* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Complete and Incomplete Metamorphosis
Survey
Document related concepts
Transcript
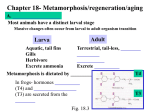
Complete and Incomplete Metamorphosis What is metamorphosis? • Metamorphosis refers to the way that certain organisms develop, grow, and change form. • Metamorphosis actually means "change". • Felt he was an outsider – Jewish in Catholic Prague – Sickly – Lonely • Perceived human beings as being trapped by authority in a hopeless world • Became frustrated at having to support his family • Had to work in a meaningless bureaucratic job where he was just another pencil pusher – Took time away from his writing Franz Kafka Two Types of Metamorphosis • COMPLETE METAMORPHOSIS has FOUR stages. • INCOMPLETE METAMORPHOSIS - has THREE stages (p.s. incomplete doesn’t mean not finished. It just means that the adult in complete metamorphosis is completely different from the larva. A nymph and its adult form are not completely different. They’re only a little different.) 4 STAGES OF COMPLETE METAMORPHOSIS • Egg • Larva • Pupa • Adult • EGG • The female lays eggs. • LARVA • Larva hatch from the eggs. They do not look like adult insects. They usually have a wormlike shape. • Caterpillars(毛虫), maggots (蛆虫), and grubs(蛴螬)are all just the larval stages of insects. Larvae molt their skin several times and they grow slightly larger. • ADULT • Inside the cocoon, the larvae change into adults. After a period of time, the adult breaks out of the cocoon. INCOMPLETE METAMORPHOSIS Let’s take a closer look at each stage! 3 STAGES OF INCOMPLETE METAMORPHOSIS • Egg • Nymph • Adult • EGG • A female insect lays eggs. These eggs are often covered by an egg case which protects the eggs and holds them together. • NYMPH • The eggs hatch into nymphs. • Nymphs looks like small adults, but usually don't have wings. • Insect nymphs eat the same food that the adult insect eats. • Nymphs shed or molt their exoskeletons (outer casings made up of a hard substance called chitin) and replace them with larger ones several times as they grow. • Most nymphs molt 4-8 times. • ADULT • The insects stop molting when they reach their adult size. By this time, they have also grown wings. ENDOCRINE- describing or relating to any gland or other group of cells that synthesizes hormones and secretes them directly into the blood, lymph, or other intercellular fluid Endocrine cells release protein and non-protein hormones Synthesis of hormones is orchestrated by the CNS Hormones effects are tissue dependent Hormonal Control of Insect Metamorphosis Temperature, Light, Stress, etc. Prothoracicotropic促前 胸腺hormone (PTTH) Brain Corpus Allatum咽侧体Prothoracic Gland前胸腺 Juvenile Hormone (JH)保 幼激素 Larva Pupa Ecdysteroid蜕皮 激素 Adult Control of Metamorphosis by Internal and External Factors Temperature, Light, Stress, etc. Brain Temperature (day degrees) Critical size matched (availability of food) Light (photoperiod) Chemicals Amount of moisture(湿度) Stress: mutagens, predators(肉食者), etc. Ecdysone: “Molting Hormone” Steroid hormone produced by prothoracic gland (lipid soluble, passes through cell membrane to the nucleus) Activates early response genes (TFs) and then late response genes (may cause differentiation,cell proliferation and migration, structural changes, apoptosis) Primes insect to respond to second hormone, EH Ec Early Response: Transcription Factors USP Binding Site Late Response: Transcription initiated by Transcription Factors Chromosome Puffing in Flies Observed in giant salivary gland chromosomes (no cell division after replication) Can be inhibited by actinomycin(放线菌素D) Puffing is where transcription is occurring. Ecdysone can be detected by fluorescent antibodies localized to the puffing Early puffs and late puffs seen in larva to pupa and pupa to adult molt Alternative Splicing of Ecdysone Receptor PremRNA Creates Several Forms of the EcReceptor Allowing Cell Type Specific Ecdysone Response Regulation of JH Levels Amount of Hormone Juvenile Hormone Ecdysone Larva Pupa Adult Low = larva stage; Medium JH levels = pupa stage; No JH = adult stage Rate of release limited by synthesis Amounts of JH also regulated by protein degradation and methyltransferase levels (can be protected by JH binding proteins, degraded by JH esterase) Metamorphosis in Action: Remember Imaginal Discs? Adult Insect: - JH Immature Insect: + JH Insect control by targeting metamorphosis Juvenile hormone mimic: Keep insects in larval stage -- Effective control for insects such as mosquitoes Juvenile hormone antagonist: Cause death of larva or early metamorphosis -- Effective control for crop pests such as hornworm Genes for juvenile hormone binding hormone and JH esterase have been identified Frogs: Tadpole to Adult Hormonal Control of Frog Metamorphosis Secretions of two hormones, thyroxine (T4) and triiodothyronine (T3) cause metamorphic changes Hormones have different effects depending on location in body Timing of changes regulated by tissue dependent hormone sensitivity Thyroid receptor is transcriptional repressor until thyroid hormone binds causing it to become a transcriptional activator Positive feedback loop is established between thyroid hormone and pituitary gland allowing incremental increases in hormone concentration Pituitary 垂体 Metamorphosis Thyroid Thyroid Hormones (T3 and T 4) Transcriptional Activation HIGH LOW Number of Receptors in Affected Tissue Amount of Hormone LOW HIGH T3 Early Response: Transcription Factors RXR Binding Site Late Response: Transcription initiated by Transcription Factors TH does not determine the developmental program, but initiates it • Changing the location of tissue or organ does not alter its response to TH • Transplant eye to tail region – Differentiates & grows into eye in response to TH while tail regresses • Transplant tail to trunk – Tail regresses while limb grows Tissue Repair Regeneration, repair & healing of injured tissues Regeneration Growth of cells and tissues to replace lost structures Requires an intact connective tissue scaffold Regeneration of vertebrates There are two types of regeneration: 1. Epimorphosis or epimorphic regeneration : This type of regeneration involve the reconstruction of the missing parts by local proliferation from the blastema, or addition of parts to remaining piece . For example: regeneration of tail, limbs and lens in anurans and urodels and other vertebrates. 2. Morpholaxis or morphollactic regeneration: This type of regeneration involving reorganization of the remaining part of the body of an animal.For example: Hydra, planaria and other invertebrates e.g. regeneration of the new individual from body pieces. Regeneration of Limb Regeneration begins in 3 phases : 1. Phase of wound healing or pre -blastema stage : Blood clotting and migration of epidermal cells from the basal layer of epidermis toward the centre of the wound. The wound is covered with epithelium which is thicker than the epidermis of the limb . 2. Phase of blastema formation : Cells accumulate beneath the epithelial covering and formed the blastema. Mesenchymal cells accumulate beneath the cap . Mesenchymal – blastemal cells differentiate into myoblasts and muscle cells, early cartilage cells and cartilage. During the dedifferentiate phase Hyaluronate (HA透 明质酸) increases in the distal stump to form blastema . As the blastema forms, the HA will be decrease. The production of HA and break down of collagen represent the establishment of migration from stump tissues . 3. Phase of dedifferentiate and morphogenesis : The blastema begins to restore the part of which the limb was deprived. Specifically, if the fore arm is removed, the blastema differentiated directly into the muscle, bone, cartilage and skin of the fore arm. Tissue response to injury. Repair after injury can occur by regeneration, which restores normal tissue, or by healing, which leads to scar formation and fibrosis. TYPES OF CELLS • Labile cells • Stable cells • Permanent cells Labile cells • have a high rate of loss and replacement and therefore high capacity for regeneration. • squamous and glandular epithelia • haemopoeitic cells in bone marrow Stable cells • do not normally proliferate but can be stimulated to do so after damage. • • • • • renal tubular cells, hepatocytes, osteoblasts, endothelial cells, fibroblasts. Permanent cells • Permanent cells : unable to divide after initial development and therefore cannot regenerate when some are lost. • Neurons • Skeletal & cardiac muscle Repair Involves • Regeneration of injured tissue by parechymal cells of the same type • Replacement by connective tissue (fibrosis), resulting in a scar • In most cases tissue repair involves both of these two processes. Repair by Connective Tissue (Fibrosis) • Fibrosis consists of four components – formation of new blood vessels (angiogenesis) – migration and proliferation of fibroblasts – deposition of ECM – maturation and reorganization of the fibrous tissue (remodeling) Biology of Aging Some age gracefully... Goldie Jean Studlendgehawn born on November 21, 1945 Animal Models in Aging Short lives, experimental results collected quickly, or over many generations; Maintained easily and inexpensively, present less complex genetic or physiological systems than humans. Caenorhabditis elegans Drosophila melanogaster Saccharomyces cerevisiae Genetically engineered animal models for exploring the basic mechanisms involved in the aging Mus musculus Theories of Aging: • • • • Oxidative Damage Telomeres Genetic Alterations with aging Mitochondrial Aging Other processes involved: • Inflammatory processes • Hormonal changes • Life style choices Oxidative stress: imbalance production/breakdown • Free radicals are normal products of metabolism • Predominant cellular free radicals are: - superoxide (O2 -) - hydroxyl (OH-) - nitrogen dioxide (NO2) superoxide - hydrogen peroxide (H2O2) Oxidative stress can lead to: • Damage to mitochondria, DNA, protein processing and cellular metabolism - lipid peroxidation - protein oxidation - DNA oxidation • This ultimately leads to: - Loss of cellular phenotype - Necrosis - or Apoptosis Endogenous defense mechanisms: O2 H2O Mitochondria P450 oxidases Catalase O2- SOD H2O2 OH+ H2O 2 GSH Aging↓ Glutathione Reductase Glutathione Peroxidase GSSG H2O Telomeres and Aging • Repetitive DNA sequences At the ends of all human Chromosomes From: Aditya Rana in Biotechnology • In humans there are 46 chromosomes; thus 92 telomeres (one per end) • Telomere is about 10 to 15 kb in length, composed of the tandem repeat sequence: TTAGGG • Without telomeres, ends of chromosomes would be “repaired”, leading to chromosome fusion and abnormal function • Telomeres regulate how many times a cell can divide. Telomere sequences shorten each time DNA replicates • Once telomeres shrink to a certain level, cells can no longer divide; hence aging From: National Institutes on Aging (not protected by copyright); official domain Summary of Telomere theory: Telomere length declines in dividing cells as we age Telomere length in bp (human blood cells) 8,000 3,000 1,500 0 35 Age in years 65 Genes that affect Aging: • • • • • Stress resistance genes Genes targeting inflammation Genes that slow basic metabolism, like IGF Overall genetic stability The problem: Altered gene expression resulting from quality control defects allows errors to accumulate as cells divide leads to cells with diminished function Many genes shown to influence life span are involved in DNA damage repair and protection. Progeria: • Two forms in humans; Werner's syndrome (adultonset progeria) and Hutchinson-Gilford syndrome (juvenile-onset progeria). • Most clinicians believe that progeria is segmental aging • Mutation in Werner’s codes for a DNA helicase (DNA repair/unwind) From: www.immortalhumans.com/982/ From:http://www.scripps.edu/~jjperry/research.html Progeria Werner’s syndrome: - chromosome instability syndromes -Inability to repair DNA -Increased rate of cancer -mutated helicase -inherited as autosomal recessive Hutchinson-Gilford: -no helicase abnormality -The pattern of inheritance is uncertain -have shorter than normal telomeres -undergo early cell senescence. Mitochondrial Aging Mitochondrial DNA is extra sensitive to damage, such as oxidative stress because it does not have repair mechanisms like normal DNA Summary of mitochondrial theory: • Decreased activity of electron transport chain complex with aging • Increased release of ROS • Alterations in mitochondrial apoptosis pathways (Bax/Bcl-2 etc) • Lack of repair mechanisms mtDNA • Slower mitochondrial turnover accumulates mtDNA mutations Genomic Alterations with Aging: Intact telomeric DNA Intact nuclear DNA Intact mtDNA Endogenous oxidative stress DNA repair Damaged/shorter telomeric DNA Cell cycle arrest senescence Damaged nuclear DNA Mutation and cancer Apoptosis Cell loss Aging Damaged mtDNA Diminished energy production