytochrome c Reductase: At complex III, the reduced coenzyme Q (QH2) molecules are reoxidized to ubiquinone (Q), and the electrons pass through a series of electron acceptors until they arrive at cytochrome c, which moves the electron from complex III to complex IV. © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation • Complex IV, Cytochrome c Oxidase: At complex IV, single electrons are transferred from cytochrome c through another set of electron acceptors to combine with hydrogen ions and oxygen (O2) to form water. This is the final stop for the electrons: 4H+ + 4e− + O2 → 2H2O © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation • Oxidative phosphorylation: The chemiosmotic model links electron transport to the generation of a proton (H+) gradient across the inner membrane. • In this model, three of the complexes (I, III, and IV) span the inner membrane and pump (relocate) protons out of the matrix and into the intermembrane space as electrons are shuttled through the complexes. • The formation of the proton gradient across the inner mitochondrial membrane provides the energy for ATP synthesis. • Protons move back into the matrix through a protein complex, called complex V, or ATP synthase. As protons flow back into the matrix through complex V, the resulting release of energy drives the synthesis of ATP. © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation © 2014 Pearson Education, Inc. 12.6 Electron Transport and Oxidative Phosphorylation Thermogenesis • If ATP cannot be produced, the energy that would have been harnessed as ATP is released as heat. This is called thermogenesis. • Some animals adapted to cold climates produce small organic molecules called uncouplers, which uncouple electron transport and oxidative phosphorylation and thereby assist in regulating their body temperature through thermogenesis. • These animals have a higher amount of a tissue called brown fat, which appears brown due to the high concentrations of mitochondria present. The cytochrome molecules present in mitochondria contain an iron ion that is responsible for the brown color. • Newborn babies have higher levels of brown fat than do adults because newborns do not have much stored fat. Brown fat deposits are located near major blood vessels that carry the warmed blood through the body, allowing a newborn to generate more heat to warm its body surface. © 2014 Pearson Education, Inc. 12.7 ATP Production Glycolysis: The oxidation of glucose produces two NADH molecules and two ATP molecules. • Glycolysis occurs in the cytosol, and electron transport draws NADH from the matrix inside the mitochondria. • The two NADH from glycolysis must be shuttled into the matrix to enter electron transport. This results in the production of five ATP. Oxidation of pyruvate: After glycolysis, the two pyruvates enter the mitochondria, where they are oxidized to produce two acetyl CoA, two CO2, and two NADH. The oxidation of two pyruvates leads to the production of five ATP. © 2014 Pearson Education, Inc. 12.7 ATP Production © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices • If glycogen and glucose are not available, cells can oxidize fatty acids to acetyl CoA through beta oxidation (β oxidation). • β oxidation includes four reactions that convert the –CH2– of the carbon to a ketone. • Once this ketone is formed, the two-carbon acetyl group splits from the fatty acyl carbon chain. One cycle of β oxidation yields one FADH2 and one NADH. © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices Reaction 1, Oxidation (Dehydrogenation). The first reaction removes one hydrogen from the alpha and beta carbons, and a double bond is formed. These hydrogens are transferred to FAD to form FADH2. Reaction 2, Hydration. In reaction 2, water is added to the and β carbon double bond as –H and –OH, respectively. Reaction 3, Oxidation (Dehydrogenation). The alcohol formed on the β carbon is oxidized to a ketone. As we have seen before in the citric acid cycle, the hydrogen from the alcohol reduces NAD+ to NADH. Reaction 4, Removal of Acetyl CoA. In the fourth reaction of the cycle, the bond between the and β carbon is broken and a second CoA is added, forming an acetyl CoA and a fatty acyl CoA shortened by two carbons. The fatty acyl CoA can be run through the cycle again. © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices Ketosis • • • • • • In the absence of carbohydrates, the body breaks down body fat through oxidation to continue ATP production. The liver will produce glucose from pyruvate through gluconeogenesis for tissues like the brain. The oxidation of large amounts of fatty acids can cause acetyl CoA to accumulate in the liver. When accumulation occurs, the two carbon acetyl units condense in the liver, forming the four-carbon ketone molecules β-hydroxybutyrate and acetoacetate and the molecule acetone. These are collectively referred to as ketone bodies. Ketosis occurs when an excessive amount of ketone bodies is present in the body. Because two of the ketones are carboxylic acids, the excessive formation of ketone bodies can cause metabolic acidosis. Acetone vaporizes easily, giving someone suffering from ketosis an odd, sweet-smelling breath upon exhalation similar to that of someone who has been drinking alcohol. © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices Energy from Amino Acids • Amino acids produce nitrogen when metabolized in the body. When excess protein is ingested, amino acids must be degraded. The α-amino group of an amino acid is removed, yielding an α-keto acid through transamination. • The α-keto acid can be converted into intermediates for other metabolic pathways. • The ammonium ions produced in this process must be excreted from the body. • The urea cycle converts ammonium ions (NH4+) into urea, which can be excreted in the urine. © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices Energy from Amino Acids • Amino acids offer a way to replenish the intermediates in the citric acid cycle. • Amino acids like alanine, containing three carbons, can enter the pathways as pyruvate. • Amino acids with four carbons are converted to oxaloacetate. • Five-carbon amino acids are converted to -ketoglutarate. • Some amino acids can enter at more than one point depending on cellular requirements. • Amino acids provide only about 10% of the required energy under normal conditions. © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices Putting It Together: Linking the Pathways • Degradation of food biomolecules begins with digestion. • When the cell requires energy and oxygen is plentiful, larger molecules are metabolized into smaller metabolites that ultimately funnel into the citric acid cycle, electron transport, and oxidative phosphorylation. • Through anabolic pathways, larger molecules can be synthesized from the smaller metabolites when necessary. © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices Putting It Together: Linking the Pathways • The biological hydrolysis products can be shifted into anabolic or catabolic pathways depending on the requirements of the cell. • Glucose can be degraded to acetyl CoA entering the citric acid cycle to produce energy or be converted to glycogen for storage in the cells. • Amino acids provide nitrogen for anabolism of nitrogen compounds, but their carbons can enter the citric acid cycle as α-keto acids if necessary. © 2014 Pearson Education, Inc. 12.8 Other Fuel Choices © 2014 Pearson Education, Inc. Chapter Twelve Summary 12.1 How Metabolism Works • Metabolism refers to biochemical reactions occurring in the body. • Catabolism refers to reactions that break down larger molecules into smaller ones. Catabolic reactions are exergonic overall, and the processes are oxidative. • Anabolism refers to reactions that synthesize larger biological molecules from smaller ones. Anabolic reactions are endergonic overall and are reductive. • In the body, biochemical reactions are usually grouped into pathways. Biochemical reactions that produce energy tend to be coupled to reactions requiring energy. • Metabolic pathways tend to be compartmentalized in different parts of the cell. • The mitochondria are the energy-producing factories of the cells. © 2014 Pearson Education, Inc. Chapter Twelve Summary 12.2 Metabolically Relevant Nucleotides • Nucleotides are used in metabolism to transfer energy throughout the cell. ATP is considered the main energy currency of the cell and produces energy when hydrolyzed to ATP + Pi. NADH and FADH2 contain a nucleotide portion and a vitamin portion. They are important coenzymes that transport hydrogens and electrons in the cell. Coenzyme A (CoA) also contains a nucleotide and vitamin portion. Each of these nucleotides has a high-energy and a low-energy form. 12.3 Digestion—From Fuel Molecules to Hydrolysis Products • Food molecules are broken down into their component parts through hydrolysis prior to absorption into the bloodstream. • Monosaccharides and amino acids travel directly through the bloodstream to the cells for absorption. • Triglycerides are packaged into lipoproteins called chylomicrons for delivery. © 2014 Pearson Education, Inc. Chapter Twelve Summary 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites • Glycolysis is a series of 10 reactions that catabolizes a six-carbon glucose molecule to two three-carbon pyruvate molecules. These 10 reactions yield two ATP and two NADH molecules per glucose. • Under aerobic conditions, pyruvate is further oxidized in the mitochondria to acetyl CoA. • In the absence of oxygen, pyruvate is reduced to lactate and regenerates NAD+ so that glycolysis can continue. • Glucose can be stored as glycogen in the liver and muscle for later use. • Fructose can undergo glycolysis. In the liver, its catabolism is unregulated and can produce excess products that become stored in the body as fat. © 2014 Pearson Education, Inc. Chapter Twelve Summary 12.5 The Citric Acid Cycle—Central Processing • The citric acid cycle occurs in the mitochondrial matrix and combines acetyl CoA (two carbons) with oxaloacetate (four carbons), producing citric acid. • Citric acid undergoes several oxidations, decarboxylation, dehydrogenation and a hydration yielding two CO2, GTP, three NADH, and one FADH2. The cycle regenerates oxaloacetate to begin again. GTP readily converts to ATP in the cell. © 2014 Pearson Education, Inc. Chapter Twelve Summary 12.6 Electron Transport and Oxidative Phosphorylation • The reduced nucleotides NADH and FADH2 become oxidized, transporting H+ and electrons through a series of enzyme complexes in the inner mitochondrial membrane. • The final electron acceptor in this process is O2, which combines with H+ to form H2O. The complexes act as proton pumps, moving protons from the matrix to the inner membrane space during electron transport. • This produces an electrochemical gradient. The protons can return to the matrix through complex V, ATP synthase, which generates ATP. • This process is known as oxidative phosphorylation. • When ATP and proton transport are uncoupled, the energy in the proton gradient is released as heat, which assists in maintaining body temperature in a process called thermogenesis. © 2014 Pearson Education, Inc. Chapter Twelve Summary 12.7 ATP Production • For every four H+ pumped into the inner membrane space and returned to the matrix, one ATP can be synthesized. • The oxidation of one NADH provides enough energy to synthesize 2.5 ATP. One FADH2 provides energy to synthesize 1.5 ATP. • Under aerobic conditions, the complete oxidation of one molecule of glucose produces a total of 32 ATP. © 2014 Pearson Education, Inc. Chapter Twelve Summary 12.8 Other Fuel Choices • Fatty acids produce ATP when glucose supplies are low. Fatty acids link to coenzyme A, forming activated fatty acyl CoA, which is transported to the mitochondria for catabolism in a reaction cycle called β oxidation. • Fatty acyl CoA is oxidized, producing a new fatty acyl CoA that is two carbons shorter and one molecule of acetyl CoA. Each turn of β oxidation produces one NADH and one FADH2. • High levels of acetyl CoA in the cell activate the ketogenesis pathway, forming ketone bodies that can lead to ketosis and acidosis. • Amino acids can produce ATP when other fuel supplies are low and the cell does not require other nitrogen-containing compounds. • When amines are removed from amino acids as ammonium ions, they are converted to urea for excretion. • The carbons from amino acids can feed into oxidative catabolism as different intermediates depending on the amino acid. © 2014 Pearson Education, Inc. Chapter Twelve Study Guide 12.1 Overview of Metabolism – Distinguish catabolism from anabolism. – Identify reactions as catabolic or anabolic. – Name the parts of a cell associated with metabolism. 12.2 Metabolically Relevant Nucleotides – Identify the metabolically relevant nucleotides. – Distinguish the low-energy and high-energy forms of the relevant nucleotides. 12.3 Digestion—From Food Molecules to Hydrolysis Products – Compare digestion of carbohydrates, lipids, and proteins. © 2014 Pearson Education, Inc. Chapter Twelve Study Guide 12.4 Glycolysis—From Hydrolysis Products to Common Metabolites – Follow a molecule of glucose through the ten reactions of glycolysis. – Discuss anaerobic and aerobic fates of pyruvate. – Contrast glycolysis for glucose and fructose. 12.5 The Citric Acid Cycle—Central Processing – Identify the reactions in the citric acid cycle. – List the energy output in the citric acid cycle. 12.6 Electron Transport and Oxidative Phosphorylation – Describe the function of each enzyme complex (I–IV) during electron transport. – Discuss the function of coenzyme Q and cytochrome c. – Describe the production of ATP at complex V using the chemiosmotic model. © 2014 Pearson Education, Inc. Chapter Twelve Study Guide 12.7 ATP Production – Convert the number of reduced nucleotides produced (NADH, FADH2) to a corresponding number of ATP. – Calculate the number of ATP produced during the oxidative catabolism of a molecule of glucose. 12.8 Other Fuel Choices – Calculate the number of ATP produced from a saturated fatty acid undergoing oxidation. – Describe the metabolic pathways of oxidation, transamination, and the urea cycle. – Identify catabolic and anabolic pathways in the cell. © 2014 Pearson Education, Inc. Reaction Summary © 2014 Pearson Education, Inc. Reaction Summary © 2014 Pearson Education, Inc.