* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Electron Transport Chain - mr-youssef-mci
Magnesium in biology wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Biochemical cascade wikipedia , lookup
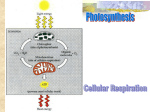
Photosynthesis wikipedia , lookup
Phosphorylation wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Mitochondrion wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Biochemistry wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Microbial metabolism wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Citric acid cycle wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Electron Transport Chain Mitochondrial Structure Electron Transport Chain Overview The ETC removes energy stored in the NADH and FADH2 molecules to: 1. create a proton gradient across the inner mitochondrial membrane 2. convert O2 to H2O. All reactions are redox reactions. Electron Transport Chain Animation ETC Animation ETC Components ETC Components: Complex I Complex 1: NADH Dehydrogenase 2 e- from NADH are transferred to Complex I Protons are pumped across the inner mitochondrial membrane (IMM) by Complex I (active Transport) ETC Components: Q e- are transferred from Complex I to ubiquinone (Q) Q is a mobile component within the IMM ETC Components: Complex III Complex III: Cytochrome b-c1 e- are transferred from Q to Complex III Protons are pumped across the IMM by Complex III ETC Components: Cyt C e- are transferred from Complex III to cytochrome c (cyt c) cyt c is a mobile component on the surface of IMM, in the intermembrane space ETC Components: Complex IV Complex IV: Cytochrome Oxidase e- are transferred from cyt c to Complex IV Protons are pumped across the IMM by Complex IV ETC Components: O2 O2 is the final electron acceptor of the ETC enough e- pass through the ETC to produce full H2O molecules FADH2 Pathway Oxidative phosphorylation. electron transport and chemiosmosis Glycolysis ATP ATP ATP H+ H+ H+ Intermembrane space Cyt c Protein complex of electron carners Q I III Inner mitochondrial membrane II FADH2 NADH+ Mitochondrial matrix IV FAD+ 2 H+ + 1/2 O2 NAD+ (Carrying electrons from, food) FADH2 Electron transport chain FAD Electron transport and pumping of protons (H+), which create an H+ gradient across the membrane Figure 9.15 H2O FADH2 Pathway 2e- are transferred from FADH2 to Complex II no protons are pumped across the IMM e- are transferred from Complex II to Q and proceed through the rest of ETC ETC Thermodynamics NADH FADH2 enters the chain 50 at a lower energy than NADH FADH2 Free energy (G) relative to O2 (kcl/mol) 40 I FMN Multiprotein complexes FAD Fe•S Fe•S II O III Cyt b 30 Fe•S 2 electrons from NADH Cyt c1 IV Cyt c Cyt a Cyt a3 20 produce a max of 3 ATP 2 electrons from FADH2 10 produce a max of 2 ATP 0 2 H + + 1 2 O2 H2O Electrochemical Proton Gradient ETC Summary 1. NADH e- transferred to O2; three proton pumps activated 2. FADH2 e- transferred to O2; two proton pumps activated 3. electrochemical proton gradient formed across IMM Electron Transport Chain Animation ETC Animation Proton Motive Force: Chemiosmosis The electrochemical gradient (chemiosmosis) produced by the ETC can now be used to generate ATP through the process of oxidative phosphorylation (OXPHOS). OXPHOS occurs through the enzyme complex ATP synthase. OXPHOS Animation ATP Synthase Complex Two components: 1. proton channel / rotor embedded in IMM 2. catalytic sites that phosphorylate ADP to ATP This is an example of facilitated diffusion (passive transport) ATP Production oxidative phosphorylation - ATP is produced as protons flow through ATP synthase. In general: 1. 1 NADH 2.5 – 3 ATP molecules 2. 1 FADH2 1.5 – 2 ATP molecules The ETC is coupled with ATP synthesis. The latter is dependent on the former. ATP Production Cellular Respiration Step Glycolysis Glycolysis Oxidative Oxidative Decarboxylation Decarboxylation Krebs Cycle Krebs Cycle Energy Molecules Produced 2 ATP 2 NADH 2 NADH 2 ATP special case 6 ATP 6 NADH 2 FADH2 2 ATP 18 ATP 4 ATP 2 ATP ATP Totals ATP Production– Aerobic Respiration Cellular Respiration Step Glycolysis Oxidative Decarboxylation Krebs Cycle TOTAL Energy Molecules Produced 2 ATP 2 NADH 2 NADH 6 NADH 2 FADH2 2 ATP ATP Totals 2 ATP 4-6 ATP 6 ATP 18 ATP 4 ATP 2 ATP 36-38 ATP Why 36-38 ATP? Electron shuttles span membrane CYTOSOL MITOCHONDRION 2 NADH or 2 FADH2 2 NADH 2 NADH Glycolysis Glucose 2 Pyruvate 2 Acetyl CoA + 2 ATP by substrate-level phosphorylation Maximum per glucose: Figure 9.16 6 NADH Citric acid cycle + 2 ATP 2 FADH2 Oxidative phosphorylation: electron transport and chemiosmosis + about 32 or 34 ATP by substrate-level by oxidative phosphorylation, depending on which shuttle transports electrons phosphorylation from NADH in cytosol About 36 or 38 ATP