* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Addition of Alcohols to Form Hemiacetals and Acetals
Homoaromaticity wikipedia , lookup
Elias James Corey wikipedia , lookup
Kinetic resolution wikipedia , lookup
Metal carbonyl wikipedia , lookup
Stille reaction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Ene reaction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Aldol reaction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
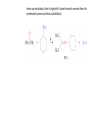
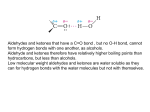
17-7 Addition of Alcohols to Form Hemiacetals and Acetals Aldehydes and ketones form hemiacetals reversibly. The reaction of alcohols with aldehydes and ketones parallels the reaction with water: These equilibria usually favor the starting carbonyl compound. Stable hemiacetals are formed from reactive carbonyl compounds (formaldehyde or 2,2,2-trichloroacetaldehyde) or when relatively strain-free, five- and sixmembered rings are formed: Acids catalyze acetal formation. In the presence of excess alcohol, two molecules of alcohol are added to an aldehyde or ketone in the acid-catalyzed reaction. The resulting compounds are called acetals (an older term for an acetal derived from a ketone is a ketal). Each step is reversible in the presence of acid. The equilibrium can be shifted towards acetal by using excess alcohol or removing water or towards aldehyde or ketone by adding excess water (acetal hydrolysis). Acetals, unlike hydrates and hemiacetals, may be isolated as pure substances by neutralizing the acid used as the catalyst. 17-8 Acetals as Protecting Groups Cyclic acetal formation protects carbonyl groups from attack by nucleophiles. 1,2-Ethanediol is a particularly effective reagent for forming acetals compared to ordinary alcohols because it forms a more stable cyclic intermediate. The stability of the cyclic acetal is due to a more favorable entropy change upon formation (2 reactant molecules become 2 product molecules) compared to a non-cyclic acetal (3 reactant molecules become 2 product molecules). Cyclic acetals can be hydrolized by aqueous acid, but are stable to many basic, organometallic and hydride reagents. These properties allow cyclic acetals to be used as protecting groups for the carbonyl function in aldehydes and ketones. The alkynyl anion attacks the carbonyl group in the unprotected 3-iodopropanal. Thiols react with the carbonyl group to form thioacetals. Thiols react with aldehydes and ketones in the presence of a Lewis acid, such as BF3 or ZnCl2, to form thioacetals. The cyclic thioacetal is stable in aqueous acid. Thioacetals are hydrolyzed by using mercuric chloride in aqueous acetonitrile (formation of insoluble mercuric sulfides drives the reaction). Thioacetals can be desulfurized to the corresponding hydrocarbon using Raney nickel: 17-9 Nucleophilic Addition of Ammonia and Its Derivatives Ammonia and primary amines add to aldehydes and ketones in a manner analogous to water and alcohols. However, the products of these reactions then lose water, forming imines and enamines. Ammonia and primary amines form imines. Amines and aldehydes or ketones react to form hemiaminals, the nitrogen analogs of hemiacetals. The hemiaminals of primary amines then lose water to form an imine (previously, Schiff base). This is the nitrogen analog of the carbonyl group. Reactions between a primary amine and an aldehyde or ketone, in which two molecules are joined with the elimination of water, are called “condensations.” When an imine is used as reaction intermediate, the carbonyl compound and amine are usually mixed with the subsequent reagent and the imine is consumed immediately upon formation. Imines can be isolated, often in high yield, if water formed is removed from the condensation process (continuous distillation). Special imines aid in the identification of aldehydes and ketones. Several amine derivatives condense with aldehydes and ketones to form imines that are highly crystalline and have sharp melting points. These derivatives are more stable than simple imines due to resonance stabilization. For instance, in an oxime: Comparison of the melting point of an unknown imine derivative to a catalog of known values was used as a method of identification of aldehydes and ketones prior to the development of spectroscopic methods.