* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Aerobic Metabolism: The Citric Acid Cycle
Multi-state modeling of biomolecules wikipedia , lookup
Butyric acid wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Mitochondrion wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Electron transport chain wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Photosynthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Biosynthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
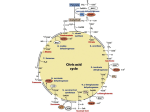
Khadijah Hanim Abdul Rahman School of Bioprocess Eng, UniMAP Sem 1, 2012/2013 Week 14: 10/12/2011 1. The citric acid cycle (also known as the tricarboxylic acid cycle, the TCA cycle, or the Krebs cycle) is a series of chemical reactions of central importance in all living cells that utilize oxygen as part of cellular respiration. 1. 2. In aerobic organisms, the citric acid cycle is part of a metabolic pathway involved in the chemical conversion of carbohydrates, fats and proteins into carbon dioxide and water to generate a form of usable energy. The citric acid cycle also provides precursors for many compounds such as certain amino acids, and some of its reactions are therefore important even in cells performing fermentation. Facultative anaerobes and obligate aerobes that use O2 to generate energy, employ the following biochemical processes: - TCA cycle - Electron transport pathway - oxidation phosphorylation In eukaryotes- occur in mitochondrion TCA cycle- metabolic pathway, 2 carbons fragments derived from organic fuel molecules are oxidized to form CO2 and the coenzymes NAD+ and FAD are reduced to form NADH and FADH2. The electron transport chain (ETC)mechanism in which electrons are transferred from reduced coenzymes to an acceptor, O2. In oxidative phosphorylation- energy released by ETC is captured in a form of a proton gradient that drives the synthesis of ATP, the energy currency of living organisms. In living organisms, both energycapturing and energy releasing processes consist primarily of redox reactions. In redox reactions electrons move between an electron donor and an electron acceptor. TCA cycle- series of biochemical reactions aerobic organisms use to release chemical energy stored in the 2carbon acetyl group in acetyl-CoA. Acetyl-CoA composed of an acetyl group derived from the breakdown of carbohydrates, lipids and some amino acid that is linked to the acyl carrier molecule Coenzyme A. Acetyl-CoA is synthesized from pyruvate Acetyl-CoA- also the product of fatty acid catabolism and certain reactions in amino acid metabolism. Its main function is to convey the carbon atoms within the acetyl group to the citric acid cycle to be oxidized for energy production. In TCA cycle- the carbon atoms are oxidized to CO2 and the high-energy electrons are transferred to NAD+ and FAD to form the reduced coenzymes NADH and FADH2. In the first reaction- a 2-carbon acetyl group condenses with a 4-carbon molecules (oxaloacetate) to form a 6-carbon molecule (citrate). During the subsequent 7 reactions, in which 2 CO2 molecules are produced and 4 pairs of electrons are removed from carbon compounds, citrate is reconverted to oxaloacetate. In one step in the cycle, substrate-level phosphorylation, high-energy molecule GTP is produced. The net reaction for TCA cycle as follows: Acetyl-CoA + 3NAD+ + FAD + GDP + Pi + 2H2O 2CO2 + 3NADH + FADH2 + CoASH + GTP + 3H+ In addition to its role in energy production, TCA cycle plays important role. Its cycle intermediates are substrates in variety of biosynthetic reactions. Pyruvate is transported out from cytosol into the mitochondrial matrix where it converted to acetyl-CoA Pyruvate acetyl-CoA- in a series of reactions catalyzed by pyruvate dehydrogenase complex enzyme The net reaction, an oxidative decarboxylation as follows: Pyruvate + NAD+ + CoASH Acetyl-CoA + NADH + CO2 + H2O + H+ The pyruvate dehydogenase enzyme complex contains 3 enzymes activities: - Pyruvate dehydrogenase - Dihydrolipoyl transacetylase - Dihydrolipoyl dehydrogenase TPP (Thiamine pyrophosphate), FAD, NAD+ and lipoic acid are the required co-enzymes for acetyl-CoA synthesis from pyruvate. The main function of acetyl-CoA is to convey the carbon atoms within the acetyl group to the citric acid cycle to be oxidized for energy production. in each turn of the cycle, acetyl-CoA from the glycolytic pathway/fatty acid catabolism enters and 2 fully oxidized carbon molecules leave as CO2. 3 molecules of NAD+ and 1 molecule of FAD are produced 1 molecule of GTP (interconvertible with ATP) is generated in a substrate-level phosphorylation reaction. TCA cycle is composed of eight reactions that occur in 2 stages: 1) The 2-carbon of acetyl group of acetylCoA enters the cycle by reacting with a 4-carbon compound oxaloacetate. 2 molecule of CO2 are subsequenly released. 2) Oxaloacetate is regenerated so it can react with another acetyl-CoA TCA cycle begins with the condensation of acetyl-CoA with oxaloacetate to form citrate: citrate which contains a tertiary alcohol is reversibly converted to isocitrate by an enzyme aconitase. During isomerization reaction, an intermediatecis-aconitate is formed by dehydration. The carbon-carbon double bond of cisaconitate is rehydrated to form isocitrate. isocitrate dehydrogenase catalyzes oxidative decarboxylation of isocitrate to form α-ketoglutarate: There are two different forms of isocitrate dehydrogenase, one requiring NAD+ as electron acceptor and the other requiring NADP+ . The overall reactions catalyzed by the two isozymes are otherwise identical. The NAD-dependent enzyme is found in the mitochondrial matrix and serves in the citric acid cycle to produce a-ketoglutarate. The NADP-dependent isozyme is found in both the mitochondrial matrix and the cytosol. It may function primarily in the generation of NADPH, which is essential for reductive anabolic reactions. The conversion of α-ketoglutarate to succinyl-CoA is catalyzed by the enzyme activities in the dehydrogenase complex: α-ketoglutarate dehydrogenase. The cleavage of the high-energy thioester bond of succinyl-CoA to form succinate, catalyzed by succinate thiokinase is coupled in mammals to the substrate-level phosphorylation of GDP. Succinate dehydrogenase catalyzes the oxidation of succinate to form fumarate: Succinate dehydrogenase is a flavoprotein using FAD to drive the oxidation of succinate to fumarate. Fumarate is converted to L-malate in a reversible stereospecific hydration reaction catalyzed by fumarase: Finally, oxaloacetate is regenerated with the oxidation of L-malate: The citric acid cycle begins with the condensation of acetyl-CoA molecule with oxaloacetate to form citrate, which is eventually reconverted to oxaloacetate. During this process, - 2 molecules of CO2 - 3 molecules of NADH - 1 molecule of FADH2 - 1 molecule of GTP Many of the enzymes in the TCA cycle are regulated by negative feedback from ATP when the energy charge of the cell is high. Such enzymes include the pyruvate dehydrogenase complex that synthesizes the acetyl-CoA needed for the first reaction of the TCA cycle. Also the enzymes citrate synthase, isocitrate dehydrogenase and alpha-ketoglutarate dehydrogenase, that regulate the first three steps of the TCA cycle, are inhibited by high concentrations of ATP. This regulation ensures that the TCA cycle will not oxidise excessive amounts of pyruvate and acetyl-CoA when ATP in the cell is plentiful. This type of negative regulation by ATP is by an allosteric mechanism Several enzymes are also negatively regulated when the level of reducing equivalents in a cell are high (high ratio of NADH/NAD+). This mechanism for regulation is due to substrate inhibition by NADH of the enzymes that use NAD+ as a substrate. This includes both the entry point enzymes pyruvate dehydrogenase and citrate synthase.