* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Problem set 3
Nitrogen-vacancy center wikipedia , lookup
Quantum entanglement wikipedia , lookup
Hidden variable theory wikipedia , lookup
Path integral formulation wikipedia , lookup
Measurement in quantum mechanics wikipedia , lookup
Renormalization group wikipedia , lookup
Coherent states wikipedia , lookup
Atomic orbital wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Noether's theorem wikipedia , lookup
Coupled cluster wikipedia , lookup
Wave function wikipedia , lookup
Dirac equation wikipedia , lookup
Density matrix wikipedia , lookup
Bell's theorem wikipedia , lookup
Ferromagnetism wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
EPR paradox wikipedia , lookup
Particle in a box wikipedia , lookup
Canonical quantization wikipedia , lookup
Quantum state wikipedia , lookup
Hydrogen atom wikipedia , lookup
Spin (physics) wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
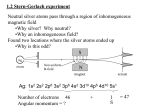
Quantum Mechanics 2, Autumn 2011 CMI Problem set 3 Due by beginning of class on Monday September 5, 2011 Angular momentum and spin 1. Recall that the angular momentum raising operator is L+ = ~eiφ (∂θ + i cot θ ∂φ ). Use this to find L− . 2. Use the above formulae for L± to find the coordinate representation of the angular momentum basis states Y11 , Y10 and Y1,−1 up to normalization. 3. Write out the 9 equations summarized in the formula for products of Pauli matrices σi σ j = δi j + ii jk σk 4. Check that these formulae hold for the Pauli matrices ! ! 0 1 0 −i σx = , σy = , 1 0 i 0 (1) ! −1 0 σz = . 0 1 (2) 5. The hamiltonian for an electron in a vertical magnetic field Bẑ is ! g|e|~B 1 0 H=E , E= . 0 −1 4m Find the spin state ψ(t) if the initial spin wavefunction is ψ(t = 0) = 6. Compute hS z i, hS x i and hS y i at time t in the state ψ(t) 7. Physically interpret the obtained expectation values for S x , S y , S z . 1 (3) √1 2 (↑ + ↓).