* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download breakdown of living things
Organ-on-a-chip wikipedia , lookup
Cell culture wikipedia , lookup
Cell encapsulation wikipedia , lookup
Tissue engineering wikipedia , lookup
Extracellular matrix wikipedia , lookup
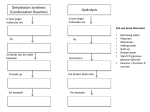
Cellular differentiation wikipedia , lookup
Signal transduction wikipedia , lookup
Endomembrane system wikipedia , lookup
UNIT 1 STRUCTURE AND FUNCTION HOW THINGS ARE BUILT AND HOW THEY WORK At every level of organization structure is arranged based on function. HOW IS LIFE ORGANIZED? 1. BIOSPHERE Zone of life, the layer of Earth’s surface where life is found 2. Biome Region with similar climate and plant type Example: desert 3. ECOSYSTEM All the organisms which share the same habitat: The community plus the abiotic factors that they use. Biotic factors - Abiotic factors 4. Community The combination of all the populations in an ecosystem 5. Population All the individuals of one species in a community 6. Organism An individual living thing 7. System A set of interacting organs working together to accomplish a common task 8. Organ A structure made up of tissues that performs a task within a body system 9. Tissue A group of cells that together carry out a specific function 10. Cell The unit of structure and function for all living things 11. Molecule 2 or more atoms bonded together. Carbohydrates, Proteins, Fats, DNA, ATP 12. Atom Building blocks of molecules C, H, O, N, P, S UNIT 1-1 MOLECULES PROTEINS Structural proteins Make up the structure of our cells. (make up us, we are made of cells) YOUR SKIN YOUR MUSCLES PROTEINS Enzymes Proteins that catalyze the reactions going on in our cells (bodies) NUCLEIC ACIDS (DNA) The instructions for building our bodies. In the nucleus of each cell. Tells the cells which proteins to make. CARBOHYDRATES Sugars and Starch Energy source for our cells (our bodies) LIPIDS Long term storage of energy. LIPIDS Cell membranes ATP The molecule that holds the energy from our food until our cells (bodies) need it. REVIEW: 5 ORGANIC MOLECULES Make up cell membranes? Enzymes? The main source of energy in our diets? Holds the energy from our food until we need it? Makes up the structure of our cells? Long term storage of energy? Fiber? Controls the chemical reactions in our bodies? GENERAL CHEMISTRY ATOM The smallest particle of matter that still has the properties of that element. The building blocks of molecules. MOLECULE Two or more atoms COVALENTLY bonded together. (Sharing pair electrons) ELEMENT Pure substance. Contains only one type of element. Oxygen = O2, Nitrogen = N2 Hydrogen = H2 COMPOUND Two or more elements bonded together H2O CO2 ATOM Oxygen O=8 FILLING THE OUTER SHELL H - Hydrogen N - Nitrogen CARBON – MOST IMPORTANT NEEDS 4 MORE ELECTRONS IONIC BONDS Ion = atom gains or loses an electron, giving it a + or – charge. IONIC BONDS Opposites attract Oppositely charged ions stick together IONS FORM CRYSTALS Salt = sodium chloride COVALENT BOND Make molecules 2 atoms share a pair of electrons to fill their outer shells. HYDROGEN BONDS POLAR MOLECULES Electrons are shared Unequally, causing a positive and negative end (pole) like a magnet. HYDROGEN BONDS Polar molecules stick to each other like weak magnets. Why is water a liquid at room temp? Why does it stick to things? Why is it a solvent? DNA HYDROGEN BONDS 3 BONDS - REVIEW Makes How molecules ? ? Makes crystals ? How ? Holds water together ? Why ? CHEMICAL FORMULAS Glucose: C6H12O6 CHEMICAL REACTIONS CO2 + H2O C6H12O6 + O2 6CO2 + 6H2O C6H12O6 + 6O2 pH Scale Measures acids and bases P. 86 IN BIOLOGY BOOK WATER SPLITS INTO IONS MORE H+ IONS = ACID MORE OH- IONS = BASE IF H+ AND OH- ARE THE SAME IT IS NEUTRAL (7) PROTEINS 2 jobs (there are more) Make up structure of cells (our bodies) Enzymes control the chemical reactions in our bodies BUILDING PROTEINS Polypeptide = long chain of amino acids DNA tells the cell the order of the amino acids AMINO ACIDS THE BUILDING BLOCKS OF PROTEINS ENZYMES Catalysts – control chemical reactions in our bodies Specific – they can only do one specific reaction They can be used over and over Changes in temperature or pH will make them lose their shape and not work. CARBOHYDRATES Monosaccharide (glucose) = single sugar Disaccharide (sucrose) = double sugar POLYSACCHARIDES Chain of many sugars Starch = made by plants to store excess glucose, we do have the enzymes to digest it Cellulose (fiber) = makes up cell walls in plant cells. We do NOT have enzymes to digest it. Glycogen (animal starch) = made by animals (us) to store excess glucose until we need it LIPIDS - FATS Long term storage of energy Make up cell membranes Nonpolar = will not mix with or stick to water FATTY ACIDS Building blocks of lipids BODY FAT Triglycerides CELL MEMBRANE Phospholipid bilayer = 2 layers of . . . . ATP The molecule that holds the energy from our food (glucose) until we need it. ATP We eat carbohydrates (starch) and digest them down to simple sugars (glucose). Our blood then brings the glucose (sugar) to our cells for energy. Our cells break down the glucose to release its energy. That energy is then stored in ATP until our bodies (cells) need it.