* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download The Quantum Mechanics of MRI
Molecular Hamiltonian wikipedia , lookup
History of quantum field theory wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
Wave function wikipedia , lookup
Renormalization group wikipedia , lookup
Magnetic monopole wikipedia , lookup
Canonical quantization wikipedia , lookup
Renormalization wikipedia , lookup
Atomic orbital wikipedia , lookup
Electron configuration wikipedia , lookup
Particle in a box wikipedia , lookup
Magnetoreception wikipedia , lookup
Electron paramagnetic resonance wikipedia , lookup
Spin (physics) wikipedia , lookup
Atomic theory wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Matter wave wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Electron scattering wikipedia , lookup
Wave–particle duality wikipedia , lookup
Hydrogen atom wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Ferromagnetism wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
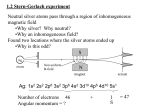
The Quantum Mechanics of MRI Part 1: Basic concepts David Milstead Stockholm University Background reading • Fundamentals of Physics, Halliday, Resnick and Walker (Wiley) • The Basics of NMR, J. Hornak (http://www.cis.rit.edu/htbooks/nmr/inside. htm ) Outline • What is quantum mechanics – Wave-particle duality – Schrödinger’s equation – Bizarreness • uncertainty principle • energy and momentum quantisation • precession of angular momentum What is quantum mechanics ? Basic concepts • • • • What is a wave ? What is a particle ? What is electromagnetic radiation ? What happens to a magnet in a magnetic field ? What is a wave ? Double slit diffraction Properties of waves: Superposition, no localisation (where is a wave??) What is electromagnetic radiation ? EM radiation is made up of electromagnetic waves of various wavelengths and frequencies Radio frequency is of most interest to us Circularly polarised light Rotating magnetic field Radiation can be produced and filtered to produce a rotating magnetic field. Light is a wave! Photon Photons Quanta of light: Energy E hf h=Planck's constant=6.626 1034 Js f =frequency v = f =wavelength, v speed Wave-particle Duality Particles also show wave-like properties Wave-length = h p Energy=hf f =frequency, p=momentum, h=6.6 1034 Js Planck's constant. We’ve just learned a basic result of quantum mechanics. Now we move onto some maths. Fundamental equation of quantum mechanics x wave function E =particle energy U =potential energy of particle Free particle A particle not confined or subject to forces. Re( x ) U 0 ; Fixed E 2 E 2m x 2 A cos kx i sin kx 2 x constant - particle can be anywhere! 2 2 p2 p ;k ; E 2m fixed wavelength and momentum h Well defined wavelength and momentum but no preferential position. "Uncertainty on position": x "Uncertainty on momentum": p 0 Wave function of a confined particle x Eg particle trapped in a tiny region of space. How do we model the wave function ? p p Solution to Schrödinger’s equation would be sum lots of sine waves with different wavelengths/momenta. x x Similarly: p y y h pz z h Question A 12-g bullet leaves a rifle at a speed of 180m/s. a) What is the wavelength of this bullet? b) If the position of the bullet is known to an accuracy of 0.60 cm (radius of barrel), what is the minimum uncertainty in its momentum? c) If the accuracy of the bullet were determined only by the uncertainty principle (an unreasonable assumption), by how much might the bullet miss a pinpoint target 200m away?. h 6.6 1034 (a) Wavelength 3.06 1034 m p 0.012 180 h 6.6 1034 (b) p 1031 kgms1 x 0.006 p 1031 (c) Displacement 200 200 9 1031m Tiny! p 0.12 180 Because h is so small, quantum effects are never seen for macroscopic objects. Hydrogen atom Solve Schrödinger’s equation for an electron around a proton in a hydrogen atom. The electron is confined due to a Coulomb potential. e 2 U 4 0 r e proton charge Component along one direction: Lz ml ml -l , - l 1 ,...., l 1, l (2l 1) different states. where h 2 Where is the electron ? Wave functions Current loops and magnetic dipoles + - Current loop N S Bar magnet Orbiting electron as a current loop Tiny current loop. dq charge e current I dt period T Magnetic moment IAnˆ e A area of loop 1 v ev ev evR IA A R2 T 2 R 2 R 2 R 2 Angular momentum: L me vr l R e L (parallel to angular momentum) 2me Gyrom agnetic ratio: l L =- e 2me nˆ into the page. eml eLz the electron Recall that U Bz Bz has fixed values of LZ 2m 2m An atom in a magnetic field • l=1 and therefore 2l+1 states E (1) 2 z Bz (2) (3) U -l • B -lz Bz (1) ml 1 lz e 2me lz eml 2me ; U e Bz (l antiparallel with B) 2me (2) ml 0 lz 0 ; U 0 (3) ml 1 lz e e Bz ; U Bz (l parallel with B ) 2me 2me Two states with shifted energy one state with no energy shift. l=1 l=0 What happens when a circulating electron is placed in a magnetic field. We know that LZ , L2 are fixed and known. L2x L2y L2 L2Z l l 1 ml2 2 constant z By symmetry, same spread of results of Lx or Ly when measurements of made. i.e. just as likely to find angular momentum X and Y . Magnetic field introduces a torque l B l B sin dL dt B d 1 dL 1 l B sin l dt L sin dt L sin L 2me eB L l e 2me y x LZ = X i.e. the angular momentum vector precesses around the magnetic field (z -axis) with frequency . This is Larmor precession. e Gyromagnetic ratio : B 2me U Angular momentum precession LZ constant and L precesses around z-axis Question Is it possible for an electron in a hydrogen atom to have L LZ ? Show that this value can be approached for large values of l. Would L LZ be possible using classical physics ? No - it can never happen otherwise we would "know" values of Lx , Ly i.e. Lx Ly 0 L2 l l 1 LZ ml L 2 For large l L2 l 2 2 Ll Max LZ l LZ (comes close) In classical physics there is no restriction on what we can and can't know. The mathematics of spin angular momentum is identical to orbital angular momentum. FN Magnetic field FS FN Ignore force not parallel to North-South axis. FS Magnetic field Magnetic field Magnetic field Magnetic field Zeeman effect with orbital and spin angular momentum In the presence of a magnetic field, multiplicities of ”spectral lines” appear 2 B B 1 2 number of lines: n 2s 1 2 Eg for l 0, s Larmor precession for spin Identical to orbital case. s ; S B s spin magnetic moment Electron: e e ge 2me z g e electron g-factor 2, me electron mass. Nucleus: e N gN 2m p (next lecture) y x g nuclear g-factor, m p proton mass. SZ S S sin Summary • Established basic quantum mechanics theory needed for NMR – wave-particle duality • Light is either photons or electromagnetic waves – Schrödinger’s equation and the wave function at the heart of QM predictions – Energy and angular momentum are quantised • Larmor precession – Angular momentum comes in two varieties (orbital and spin) The Quantum Mechanics of MRI Part 2: Understanding MRI David Milstead Stockholm University Outline • Spin - reminder • Fermions and bosons • Nuclear energy levels Identical in form to orbital case. S 2 s ( s 1) 2 Spin S z ms B s S s spin magnetic moment Electron: e e ge 2me z g e electron g-factor 2, me electron mass. y Nucleus: e N gN 2m p (next lecture) x g nuclear g-factor, m p proton mass. S SZ S sin Gyromagnetic ratio Why do they have different values ? Fermions and bosons • Fermions – Spin 1/2, 3/2, 5/2 objects – Electrons, protons and neutrons have spin 1/2 – Tricky bit comes when combining their spins to form the spin of, eg, an atom or a nucleus • Bosons – Integer spin objects Similar shell structure for nuclear physics as for atomic physics • Need to fill up, shell by shell • Pairs of protons and neutrons cancel each other’s spins. • Pauli’s exclusion principle ensures that many shells are filled. • Nuclei with uneven (even) atomic number have half-integer (integer) spin • Nuclei with even atomic and mass numbers have zero spin. • Unpaired neutrons/protons provide the spin for MRI. Question Nature would prefer all electrons to be in the lowest shell and all nucleons protons/neutrons) in the lowest shell ? Why doesn’t this happen ? Usefulness for MRI • Need isotopes with unpaired protons (to produce signal for MRI) • Most elements have isotopes with nonzero nuclear spin • Natural abundance must be high enough for MRI to be performed. Spins of various nuclei Now we can understand MRI We know the basics: A uniform magnetic field B0 A short pulse of a rf field B1 A system out of equilibrium measurement of return to equilibrium. Why is (a) B1 B0 ? (b) The rf pulse shorter than recovery time ? No magnetic field Apply an external magnetic field B0 Spins precess at Larmor frequency . Gyromagnetic ratio: B Precessions incoherent: Total spin S S Z , S x S y 0 SZ 1 2 In fact, there are continual transitions and interactions from thermal energy. Another look at the system Split into ”spin-zones”. For uniform system we can regard the macroscopic system as giving a single magnetisation. Conventional to talk about magnetisation M : N M s ; N no. dipoles, V volume V Putting together what we’ve learned Need to know its energy splitting. s S e gN 2m p S =spin angular momentum g nuclear g-factor , m p proton mass. E sZ B0 ; sZ S z E 2 sZ B0 g pe 2m p e 1 e gN gN 2m p 2 4m p B0 0 Frequency of light needed for excitation 0 Larmor frequency: B0 g pe 2m p B0 eB0 g N 0 2m p How many nuclei can be excited ? 0 0 Energy level population Nature has a preference for the lowest energy states. In thermal equilibrium the lowest states are a bit more populated than the higher energy states. 2 x Taylor expansion: e x 1 x ... 2! E N E e 1 N k BT k BT k B Boltzmann's constant = 1.38 10 23 J/K T =room temperature E g pe 2m p 310 K B0 B0 0 B0 1.5 T 42.576 106 2 E 4.2 1026 J N 1.000009 N N N g pe N N N N 1 B0 N 2 2 m p k BT Changing the spin populations Tiny differences in, eg, 0.02ml of water 15 expect 6×10 more in parallel state. Question Chemists can excite certain samples using UV light. Would you expect N to be substantially different in this case ? What does this imply for N the relative size of the sample of a MRI scan and a test made by chemists with infrared light ? 3 108 UV-light 10 m f 3 1016 s 1 8 10 E 6.6 1034 3 1016 2 1017 J 8 c N E 2 1017 1 1 1 5000 N k BT 1.4 1023 300 In fact, approximation no longer valid since it is no longer a small difference!! A chemist can use far smaller samples since the energy gap is larger and there are far more in the lower energy state. Exciting the nuclei - Rf pulse Rotating magnetic field B1 Typical pulse duration ~1ms. Two ways to think about the pulse. Both are needed to understand MRI. Excitation B0 B1 Pulse of rf- Rotating magnetic field B1 The rf pulse acts in two ways: (1) The photons are absorbed, saturating the system and reducing M Z 0. (2) The rotating magnetic field B1 acts on the magnetisation vector to rotate into the complex plane. Individual dipole: s B1 ; Use magnetisation: dS 1 d s s B1 dt dt 1 dM M B1 - simple form of the Bloch equations. dt Rotating frame of reference B0 B1 X’ Easier to understand if a rotating frame of reference (with Larmor frequency) is used. Rotate x, y co-ordinates x ', y ' Magnetic field constant on x ' axis. y’ Different types of pulses 90o 180o Duration of pulse and size of B1 determine angle of rotation of magnetisation: Spins precess around B1 t Consider a full rotation of 2 in 1ms B1 2 42.58 MHz/T 1 1 103 42.58 106 1T B1 B0 . B1 B0 105 T Question When applying B1 a new magnetic field is obtained: Bn B1 B0 . Why don't the protons just align with respect to that field ? B0 Bn What happens next ? Consider a 90o rotation. The magnetisation vector is in the x - y plane. The rf -pulse is turned off. The system must return to equilibrium. We have two components: M Z and M xy . Longitudinal relaxation Spin-lattice effect: higher energy state interacts with lower energy state due and lose energy through rotation and vibration . Relaxation times for different materials Transverse relaxation In the laboratory frame Dipole moments are initially in phase. Mxy precesses and decays. As it precesses phase decoherence occurs. Complicated process: contributing factor non-uniform magnetic field over sample different precession rates for different regions. Free induction decay As the transverse magnetisation decays, a changing magnetic field is produced: =an emf is produced in a receiver. t The signal is a decaying sinusoidal wave with lifetime T2* . Measurement of gives information on composition of sample. Free induction decay Easier to interpret a single line on a "frequency" spectrum. Use a Fourier transform to move from damped exponential to signal. Summary • Basic quantum mechanics at the heart of nuclear magnetic resonance – Angular momentum quantisation – Energy quantisation • Features of a MRI experiment investigated.