* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Week 03 Lecture notes
Biochemical cascade wikipedia , lookup
Magnesium in biology wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Electron transport chain wikipedia , lookup
Photosynthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
Biochemistry wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
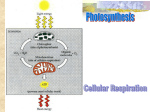
Learning Objectives • How do cells store energy? • How do cells use energy? • What types of reactions occur in cells? Which one requires energy as an input? Which one releases energy? • • • • • • What is the activation energy of a reaction? How do catalysts speed up reactions? How do enzymes work? What is a biochemical pathway? How does an enzyme’s environment affect its function? How does ATP provide energy for the cell? Learning Objectives • Consider the location of photosynthesis: Where does the light reaction occur? Where is sugar synthesized? • • • The flow of energy is the flow of electrons In very general terms, what does the electron transport system do? Compare the proton pump to previous membrane transport mechanisms we’ve looked at How does it relate to ATP synthesis? • • • Carbon dioxide + water + light = sugar + oxygen + water Both photosynthesis and cellular respiration use an electron transport chain Glycolysis Cytoplasm Anaerobic—Doesn’t require oxygen • What role does fermentation play in metabolism? Chemical Reactions • The starting molecules of a chemical reaction are called the reactants or substrates • The output molecules from the reaction are called products What are the substrates? What are the products? Where is the energy? Is the energy kinetic or potential? Endergonic vs. Exergonic There are two kinds of chemical reactions endergonic reactions have products with more energy than the reactants • these reactions require an input of energy exergonic reactions have products with less energy than the reactants • these reactions tend to occur spontaneously Is polysaccharide formation endergonic or exergonic? Exergonic Reactions Question: • If exergonic reactions tend to occur spontaneously, why haven’t they all done so? Activation Energy • All chemical reactions require an initial input of energy called activation energy the activation energy initiates a chemical reaction by destabilizing existing chemical bonds Catalysis Reactions become more likely to happen if their activation energy is lowered this process is called catalysis catalyzed reactions proceed much faster than noncatalyzed reactions . Enzymes Enzymes are the catalysts used by cells to perform particular reactions enzymes bind specifically to a molecule and stress the bonds to make the reaction more likely to proceed the active site is the site on the enzyme that binds to a reactant the site on the reactant where the enzyme binds is called the binding site Figure 5.5 An enzyme’s shape determines its activity Induced fit How Enzymes Work • The binding of a reactant to an enzyme causes the enzyme’s shape to change slightly this leads to an “induced fit” where the enzyme and substrate fit tightly together as a complex the enzyme lowers the activation energy for the reaction the enzyme is unaffected by the chemical reaction and can be re-used Essential Biological Process 5A: How Enzymes Work Animation: How Enzymes Work Please note that due to differing operating systems, some animations will not appear until the presentation is viewed in Presentation Mode (Slide Show view). You may see blank slides in the “Normal” or “Slide Sorter” views. All animations will appear after viewing in Presentation Mode and playing each animation. Most animations will require the latest version of the Flash Player, which is available at http://get.adobe.com/flashplayer. Animation: Enzyme Action and the Hydrolysis of Sucrose Please note that due to differing operating systems, some animations will not appear until the presentation is viewed in Presentation Mode (Slide Show view). You may see blank slides in the “Normal” or “Slide Sorter” views. All animations will appear after viewing in Presentation Mode and playing each animation. Most animations will require the latest version of the Flash Player, which is available at http://get.adobe.com/flashplayer. Inquiry & Analysis Do Enzymes Physically Attach to Their Substrates? Biochemical Pathways • Catalyzed reactions may occur together in sequence the product of one reaction is the substrate for the next reaction until a final product is made the series of reactions is called a biochemical pathway Figure 5.6 A biochemical pathway Animation: A Biochemical Pathway Please note that due to differing operating systems, some animations will not appear until the presentation is viewed in Presentation Mode (Slide Show view). You may see blank slides in the “Normal” or “Slide Sorter” views. All animations will appear after viewing in Presentation Mode and playing each animation. Most animations will require the latest version of the Flash Player, which is available at http://get.adobe.com/flashplayer. Factors Affecting Enzymes • Temperature and pH affect enzyme activity enzymes function within an optimum temperature range • when temperature increases, the shape of the enzyme changes due to denaturing of the protein chains enzymes function within an optimal pH range • the shape of enzymes is also affected by pH • most human enzymes work best within a pH range of 6 - 8 – exceptions are stomach enzymes that function in acidic ranges Figure 5.7 Enzymes are sensitive to their environment Why might a human enzyme need to function at low pH? Enzyme Regulation • Cells can control enzymes by altering their shape allosteric enzymes are affected by the binding of signal molecules • some signals act as repressors – inhibit the enzyme when bound • other signals act as activators – change the shape of the enzyme so that it can bind substrate Essential Biological Process 5B: Regulating Enzyme Activity Enzyme Inhibition Feedback inhibition is a form of enzyme inhibition where the product of a reaction acts as a repressor competitive inhibition • the inhibitor competes with the substrate for the active site • the inhibitor can block the active site so that it cannot bind the substrate non-competitive inhibition • the inhibitor binds to the allosteric site and changes the shape of the active site so that no substrate can bind Which type of feedback inhibition is more common? Why do you think that is the case? Figure 5.8 How enzymes can be inhibited Animation: Feedback Inhibition of Biochemical Pathways Please note that due to differing operating systems, some animations will not appear until the presentation is viewed in Presentation Mode (Slide Show view). You may see blank slides in the “Normal” or “Slide Sorter” views. All animations will appear after viewing in Presentation Mode and playing each animation. Most animations will require the latest version of the Flash Player, which is available at http://get.adobe.com/flashplayer. ATP: The Energy Currency of the Cell • The energy from the sun or from food sources must be converted to a form that cells can use adenosine triphosphate (ATP) is the energy currency of the cell Structure of ATP The structure of ATP suits it as an energy carrier each ATP molecule has three parts 1. 2. 3. a sugar an adenine nitrogenous base a chain of three phosphate groups the phosphates are negatively charged and it takes a lot of chemical energy to hold them together the phosphates are poised to come apart ATP Hydrolysis • When the endmost phosphate group is broken off an ATP molecule, energy is released ATP ADP + Pi + energy • The Pi represents inorganic phosphate ATP: Coupled Reactions • Coupled reactions usually endergonic reactions are coupled with the breakdown of ATP • more energy than is needed is released by the breakdown of ATP so heat is given off • ATP cycles in the cell with respect to its energy needs Figure 5.10 The ATP-ADP Cycle How ATP Is Made • • ATP powers many key cell activities Cells use two processes to convert energy from the sun and from food molecules into ATP photosynthesis • some cells convert energy from the sun into ATP and then use it to make sugars that store potential energy cellular respiration • cells break down the potential energy in sugars and convert it ATP Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. TABLE 5.1 How Cells Use ATP Energy to Power Cellular Work ATP Table 5.1 How Cells Use ATP Energy to Power Cellular Work P Product ATP ADP ATP Reactant Biosynthesis Contraction Chemical Activation Cells use the energy released from the exergonic hydrolysis of ATP to drive endergonic reactions like those of protein synthesis, an approach called energy coupling. In muscle cells, filaments of protein repeatedly slide past each other to achieve contraction of the cell. An input of ATP is required for the filaments to reset and slide again. Proteins can become activated when a highenergy phosphate from ATP attaches to the protein, activating it. Other types of molecules can also become phosphorylated by transfer of a phosphate from ATP. Outside of cell Sugar Outside of cell Na+ Vesicle or organelle Other associated proteins Coupled transport protein Ion pump Na+–K+ pump Motor protein Connector proteins ATP K+ ATP Inside of cell ATP Inside of cell Microtubule Importing Metabolites Active Transport: Na+– K+ Pump Cytoplasmic Tansport Metabolite molecules such as amino acids and sugars can be transported into cells against their concentration gradients by coupling the intake of the metabolite to the inward movement of an ion moving down its concentration gradient, this ion gradient being established using ATP. Most animal cells maintain a low internal concentration of Na+ relative to their surroundings, and a high internal concentration of K+. This is achieved using a protein called the sodium- potassium pump, which actively pumps Na+ out of the cell and in, using energy from ATP. Within a cell’s cytoplasm, vesicles or organelles can be dragged along microtubular tracks using molecular motor proteins, which are attached to the vesicle or organelle with connector proteins. The motor proteins use ATP to power their movement. Cell Macrophage cell Actin ATP Heat Flagellum H2O ATP + ATP H+ + Pi + ADP Flagellar Movements Cell Crawling Heat Production Microtubules within flegella slide past each other to produce flagellar movements. ATP powers the sliding of the microtubules. Actin filaments in a cell’s cytoskeleton continually assemble and disassemble to achieve changes in cell shape and to allow cells to crawl over substrates or engulf materials. The dynamic character of actin is controlled by molecules bound to actin filaments. The hydrolysis of the ATP molecule releases heat. Reactions that hydrolyze ATP often take place in mitochondria or in contracting muscle cells and may be coupled to other reactions. The heat generated by these actions can be used to maintain an organism’s temperature. An Overview of Photosynthesis All of the energy used by almost all living cells ultimately comes from the sun plants, algae, and some bacteria capture the sunlight energy by a process called photosynthesis only about 1% of the available energy in sunlight is captured What biological theme from Chapter 1 does this relate to? Overview of Photosynthesis What other biological theme could we relate this to? The Chloroplast Only the leaf cells of plants contain chloroplasts the chloroplast contains internal membranes called thylakoids the stroma is a semi-liquid substance that surrounds the thylakoids (like the cytoplasm of the chloroplast) Why are the thylakoids green? Photosystems-Pigments Embedded in the thylakoid membrane are photosystems that contain pigments the primary pigment molecule in most photosystems is chlorophyll the pigments act as an antenna to capture energy from sunlight Photosynthesis takes places in three stages 1. capturing energy from sunlight 2. using the captured energy to produce ATP 3. using the ATP to make carbohydrates from CO2 in the atmosphere Inside the chloroplast, but outside the thylakoid Photosynthesis: Role of Electrons Photosystems transfer light energy to electrons The flow of energy through the cell is the flow of electrons The energy from the electron is used to make ATP (Electron Transport System) The electron itself is donated to the formation of glucose Electrons are transferred from water to glucose Revisiting our biological theme from Ch.1: The flow of energy How Plants Capture Energy from Sunlight The main pigment in plants is chlorophyll Chlorophyll absorbs light at the end of the visible spectrum, mainly blue and red light Chlorophyll pigments and protein molecules occur as complexes within the thylakoid membrane Light energy is first captured by a chlorophyll pigment in a photosystem The energy is passed along to an electron, which subsequently passes through the electron transport system (ETS) Figure 6.2 How a photosystem works ETS Water Electron Transport System • The electron transport system (ETS) receives the excited electron from the electron acceptor the ETS is comprised of proteins that are embedded in the thylakoid membrane one of these proteins acts as a proton pump to move a proton from the stroma into the thylakoid space at the end of the ETS, the electron is used to generate O2 Electron Transport System Energized electrons are passed through the ETS in the thylakoid membrane • Its energy is used to power a “proton pump” that transports protons against their concentration gradient – Active transport How is this like the sodium-potassium pump? Animation: Proton Pump Please note that due to differing operating systems, some animations will not appear until the presentation is viewed in Presentation Mode (Slide Show view). You may see blank slides in the “Normal” or “Slide Sorter” views. All animations will appear after viewing in Presentation Mode and playing each animation. Most animations will require the latest version of the Flash Player, which is available at http://get.adobe.com/flashplayer. ATP synthase Protons diffuse across membrane (“down” their concentration gradient) through the enzyme ATP synthase • Passage of protons through ATP synthase turns it, catalyzing ATP synthesis. • ATP and electrons produced by ETS are used to make glucose in the stroma. • Energy from the sun is stored by photosynthesis https://www.youtube.com/watch?v=PjdPTY1wHdQ Building New Molecules • The synthesis of sugar from CO2 employs the Calvin cycle the products of the lightdependent reactions are used • ATP energy drives the cycle Where Is the Energy in Food? The energy for living is obtained by breaking down the organic molecules originally produced in plants the energy invested in building the organic molecules is retrieved by stripping away electrons and using them to make ATP this process is called cellular respiration The Food Reaction • Eukaryotes produce the majority of their ATP by harvesting electrons from the food molecule glucose: C6H12O6 + 6 O2 6 CO2 + 6 H2O + energy • The reactants are glucose and oxygen, and the products are carbon dioxide, water, and energy (heat or ATP) • In cellular respiration, oxygen is the final electron acceptor Cellular Respiration • The “opposite” of photosynthesis; high-energy electrons are harvested by breaking down sugars • Electrons pass through ETS in mitochondria Again, the energy from the electron is used to power proton pumps and generate a proton gradient across the membrane Flow of protons down the gradient through ATP synthase produces ATP This ATP provides the energy for the cell We’re still following the flow of energy through the cell Figure 7.3 An overview of cellular respiration Glycolysis 1st step in breakdown of glucose • Occurs in cytoplasm, not in mitochondria • Only produces 2 ATP • 3-carbon sugars produced by glycolysis are then used by mitochondria This is the only way organisms can derive energy from food in the absence of oxygen Acetyl-CoA • CoA is an important molecule in the Krebs cycle • As the Krebs cycle proceeds, CoA becomes acetyl-CoA If energy is needed, acetyl-CoA is used in production of ATP If energy is not needed, acetyl-CoA is used to produce energystoring fat. Krebs Cycle • Occurs in mitochondria • High-energy electrons are harvested from byproducts of glycolysis • The energy from the electrons is used by the ETS to produce lots of ATP (>30) Involves proton pumps and ATP synthase • Carbon dioxide is a byproduct How does this differ from photosynthesis? ETC-Powered Proton Pumps High-energy electrons are transferred to a series of proteins embedded in the mitochondrial membrane Called the electron transport chain Many of the proteins in the ETC operate as proton pumps, pumping protons into the intermembrane space of the mitochondrion The last transport protein donates the electrons to hydrogen and oxygen in order to form water Figure The electron transport chain Chemiosmosis ATP Mitochondria use chemiosmosis to make ATP as the concentration of protons builds up in the intermembrane space, the protons diffuse back across the membrane (down their concentration gradient) through ATP synthase channels their passage powers the production of ATP from ADP Fermentation Alternative to Krebs cycle that occurs in absence of oxygen Does not require mitochondria • bacteria carryout more than a dozen kinds of fermentation • eukaryotic cells are capable of only a few types of fermentation – Ethanol – Lactic acid Glucose Is Not the Only Food Molecule Cells also get energy from foods other than sugars proteins are first broken down into their individual amino acids • deamination removes amino groups so molecules can take part in the Krebs cycle fats are first broken down into fatty acids