* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cell Metabolism Review
Nicotinamide adenine dinucleotide wikipedia , lookup
Electron transport chain wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
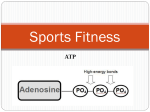
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Photosynthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
CELL & MOLEC BIOL HANDOUT REVIEW OF CELL METABOLISM The following summarizes a few facts to know from the chapter and class about metabolism. General Understand energy coupling and how it applies to specific rxns during metabolism. During catabolism (breakdown rxns of metabolism), energy is released in the form of ATP. Often energy is also released in the form of electrons (i.e. the organic molecule being broken down is oxidized). These electrons are carried by electron carriers such as NADH and FADH2 (Note: when not carrying electrons the molecules are in the form: NAD+ and FAD) ATP is a central energy transfer molecule in the cell. Glycolysis - is the breakdown of glucose (a 6 carbon monosaccharide) to 2 molecules of pyruvate (a 3 carbon compound) - uses 2 ATP’s to activate the intermediates, then releases 4 ATP’s for a net yield of 2 ATP’s - in general, under anaerobic conditions glucose is only partially broken down; a few additional reactions are required; this is known as fermentation - occurs in the cytosol of the cell Under aerobic conditions, the breakdown of glucose can continue to completion (i.e. completely oxidized to CO2) by Cellular Respiration. In cellular respiration, pyruvate enters the mitochondria where it is completely oxidized to CO2 by going through two more steps: Acetyl Coenzyme A (AcCoA) formation (also known as oxidative decarboxylation), then the Citric Acid Cycle (also known as Kreb’s Cycle). For each pyruvate, one CO2 is lost during AcCoA formation, the other 2 are lost during the Citric Acid Cycle. From one original molecule of glucose, two more ATPs are produced during the Citric Acid Cycle. Electrons removed during each of the three steps of glucose breakdown are carried by NADH and FADH2 to the Electron Transport Chain (ETC) where additional molecules of ATP are produced from the oxidation steps of the ETC (~34 additional ATPs per original molecule of glucose). Plants and some bacteria can harvest light energy and convert it to a form that is usable by other living organisms in a process known as Photosynthesis. In plants, photosynthesis occurs in chloroplasts. In the first half of photosynthesis, light energy is trapped and converted to chemical energy in the form of ATP and NADPH. In the second half of photosynthesis, the reactions of the Calvin Cycle use the energy stored in these two molecules to convert carbon dioxide to carbohydrate, which is a less temporary energy storage form. Notice the activation of the precursors for biosynthetic pathways: polysaccharides (Fig3.20), protein synthesis (Fig 3.23), nucleic acids (Fig 3.25). Be sure and go back and relate what we talked about in class to the more detailed description in the book. Although you do not need to know all of the details in the book, many concepts were interwoven in our class discussion of metabolism and will stick in your mind better by doing this comparison.