* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Protein Degradation As discussed in last the last lecture, newly
Survey
Document related concepts
Homology modeling wikipedia , lookup
Degradomics wikipedia , lookup
Circular dichroism wikipedia , lookup
Protein domain wikipedia , lookup
Bimolecular fluorescence complementation wikipedia , lookup
Protein folding wikipedia , lookup
Protein moonlighting wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein structure prediction wikipedia , lookup
Protein purification wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Western blot wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Transcript
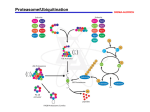
Protein Degradation As discussed in last the last lecture, newly synthesized proteins are folded and glycosylated in the ER in preparation for their transport to other organelles. However, life is not always perfect in the ER: proteins may turn out to have mutations that prevent proper folding, or poisonous compounds may inhibit glycosylation. Similarly, some of the proteins in the cytoplasm may also be misfolded, or damaged by oxygen radicals or other compounds, and aggregate. If all this junk is allowed to accumulate, the cell would die. Therefore, the cell rapidly breaks such protein down. Hence a need for a proteolysis pathway. In addition to removal of misfolded proteins, proteolysis is also important in recycling or turnover of proteins and amino acids, as well as regulation of processes such as cell cycle, transcription, cell signaling, and apoptosis. Proteolysis takes place primarily in the cytoplasm. So why don’t “normal” cytosolic proteins get chewed up? The first reason is that protein degradation occurs in defined compartments that are excluded from the cytosol. Secondly, proteins that are destined to be degraded are often marked. We’ll look at the organelles or machines involved in protein degradation, and then see how ER proteins, which are excluded from the cytosol, get degraded. I. Lysosomes. The lysosome is a membrane bound organelle that contains enzymes involved in the degradation of a variety of substances. ( MBOC p. 610 Fig. 13-17) Lysosomes are the main organelle for the degradation of extracellular materials, which are collected by endocytosis, which will be discussed in detail in later lectures. Lysosomes also digest entire organelles or parts of the cytoplasm that are engulfed in a process known as autophagy. ( MBOC p. 614 Fig. 13-22) There is yet a third, more specific process. During stress situations such as starvation, there is a large turnover of proteins in the cell. Studies have shown that RNase A, and other cytosolic proteins bearing the amino acids sequence KFERQ, are directly and selectively transported to the lysosome for degradation (V&V p. 1011). About 40-50 acid hydrolases reside in the lysosome. In addition to proteases, there are nucleases, glycosylases, lipases, phospholipases etc. They are acid hydrolases because their optimal pH for activity is about 5, which is the pH inside lysosomes. The cell is smart in that if a lysosome accidentally ruptures and releases its contents to the cytosol, the cell won’t be chewed up, because the enzymes will not be active. If you isolate lysosomes, and put them in a test tube with solution that has ADP and Pi, ATP is made and the pH of the solution goes up. What does this tell you? As you could guess by now, the internal pH is maintained by a proton pump that derives its energy through ATP hydrolysis. This pump is formally known as the vesicular ATPase, or VATPase. A variety of proteases exists inside the lysosome (V&V p. 1011). These include carboxypeptidase, aminopeptidase, and endopeptidase. Most of them belong to the class known as cathepsins. Many of these are cysteine proteases. Like the serine proteases, they also have a catalytic triad, this time consisting of Cys-His-Asn. II. The Ubiquitin-Proteasome Pathway This is the major pathway by which cytosolic proteins are selectively degraded during normal metabolic growth. As mentioned, it is also very important for regulation of processes such as cell cycle control and cell signaling. A. Ubiquitination The first step is the attachment of a signal/flag to the protein that is to be degraded. In this case the signal is a polyubiquitin chain. Ubiquitin is a 72 amino acids polypeptide that is very abundant, as its name suggests. Linkage of ubiquitin to the “condemned” protein occurs in 3 enzyme-catalyzed steps (see V&V p. 1012 Fig. 30-59). 1. E1- ubiquitin – a thioester bond is formed between the C-terminal amino acids of ubiquitin and an internal Cys residue of E1, the ubiquitin activating enzyme. This is an ATP dependent process. 2. E2- ubiquitin – an exchange reaction occurs, and the ubiquitin is transferred to a sulfhydryl group on E2, the ubiquitin-conjugating enzyme. The thioester bond is preserved and no ATP is required in this reaction. 3. E2-ubiquitin then interacts with E3, to which the substrate is specifically bound. E3 is known as the ubiquitin-protein ligase. It transfers the activated ubiquitin to the ε-NH2 group on a lysine residue in the protein substrate, thus establishing an amide linkage. In subsequent reactions, additional ubiquitin molecules are added to an ε-NH2 group on a lysine residue of the previously conjugated ubiquitin molecule to form a multiubiquitin chain linked by isopeptide bonds. B. The specificity of the system is conferred primarily by E3. To date there are 4 main classes that have been identified (refer to the handout on protein degradation). 1. N-end rule E3s. The residue at the N-terminus of proteins affect their half-lives (see V&V p. 1014 and MBOC p. 220). E3α is one of the E3s that have been identified. It has two distinct sites that recognize either basic (type1) or bulky-hydrophobic (type 2) residues. It also recognizes other non-N-end rule substrates via its “body site”. 2. HECT-domain E3s. (homologous to the E6-AP C-terminus). E6-AP is a cellular protein that forms a complex with the human papillomavirus protein E6 (thus AP=associated protein). Together they act like an E3 to target p53, a tumor suppressor protein in the cell, for degradation. The conserved HECT domain is in the C-terminus of these E3s, and has catalytic function, and an invariable Cys residue. The N-terminus of HECT-domain E3s is not highly conserved. Rsp5 is an example of a HECT-domain E3. It contains a calcium-binding domain, C2, and 3 WW domains in the amino-terminal two-thirds of the protein. The WW domains are in part responsible for substrate recognition. 3. Cyclosome/APC (anaphase promoting complex). Cyclosomes are inactive during interphase. They are activated by a mitotic kinase. When phosphorylated, they specifically recognize cell-cycle regulators such as mitotic cyclins that contain a 9 amino acids motif termed the destruction box. While N-end rule and HECT-domain E3s are single polypeptides, the cyclosome has several subunits. Cells that have mutations in any of these subunits fail to degrade B-type cyclins, cannot complete or exit from mitosis. 4. Phosphoprotein-ubiquitin ligase complexes. These complexes also degrade cell cycle regulators. However, their substrates need to be phosphorylated before these E3s will recognize them. While certain subunits in these complexes are invariant, others can be swapped to alter the substrate specificity. C. Proteasome Structure (see MBOC p. 219 Fig. 5-37): The core of the proteasome is a 20S complex composed of 4 stacked rings. There are 2βrings on the inside and 2 α rings on the outside. Each ring is composed of 7 subunits. A central channel is present in the 20S complex. This arrangement is reminiscent of GroEL. The opening at the top created by the alpha subunits is about 13 Å and is just big enough for an unfolded chain to enter. The 19S Cap complex normally sits on top of the alpha ring, rather like GroES. The 19S cap consists of more than 15 subunits, including ATPases. The functions of the 19S complex includes recognition of condemned proteins via the ubiquitin chain (another way to prevent the accidental degradation of cytoplasmic proteins), release and subsequent hydrolysis of the ubiquitin chain, unfolding of the condemned polypeptide, and translocation of the unfolded protein into 20S. It may also control the exit of degradation products. The step(s) that require energy has not been clearly demonstrated, though it is highly likely that both unfolding of the protein and its translocation are coupled to ATP hydrolysis. Even though the α and βsubunits are closely related in sequence, only the βsubunits have protease activity. Chymotrypsin-like, trypsin-like protease activity, and an activity that cleaves after glutamate residues have been identified. As the polypeptide is fed through the 20S complex, it gets degraded by the βsubunits, and peptides of 7-10 amino acids are released from the proteasome. Thus, the architecture of the proteasome machine segregates the proteolytic component from the cytoplasm, and also prevents cytosolic proteins from accidentally falling in and getting degraded. An inhibitor of the proteasome has been identified. It is called lactacystin. Studies using this inhibitor showed that most cytosolic proteins are degraded by the proteasome. III. What about ER proteins? So far we’ve seen that proteolysis in the cell takes place in the cytoplasm, albeit in structurally confined spaces. Are there proteases in the ER that degrade misfolded proteins? If so, how are they separated from resident and newly synthesized proteins? Are there proteasomes in the ER? These large structures have never been observed in the ER. Studies of human cytomegalovirus (HCMV) suggest that proteins that aren’t properly folded, or not glycosylated correctly are secreted out of the ER by retrograde transport. When most viruses infect a cell, the cell responds by putting some of the foreign protein on a flagpole on the surface of the cell to tell T cells to come kill it. These flagpoles are known as major histocompatibility (MHC) antigens. MHC class I heavy chains bind to peptides of about 7-9 amino acids (yes, you guessed it, produced by the proteasome) and its partner β2-microglobulin. This complex is transported to the surface of the cell. In HCMV infected cells, the heavy chains are co-translationally transported into the ER and glycosylated, as in normal uninfected cells, but then are quickly deglycosylated, transported back to the cytoplasm and degraded. This degradation is inhibited by lactacystin, suggesting the proteasome is involved. Furthermore, researchers have been able to detect an association between Sec61p and the heavy chain. Since proteasomes have also been observed to be closely associated with ER membranes, one model is that retrograde transport of errant polypeptides from the ER is tightly coupled to degradation by the proteasome. How the ER distinguishes a protein that is in the process of folding from those that are not able to fold, and how these errant proteins are delivered to the translocation machinery to be exported back to the cytoplasm is not known. IV. The Unfolded Protein Response What we do know is an interesting phenomenon known as the unfolded protein response (UPR). When a lot of “junk” proteins accumulate in the cell, for example by treating cells with reducing agents, or by preventing glycosylation, the ER sends out a signal that upregulates resident ER proteins. These include BiP, protein disulfide isomerase, and peptidyl-prolyl isomerase, which as you learnt last time, help proper folding of proteins. The mechanism by which this response is mediated is very interesting. All the genes that are upregulated in the UPR have a cis-acting element in their control region that is bound by the transcription factor HAC1 (refer to the handout). During normal growth, HAC1 is produced but quickly degraded by the ubiquitin/proteasome pathway. However, during UPR, the mRNA of HAC1 is spliced and produces a stable protein that can then upregulate the response genes. It turns out that an ER transmembrane protein, Ire1p, somehow senses the abundance of junk in the ER. The cytosolic side of this protein is partly a serine/threonine kinase, and partly an endoribonuclease. Ire1p is responsible for cleaving the HAC1 mRNA, which is then ligated by RLG1, a tRNA ligase.