* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Electron Transport and oxidative phosphorylation (ATP Synthesis)
Survey
Document related concepts
Mitochondrion wikipedia , lookup
Photosynthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Metalloprotein wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Citric acid cycle wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Light-dependent reactions wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Transcript
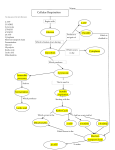
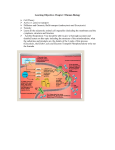
Electron Transport and oxidative phosphorylation (ATP Synthesis) Dr. Abir Alghanouchi Biochemistry department Sciences college All of the reactions involved in cellular respiration can be grouped into three main stages ` Glycolysis – occurs in cytoplasm ` The Krebs cycle – occurs in matrix of mitochondria ` Electron transport – occurs across the mitochondrial membrane 2 ` Process in which ATP is formed as a result of transfer of electrons from NADH or FADH2 by a series of electron carriers ` The electron transport chain generates no ATP directly. Rather, its function is to break the large free energy drop from food to oxygen into a series of smaller steps that release energy During respiration, most energy flows from glucose ‐> NADH ‐> electron transport chain ‐> proton‐motive force ‐> ATP. 3 Phosphorylation process Oxidative process O2 einner membrane H 2O H+ ATP Synthase ADP+ Pi H+ outer membrane ATP intermembrane space matrix Figure: Essential features of oxidative phosphorylation ¾ Redox reactions of respiratory chain use electrons to reduce oxygen to water ¾ Energy generated moves protons from matrix to intermembrane space ¾ Inward movement of protons recovers this energy to promote formation of ATP in the matrix. 4 Protein complex Electron carrier Inner mitochondrial membrane Electron flow Electron transport chain ATP synthase 5 ATP yield ` Only 4 of 38 ATP ultimately produced by respiration of glucose are derived from substrate‐level phosphorylation (2 from glycolysis and 2 from TCA) ` The vast majority of the ATP (90%) comes from the energy in the electrons carried by NADH and FADH2 6 High-energy electrons carried mainly byNADH High-energy electronscarried by NADH Cytosol Mitochondrion Glycolysis Glucose 2 Pyruvic acid 2 Acetyl‐ CoA Krebs Cycle Electron Transport Maximum per glucose: by direct synthesis by direct synthesis by ATP synthase A Road Map for Cellular Respiration 7 ` Chemical reactions that transfer electrons from one substance to another are called oxidation‐reduction reactions ` REDOX short for oxidation‐reduction reactions 8 REDOX FACTS ` A:H A Reductant ' Oxidant + e‐ ` B B:H Oxidant + e‐ ' Reductant (acceptor) (donor) ` ` Both oxidation and reduction must occur simultaneously The reductant of one pair donates electrons and the oxidant of the other pair accepts the electrons Red1 (AH) + Ox2 (B) Î Ox1(A) + Red2(BH) 9 ` Electrons can move through a chain of donors and acceptors ` In the electron transport chain, electrons flow down a gradient ` Electrons move from a carrier with low reduction potential (high tendency to donate electrons) toward carriers with higher reduction potential (high tendency to accept electrons) 10 ` Potential (EO): measure of the tendency of oxidant to gain electrons, to become reduced, a potential energy. ` ∆EO: Standard reduction potential difference between two half reactions 11 Succinate Eo' = 0.03V ∆Eo' = 0.07V I NADH Eo' = -0.32V II Coenzyme Q Eo' = 0.10V ∆Eo' = 0.42V III ∆Eo' = 0.19V Cytochrome C Eo' = 0.29V IV ½ O2 Eo' = 0.82V ∆Eo' = 0.53V electron flow ¾ The components of the RC are arranged in order of increasing redox potential ¾ The ∆Eo′ values are the potential differences across the four complexes ( that span the mitochondrial inner membrane) 12 Succinate Eo' = 0.03V ∆Eo' = 0.07V I NADH Eo' = -0.32V II Coenzyme Q Eo' = 0.10V ∆Eo' = 0.42V III ∆Eo' = 0.19V Cytochrome C Eo' = 0.29V IV ½ O2 Eo' = 0.82V ∆Eo' = 0.53V electron flow ¾ The overall voltage drop from NADH E0′ = ‐(‐0.32 V) to O E0′ = +0.82 V is ∆Eº′ = 1.14 V 13 RC exists as four large, multi‐ subunit protein complexes ` ` ` ` The respiratory electron transport chain complex I is a NADH‐ ubiquinone reductase complex II is succinate dehydrogenase complex III is the ubiquinone ‐ cytochrome c reductase complex IV is cytochrome oxidase 14 Figure: Complex I of the respiratory chain that links NADH and coenzyme Q. ` NADH Dehydrogenase (NADH‐ubiquinone reductase) accepts 2e‐ from NADH and transfers them to ubiquinone (coenzyme Q), an electron carrier ` Uses two bound cofactors to accomplish this: FMN (Flavin mononucleotide) and 6 iron‐sulfur (Fe‐S) protein 15 Complex II: Succinate-CoQ reductase Prosthetic groups: FAD; Fe-S Succinate FAD SDH Fumarate ` ` ` FADH2 CoQ SDH is succinate dehydrogenase an enzyme of the citric acid cycle (associated with membrane) 2 e‐ transferred from succinate to CoQ 1 mole FADH2 produced 16 Electrons from complex I or II CoQ cyt b/cyt c1 Complex III: cytochrome reductase Prosthetic groups: heme b; heme c1; Fe-S cyt c Figure: Complex III of the respiratory chain linking CoQ and cytochrome C. ` Is composed of cytochome b, cytochrome C1 and iron sulphur proteins ` Accepts e‐ from coenzyme Q and transfers e‐ to cytochrome c coupled with the transfer of protons from the matrix to the intermembrane space 17 Figure: Complex IV ‐cytochrome oxidase‐ reducing oxygen to water ` Contains cytochromes a/a3 and 2 Cu ions involved in e‐ transfers ` Cytochrome oxidase passes electrons from cytochrome c through a series of heme groups and Cu ions to O2, reducing it to H2O (end product) 18 Coenzymes and cytochromes in the complexes act as e‐ donors & acceptors 19 ` Flavin MonoNucleotide (FMN), in Complex I, functions like FAD (which is an electron acceptor that helps electron transfer during Krebs Cycle and Electron Transport Chain in cellular respiration). ` iron‐sulfur (Fe‐S proteins): Fe‐S centers transfer e‐ in Complexes I, II and III ` Coenzyme Q (ubiquinone), lipid soluble, floats in the membrane and doesn’t require protein ` Cytochromes (b, c1, c, a, a3; contain heme): transfer e‐ in Complexes III and IV, Cytc is the only soluble cytochrome ` NAD+, FMN, CQ are carriers of e‐ and hydrogen while cytochromes are carriers of electrons only. 20 ` ATP‐synthase (complex V), present in the inner mitochondrial membrane, actually makes ATP from ADP and Pi. ` ATP used the energy of an existing proton gradient to power ATP synthesis. ◦ This proton gradient develops between the intermembrane space and the matrix. ◦ This concentration of H+ is the proton‐motive force. 21 ¾ The ATP synthase molecules are the only place that will allow H+ to diffuse back to the matrix ¾ This flow of H+ is used by the enzyme to generate ATP a process called chemiosmosis. ¾ Chemiosmosis: (osmos = push) is the oxidative phosphorylation that results in ATP production in the inner membrane of mitochondria. 22 Properties of ATP Synthase ` Multisubunit transmembrane protein ` Molecular mass = ~450 kDa ` Functional units ◦ F0: water‐insoluble transmembrane protein (up to 8 different subunits) ◦ F1: water‐soluble peripheral membrane protein (5 subunits) ,contains the catalytic site for ATP synthesis Flow of 3 protons through ATP synthase leads to phosphorylation of 1 ADP 23 ` Cytosolic NADH (glycolysis) must enter the mitochondria to fuel oxidative phosphorylation but NADH and NAD+ cannot diffuse across the inner mitochondrial membrane ` Two shuttle systems for reducing equivalents: 1. Glycerol phosphate shuttle: insect flight muscles 2. Malate Malate‐aspartate shuttle: predominant in liver and other mammalian tissues 24 25 ` In muscle and brain ` Each NADH converted to FADH2 inside mitochondrion ◦ FADH2 enters later in the electron transport chain ◦ Produces 2 ATP 26 27 ` In liver and heart ` NADH oxidized while reducing oxaloacetate to malate ◦ Malate dehydrogenase ` Malate crosses membrane 28 ` Malate reoxidized to oxaloacetate ◦ Malate dehydrogenase ◦ NAD+ reduced to NADH ` NADH via electron transport yields 3 ATP 29 Respiratory inhibitors ¾ These compounds prevent the passage of e‐ by binding a component of the ETC blocking the oxidation/reduction reaction 30 31 Complex designation Functional groups Function I– ----------NADH-Q reductase FMN (flavin ----------mono‐ nucleotide); Fe‐S oxidizes NADH to NAD+; Rotenone ----------------------transfers electrons to coenzyme Q II – SuccinateQ reductase ----------- FAD; Fe‐S oxidizes succinate to fumarate with reduction of FAD to --------------FADH2; electron transfer to CoQ III Cytochrome ----------reductase heme b; heme c 1; ---------Fe‐S transfers electrons between coenzyme Q -------------and cytochrome C (C becomes reduced) Antimycin A ---------- IV ----------C Cytochrome oxidase heme a‐a3; ---------Cu oxidizes cytochrome C; ------------reduces ½O 2 to H2O Carbon monoxide ---------Cyanide ---------- Inhibitors 32 ` Lippincot's Illustrated Reviews Biochemistry ` Lechinger's Principles of Biochemistry 4th edition. D. L. Nelson and M.M. Cox, Worth Publishers. ` Harpers illustrated biochemistry 25th edition. Robert K. Murray; Darly K. Granner: Victor W Rodwell ` www.rpi.edu/dept/bcbp/molbiochem ` www.med.ufl.edu/biochem/rcohen/rcohen.html ` http://courses.cm.utexas.edu/jrobertus/ch339k 33 34