* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Intracellular Respiration
Fatty acid metabolism wikipedia , lookup
Mitochondrion wikipedia , lookup
Metalloprotein wikipedia , lookup
Blood sugar level wikipedia , lookup
Phosphorylation wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Electron transport chain wikipedia , lookup
Microbial metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Biochemistry wikipedia , lookup
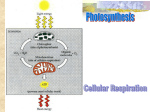
Intracellular Respiration Energy and matter necessary for metabolism are provided by Intracellular respiration C6H12O6 + 6O2 → 6CO2 + 6H2O +36ATP Photosynthesis 6CO2 + 6H2O Sunlight → C6H12O6 + 6O2 -Matter is always recycled, energy is not A. Carbohydrates, fats, and proteins can all be used as fuel 1. catabolism of glucose yields 686 kcal/mole of energy, some is used to produce ATP, some is lost as entropy B. ATP is synthesized with energy of electrons that are relocated from food to ATP through a series of redox reactions 1. oxidation – the loss of electrons (usually pulled by electronegative oxygen) a. in respiration Glucose is oxidized, releasing energy b. Oxygen, in turn, is reduced 2.reduction the addition of electrons 3. hydrocarbons, and molecules that have a lot of Hydrogen(sugars, fats) are sources of electrons that can be pulled by Oxygen 4. Glucose is broken down in a series of steps, each catalyzed by a specific enzyme a. hydrogen atoms are transferred from glucose by Dehydrogenase enzymes to coenzymes that are Hydrogen acceptors 1. NAD+ (oxidized) Nicotinamide adenine Dinucleotide, accepts electrons and becomes 2. NADH (reduced) 5. ATP is synthesized as the electrons carried by NADH lose their energy when transferred to oxygen during the ETC (electron transport chain) (mitochondria) a. at the end of ETC, Oxygen combines with the electrons and Hydrogen to form water. C. Respiration occurs in three stages 1.Glycolysis – 2.Krebs Cycle 3.ETC D. Glycolysis a. glucose is broken into 2 (3C) compounds, 2 NADH, and 2ATP b. occurs in the cytoplasm of both c. d. e. f. eukaryotic and prokaryotic cells. common to all life forms, this data to supports Evolution (common ancestor) Glucose 6C is oxidized and splits into two Pyruvates, which are 3C compounds. 10 steps in process, each with specific enzymes Energy Harvest Phase consumes 2 ATP 4 ATP are synthesized during the Energy Payoff Phase by substrate-level phosphorilation 1. enzymes: Phosphoglycerokinase and Pyruvate Kinase, transfer a phosphate to ADP to form ATP 2. two ATPs are gained in net g. h. 2 NADH molecules are made through the reduction of NAD+ 1. these go to the ETC, where each contributes enough energy to produce 3 ATPs Summary of molecules produced: 1. 2 (3C) pyruvates 2. 2 ATPs 3. 2 NADHs i. 10 steps of Glycolysis 1. These steps occur 2Xs. Once for each of the two G3Ps produced per glucose E. Krebs Cycle a. occurs in the mitochondrial matrix, where the specific Krebs cycle enzymes reside b. only occurs in Eukaryotic cells. c. Only occurs if Oxygen is present d. Pyruvate is oxidized to CO2 e. Cycle turns 2Xs per glucose f. Per cycle, one ATP is produced through substratelevel phosphorilation, 2 total ATP per glucose g. 3 NADHs produced per cycle, 6 in total per glucose h. one FADH2 (Flavin Adenine Dinucleotide) is produced per cycle, 2 per glucose i. prior to cycle, pyruvate enters the mitochondria (only if O2 is present) 1. pyruvate is oxidized to an acetyl CoA molecule a. a carboxyl group is removed as CO2 b. a pair of electrons and H are transferred to reduce NAD+ to NADH j. Cycle’s pathway elucidated by Hans Krebs in 1930’s 1. He named it the Citric Acid Cycle. It has 8 steps. a.cycle starts when acetyl combines with Oxaloacetate, a 4C cpd. to form citrate b. the acetate is ultimately degraded into 2 CO2 k. Summary of Krebs Cycle Cycle turns 2Xs/glucose (once per pyruvate) F. ETC The Electron Transport Chain a. in the cristae, the folded inner membrane of the mitochondria b. electrons from NADH & FADH2 move from molecule to molecule in a series of redox reactions (the electron transport chain), until they combine with O2 and H to form H2O c. as e- pass along the chain, their energy is harnessed and used to power the synthesis of ATP via oxidative phosphorilation d. NADH’s electrons are transferred to the first molecule in the ETC, a flavoprotein, FMN- flavin mononucleotide e. then the electrons pass through a series of redox reactions through Q-ubiquinone and the cytochromes f. the FADH2 electrons enter at a later point in the chain, into the Fe-S protein, then to Q, then to the cyt. g. electrons are passed by increasingly electronegative molecules in the chain until the are passed to oxygen, the most electronegative h. for every 2 electron carriers (4e-), one O2 is reduced to 2 H2O i. Proton Motive Force- a proton gradient, produced by the movement of electrons along the transport chain, as the exergonic flow of electrons pumps H+ into the intermembrane space. 1. this creates a proton-motive force 2. these H+ protons diffuse through ATP synthase. Their exergonic energy is used to power ATP synthesis j. ATP Synthase- a protein complex enzyme, on the cristae membrane of the mitochondria, that makes ATP from ADP + Pi k.Chemiosmosis = the process of coupling the redox reactions of electron transport to the synthesis of ATP. G. Summary of energy gain from aerobic respiration = 38 ATP 1. each NADH yields 3 ATPs 2. each FADH2 yields 2 ATPs 3. glycolysis: 2 ATP 2 NADH = 6 ATP Total = 8 ATP 4. Krebs Cycle per glucose 2 ATP 2 NADH = 6 ATP 6 NADH = 18 ATP 2 FADH2 = 4 ATP Total = 30 ATP H. Efficiency of Intracellular Respiration 1. the exergonic oxidation of glucose releases 686 kcal/mole of energy 2. the endergonic synthesis of ATP requires 7.3 kcal/mole of energy 3. the efficiency of respiration is 7.3 kcal/mole x 38 ATP/glucose/686kcal/mole of glucose x 100 = 40% 4. 60% of the energy is lost as heat I. Fermentation-to produce ATP w/o O2 1. prokaryotes must regenerate NAD+ AKA Anaerobic Respiration a. in glycolysis NAD+ is the oxidizing agent that pulls the e- from glucose to form: 2 pyruvates derivatives 2 NADH, and 2 ATPs. 2. also occurs in Eukaryotes in the absence of Oxygen (anaerobic) to generate 2 ATP through substrate level phosphorilation 3. 2 types of fermemtations: a. alcoholic: pyruvate is converted to: - ethanol CH3-CH2-OH - CO2 This pathway used in the production of wine, beer, and alcoholic beverages b. lactic acid: pyruvate is converted to: - lactate CH3-CHOH-COO This process used by muscles when oxygen concentration is low J. 38 ATP Aerobic Respiration Vs. 2 ATP Anaerobic from the oxidation of molecule of glucose K. Carbohydrates, fats, and proteins can all be catabolized