* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download complex patterns of inheritance
Gene therapy of the human retina wikipedia , lookup
Essential gene wikipedia , lookup
Gene nomenclature wikipedia , lookup
Gene therapy wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Genetic engineering wikipedia , lookup
Oncogenomics wikipedia , lookup
Y chromosome wikipedia , lookup
Point mutation wikipedia , lookup
Gene desert wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Ridge (biology) wikipedia , lookup
Genome evolution wikipedia , lookup
Transgenerational epigenetic inheritance wikipedia , lookup
Gene expression programming wikipedia , lookup
Minimal genome wikipedia , lookup
Dominance (genetics) wikipedia , lookup
Skewed X-inactivation wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Biology and consumer behaviour wikipedia , lookup
History of genetic engineering wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Gene expression profiling wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Quantitative trait locus wikipedia , lookup
Genome (book) wikipedia , lookup
Epigenetics of human development wikipedia , lookup
Designer baby wikipedia , lookup
Genomic imprinting wikipedia , lookup
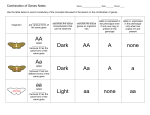
96 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication 17. Complex Patterns of Inheritance © The McGraw−Hill Companies, 2008 COMPLEX PATTERNS OF I NHERITANCE CHAPTER OUTLINE 17.1 17.2 Gene Interactions 17.3 17.4 Extranuclear Inheritance: Organelle Genomes Genes on the Same Chromosome: Linkage, Recombination, and Mapping X Inactivation, Genomic Imprinting, and Maternal Effect Snail shells (Lymnaea peregra) that coil to the right or left. The direction of coiling of a snail’s shell is an example of a complex inheritance pattern. n Chapter 16, we examined inheritance patterns in which the outcome of a single trait was governed by a single gene. In the cases we considered, the alleles segregated and assorted independently, allowing us to predict the phenotypes of offspring from the genotypes of their parents. These phenotypes occurred in definite ratios and they did not overlap—a pea plant was either tall or dwarf; a blood type was either A, B, or O. The inheritance patterns of most traits are more complex, however, and in this chapter we will examine some of the factors that complicate the prediction of phenotypes. In the first section of the chapter, we will consider how two or more different genes may affect the outcome of a single trait. For example, we examine continuously varying traits like human skin color, and you will see how the interaction of multiple genes and environmental influences can produce such a continuum. In the rest of the chapter we will consider inheritance patterns that defy Mendel’s laws of inheritance. First we discuss genes that are linked on the same chromosome and therefore do not assort independently. Next we consider the genes found in chloroplasts and mitochondria, which defy the law of segregation. Don’t worry, Mendel’s laws do describe most inheritance patterns, and they accurately reflect the behavior of chromosomes during meiosis. However, as you will learn in this chapter, they simply don’t apply to all of the genes that eukaryotic organisms possess. We will end the chapter with a discussion of three inheritance patterns, X inactivation, genomic imprinting, and maternal effect, that were not easily explained until researchers began to unravel genetic events that occur at the cellular and molecular levels. As you will learn, males and females don’t always regulate their genes in the same way, and this can lead to seemingly bizarre patterns of inheritance that are distinct from X-linked and sex-influenced inheritance patterns, I which we considered in the previous chapter. An exciting advance over the past few decades has been a better understanding of such unusual patterns of inheritance. Studies of complex inheritance patterns such as those described in this chapter have helped us appreciate more fully how genes influence phenotypes. These studies have revealed an astounding variety in the ways that inheritance occurs. The picture that emerges is of a wonderful web of diverse mechanisms by which genes give rise to phenotypes. Table 17.1 provides a summary of the most common patterns of inheritance. 17.1 Gene Interactions The study of single genes was pivotal in establishing the science of genetics. This focus allowed Mendel to formulate the basic laws of inheritance for traits with a simple dominant/ recessive inheritance pattern. Likewise, this approach helped later researchers understand inheritance patterns involving incomplete dominance and codominance, as well as traits that are influenced by an individual’s sex. In reality, however, all or nearly all traits are influenced by many genes. For example, in both plants and animals, height is affected by genes that encode proteins involved in the production of growth hormones, cell division, the uptake of nutrients, metabolism, and many other functions. A defect in any of these genes is likely to have a negative impact on an individual’s height. If height is controlled by many genes, you may be wondering how Mendel was able to study the effects of a single gene that produced tall or dwarf pea plants. The answer lies in the genotypes of his strains. Although many genes affect the height of pea plants, Mendel chose true-breeding strains that differed Brooker−Widmaier−Graham−Stiling: Biology 350 III. Nucleic Acid Structure and DNA Replication 97 © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance UNIT III – CHAPTER 17 Table 17.1 Different Types of Inheritance Patterns Type Description Mendelian Inheritance patterns in which a single gene affects a single trait, and the alleles segregate and assort independently. These patterns include simple dominant/recessive traits, X-linked traits controlled by a single gene, incomplete dominance, codominance, and sex-influenced traits (refer back to Table 16.1). Epistasis A type of gene interaction in which the alleles of one gene mask the effects of a dominant allele of another gene. Continuous variation Inheritance pattern in which the offspring display a continuous range of phenotypes. This pattern is produced by the additive interactions of several genes, together with environmental influences. Linkage Inheritance patterns involving two or more genes that are close together on the same chromosome. These genes do not assort independently. Extranuclear inheritance Transmission pattern of genes found in the DNA of mitochondria or chloroplasts, which are inherited independently of genes in the nucleus and do not segregate during meiosis. Usually these genes are inherited from the mother. X inactivation Phenomenon of female mammals in which one X chromosome is inactivated in every somatic cell, producing a mosaic phenotype. Most genes on the inactivated X chromosome are not expressed. Genomic imprinting Inheritance pattern in which an allele from one parent is inactivated in the somatic cells of the offspring, while the allele from the other parent is expressed. Maternal effect Inheritance pattern in which the genotype of the mother determines the phenotype of the offspring. This occurs because maternal effect genes of the mother provide gene products to developing egg cells. or more alleles. This phenomenon is called a gene interaction. As you will see, allelic variation at two or more loci may affect the outcome of traits in different ways. First we will look at interactions in which an allele of one gene prevents the expression of an allele of a different gene. Then we will discuss interactions in which multiple genes have additive effects on a single trait. These additive effects, together with environmental influences, account for the continuous phenotypic variation that we see for most traits. An Epistatic Gene Interaction Occurs When the Allele of One Gene Masks the Phenotypic Effects of a Different Gene In some gene interactions, the alleles of one gene mask the expression of the alleles of another gene. This phenomenon is called epistasis (Greek ephistanai, stopping). An example is the unexpected gene interaction discovered by William Bateson and Reginald Punnett in the early 1900s, when they were studying crosses involving the sweet pea, Lathyrus odoratus. A cross between a true-breeding purple-flowered plant and a truebreeding white-flowered plant produced an F1 generation with all purple-flowered plants and an F2 generation with a 3:1 ratio of purple- to white-flowered plants. Of course, Mendel’s laws predicted this result. The surprise came when the researchers crossed two different varieties of white-flowered sweet peas (Figure 17.1). All of the F1 generation plants had purple flowers! When these plants were allowed to self-fertilize, the F2 generation had purple-flowered and white-flowered plants in a 9:7 ratio. From these results, Bateson and Punnett deduced that two different genes were involved. To have purple flowers, a plant must have one or two dominant alleles for each of these genes. The relationships among the alleles are as follows: C (one allele for purple) is dominant to c (white) P (an allele for purple of a different gene) is dominant to p (white) with regard to only one of these genes. As a hypothetical example, let’s suppose that pea plants have 10 genes affecting height, which we will call K, L, M, N, O, P, Q, R, S, and T. The genotypes of two hypothetical strains of pea plants may be: Tall strain: KK LL MM NN OO PP QQ RR SS TT Dwarf strain: KK LL MM NN OO PP QQ RR SS tt In this example, the tall and dwarf strains differ at only a single locus. One strain is TT and the other is tt, and this accounts for the difference in their height. If we make crosses of tall and dwarf plants, the genotypes of the F2 offspring will differ with regard to only one gene; the other nine genes will be identical in all of them. This approach allows a researcher to study the effects of a single gene even though many genes may affect a single trait. In this section, we will examine situations in which a single trait is controlled by two or more genes, each of which has two cc masks P, or pp masks C, in either case producing white flowers A plant that was homozygous for either c or p would have white flowers even if it had a purple-producing allele at the other locus. Epistatic interactions often arise because two or more different proteins are involved in a single cellular function. For example, two or more proteins may be part of an enzymatic pathway leading to the formation of a single product. This is the case for the formation of a purple pigment in the sweet pea strains we have been discussing: Enzyme C Colorless precursor Enzyme P Colorless intermediate Purple pigment In this example, a colorless precursor molecule must be acted on by two different enzymes to produce the purple pig- 98 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication 17. Complex Patterns of Inheritance © The McGraw−Hill Companies, 2008 COMPLEX PATTERNS OF INHERITANCE 351 or enzyme P. When either of these enzymes is missing, the plant cannot make the purple pigment and has white flowers. P White variety #1 CCpp White variety #2 ccPP F1 All purple CcPp Self-fertilization F2 CP Cp cP cp CP CCPP Purple CCPp Purple CcPP Purple CcPp Purple Cp CCPp Purple CCpp White CcPp Purple Ccpp White cP CcPP Purple CcPp Purple ccPP White ccPp White cp CcPp Purple Ccpp White ccPp White ccpp White Figure 17.1 Epistasis in the sweet pea. The color of the sweet pea flower is controlled by two genes, each with a dominant and a recessive allele. Each of the dominant alleles (C and P) encodes an enzyme required for the synthesis of purple pigment. A plant that is homozygous recessive for either gene (cc or pp) cannot synthesize the pigment and will have white flowers. Biological inquiry: In a Ccpp individual, which functional enzyme is missing? Is it the enzyme encoded by the C or P gene? ment. Gene C encodes a functional protein, enzyme C, that converts the colorless precursor into a colorless intermediate. The recessive c allele results in a lack of production of enzyme C in the homozygote. Gene P encodes the functional enzyme P, which converts the colorless intermediate into the purple pigment. Like the c allele, the p allele results in an inability to produce a functional protein. A plant that is homozygous for either of the recessive alleles will not make any functional enzyme C Polygenic Inheritance and Environmental Influences Produce Continuous Phenotypic Variation As we have just seen, an epistatic interaction causes the alleles of one gene to mask the effects of a different gene. Let’s now turn to another way that the alleles of different genes may affect the phenotype of a single trait. In many cases, the effects of alleles may be additive. This has been observed for many traits, particularly those that are quantitative in nature. Until now we have discussed the inheritance of traits with clearly defined phenotypic variants, such as red or white eyes in fruit flies. These are known as discrete traits, or discontinuous traits, because the phenotypes do not overlap. For most traits, however, the phenotypes cannot be sorted into discrete categories. The majority of traits in all organisms are continuous traits, also called quantitative traits, which show continuous variation over a range of phenotypes. In humans, quantitative traits include height, weight, skin color, metabolic rate, and heart size, to mention a few. In the case of domestic animals and plant crops, many of the traits that people consider desirable are quantitative in nature, such as the number of eggs a chicken lays, the amount of milk a cow produces, and the number of apples on an apple tree. Consequently, much of our modern understanding of quantitative traits comes from agricultural research. Quantitative traits are polygenic, which means that several or many genes contribute to the outcome of the trait. For many polygenic traits, genes contribute to the phenotype in an additive way. Another important factor is the environment. As we saw in Chapter 16, the environment plays a vital role in the phenotypic expression of genes. Environmental factors often have a major impact on quantitative traits. For example, an animal’s diet affects its weight, and the amount of rain and sunlight that fall on an apple tree affect how many apples it produces. Because quantitative traits are polygenic and greatly influenced by environmental conditions, the phenotypes among different individuals may vary substantially in any given population. As an example, let’s consider skin pigmentation in people. This trait is influenced by several genes that tend to interact in an additive way. As a simplified example, let’s consider a population in which this trait is controlled by three genes, which we will designate A, B, and C. Each gene has a dark allele, designated AD, BD, or CD, and a light allele, designated AL, BL, or CL, respectively. All of the alleles encode enzymes that cause the synthesis of skin pigment, but the enzymes encoded by dark alleles cause more pigment synthesis than the enzymes encoded by light alleles. Figure 17.2 considers a hypothetical case in which people who were heterozygous for all three genes produced a large population of offspring. The bar graph shows the genotypes of the offspring, grouped according to the total number of dark alleles. As shown by the shading of the figure, skin pigmentation increases as the number of dark alleles increases. Brooker−Widmaier−Graham−Stiling: Biology 352 III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 99 UNIT III – CHAPTER 17 ADA LB DB LC DC L ADALB DB LC DC L Fraction of people with same phenotype 20/ 15 64 /64 ADALB DB LC DC L 10/ 5 Same population raised in a sunnier environment ADADB LB LC LC L ADADB DB LC LC L ALALB DB DC DC D ALALB DB DC LC L ADADB LB LC DC L ADADB LB LC DC D ALALB LB LC DC D ADALB DB DC LC L ADADB DB DC LC L ADALB LB LC LC L ADALB DB LC LC L ADALB LB LC DC D ALADB LB DC DC D ALADB DB DC DC D ALALB DB LC LC L ADALB LB LC DC L ALALB DB LC DC D ALADB DB DC LC D ADADB LB DC DC D 64 /64 0 Number of light alleles Number of dark alleles ALALB LB LC DC L ALALB DB LC DC L ALALB DB DC DC L ADADB LB DC LC D ADADB DB DC LC D ADADB DB DC DC D 6 5 4 3 2 1 0 0 1 2 3 4 5 6 ALALB LB LC LC L Light Dark Pigmentation Figure 17.2 Continuous variation in a polygenic trait. Skin color is a polygenic trait that can display a continuum of phenotypes. The bell curve on the left (solid line) shows the range of skin pigmentation in a hypothetical human population. The bar graphs below the curve show the additive effects of three genes that affect pigment production in this population; each bar shows the fraction of people with a particular number of dark alleles (AD, BD, and CD ) and light alleles (AL, BL, and CL). The bell curve on the right (dashed line) represents the expected range of phenotypes if the same population was raised in a sunnier environment. Offspring who have no dark alleles or no light alleles—that is, who are homozygous for all three genes—are fewer in number than those with some combination of dark and light alleles. As seen in the bell-shaped curve above the bar graph, the phenotypes of the offspring fall along a continuum. This continuous phenotypic variation, which is typical of quantitative traits, is produced by genotypic differences together with environmental effects. A second bell-shaped curve (the dashed line) depicts the expected phenotypic range if the same population of offspring had been raised in a sunnier environment, which increases pigment production. These two curves illustrate how the environment can also have a significant influence on the range of phenotypes. In our discussion of genetics, we tend to focus on discrete traits because this makes it easier to relate a specific genotype with a phenotype. This is usually not possible for continuous traits. For example, as depicted in the middle bar of Figure 17.2, seven different genotypes can produce individuals with a medium amount of pigmentation. Nevertheless, it is important to emphasize that the majority of traits in all organisms are continuous, not discrete. Most traits are influenced by multiple genes, and the environment has an important impact on the phenotypic outcome. 17.2 Genes on the Same Chromosome: Linkage, Recombination, and Mapping In all of the inheritance patterns we have studied so far, the alleles segregate and assort independently as predicted by Mendel’s laws. As we have seen, phenotypes can be influenced by a variety of factors, including gene interactions and environmental effects, that make it difficult to relate genotype to phenotype. Even so, if we understand all of these factors and take them into account, we can see that each of the genes is transmitted according to Mendel’s laws. In the rest of this chapter, we will consider inheritance patterns in which the outcome of a cross violates one of Mendel’s laws. In this section, we focus on transmission patterns that do not conform to the law of independent assortment. We will begin by examining the first experimental cross that demonstrated this pattern. You will learn that this pattern was explained by Thomas Hunt Morgan, who proposed that genes located close to each other on the same chromosome tend to be inherited as a group. Finally, we will see how crossing over between such genes provided the first method of mapping genes on chromosomes. 100 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication 17. Complex Patterns of Inheritance © The McGraw−Hill Companies, 2008 COMPLEX PATTERNS OF INHERITANCE Bateson and Punnett’s Crosses of Sweet Peas Showed That Genes Do Not Always Assort Independently In Chapter 16, we learned that the independent assortment of alleles is due to the random alignment of homologous chromosomes during meiosis (refer back to Figure 16.11). But what happens when the alleles of different genes are on the same chromosome? A typical chromosome contains many hundreds or even a few thousand different genes. When two genes are close together on the same chromosome, they tend to be transmitted as a unit, a phenomenon known as linkage. A group of genes that usu- Figure 17.3 ally stay together during meiosis is called a linkage group, and the genes in the group are said to be linked. In a two-factor cross, linked genes do not follow the law of independent assortment. The first study showing linkage between two different genes was a cross of sweet peas carried out by William Bateson and Reginald Punnett in 1905. A surprising result occurred when they conducted a cross involving two different traits, flower color and pollen shape (Figure 17.3). One of the parent plants had purple flowers (PP) and long pollen (LL); the other had red flowers (pp) and round pollen (ll). As Bateson and Punnett expected, the F1 plants all had purple flowers and long pollen (PpLl). The unexpected result came in the F2 generation. A cross of sweet peas showing that independent assortment does not always occur. HYPOTHESIS The alleles of different genes assort independently of each other. STARTING MATERIALS True-breeding sweet pea strains that differ with regard to flower color and pollen shape. 1 Experimental level Conceptual level PPLL ppll Cross a plant with purple flowers and long pollen to a plant with red flowers and round pollen. Purple flowers, long pollen 2 Red flowers, round pollen Observe the phenotypes of the F1 offspring. PpLl Purple flowers, long pollen 3 Meiosis Allow the F1 offspring to self-fertilize. PL and pl gametes — more frequent Pl and pL gametes — less frequent Purple flowers, long pollen 4 353 Purple flowers, long pollen Fertilization Observe the phenotypes of the F2 offspring. Purple flowers, Purple flowers, Red flowers, Red flowers, long pollen round pollen long pollen round pollen 15.6 : 1.0 : 1.4 : 4.5 F2 offspring having phenotypes of purple flowers, long pollen or red flowers, round pollen occurred more frequently than expected from Mendel’s law of independent assortment. Brooker−Widmaier−Graham−Stiling: Biology 354 5 III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 101 UNIT III – CHAPTER 17 THE DATA Phenotypes of F2 offspring Purple flowers, long pollen Purple flowers, round pollen Red flowers, long pollen Red flowers, round pollen Observed number Observed ratio Expected number Expected ratio 296 19 27 85 15.6 1.0 1.4 4.5 240 80 80 27 9 3 3 1 Although the offspring displayed the four phenotypes predicted by Mendel’s laws, the observed numbers of offspring did not conform to the predicted 9:3:3:1 ratio. Rather, as seen in the data in Figure 17.3, the F2 generation had a much higher proportion of the two phenotypes found in the parental generation: purple flowers with long pollen, and red flowers with round pollen. These results did not support the law of independent assortment. Bateson and Punnett suggested that the transmission of flower color and pollen shape was somehow coupled, so that these traits did not always assort independently. Although the law of independent assortment applies to many other genes, in this example, the hypothesis of independent assortment was rejected. Linkage and Crossing Over Produce Parental and Recombinant Phenotypes The chromosomes next to the flies in Figure 17.4 show the arrangement of these alleles. If the two genes are on the same chromosome, we know the arrangement of alleles in the P generation flies because these flies are homozygous for both genes (bbcc or bbcc). In the P generation female on the left, b and c are linked, while b and c are linked in the male on the right. Let’s now look at the outcome of the crosses in Figure 17.4. As expected, the F1 offspring (bbcc) all had gray bodies and straight wings, confirming that these are the dominant traits. In the next cross, F1 females were mated to males that were homozygous for both recessive alleles (bbcc). A cross in which an individual with a dominant phenotype is mated with a homozygous recessive individual is called a testcross, as described in Chapter 16. In the crosses we are discussing here, the purpose of the testcross is to determine whether the genes for body color and wing shape are linked. If the genes were on different chromosomes and assorted independently, this testcross should have produced equal numbers of F2 offspring with the four possible phenotypes. The observed numbers, shown above the F2 phenotypes, clearly conflict with this prediction based on independent assortment. The two most abundant phenotypes are those with the combinations of characteristics in the P generation: gray bodies and straight wings or black bodies and curved wings. These offspring are called nonrecombinants because their combination of traits has not changed from the parental generation. They are also termed parental types. The smaller number of offspring that have a different combination of traits—gray bodies and curved wings or black bodies and straight wings—are recombinants or nonparental types. How do we explain the occurrence of recombinants when genes are linked on the same chromosome? As shown beside the flies of the F2 generation in Figure 17.4, each recombinant individual has a chromosome that is the product of a crossover. The crossover occurred while the F1 female fly was making egg cells. Although Bateson and Punnett realized their results did not conform to Mendel’s law of independent assortment, they did not provide a clear explanation for their data. A few years later, Thomas Hunt Morgan obtained similar ratios in crosses of fruit flies while studying the transmission pattern of genes located on the X chromosome. Like Bateson and Punnett, Morgan observed many more F2 offspring with the parental combination of traits than would be predicted on the basis of independent assortment. To explain his data, Morgan proposed these ideas: 1. When different genes are located on the same chromosome, the traits that are determined by those genes are most likely to be inherited together. 2. Due to crossing over during meiosis, homologous chromosomes can exchange pieces of chromosomes and create new combinations of alleles (refer back to Figure 15.17). 3. The likelihood of crossing over depends on the distance between two genes. Crossovers between homologous chromosomes are much more likely to occur between two genes that are farther apart in the chromosome compared to two genes that are closer together. To illustrate the first two of these ideas, Figure 17.4 considers a series of crosses involving two genes that are linked on the same chromosome in Drosophila. The P generation cross is between flies that are homozygous for alleles that affect body color and wing shape. The female is homozygous for the wild-type alleles that produce gray body color (bb) and straight wings (cc); the male is homozygous for mutant alleles that produce black body color (bb) and curved wings (cc). Note that the symbols for the genes are based on the name of the mutant allele; the wild-type allele is indicated by a superscript plus sign (+). 102 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 355 COMPLEX PATTERNS OF INHERITANCE P Gray body, straight wings Black body, curved wings b b c c b b c c Homozygous recessive bbcc Homozygous dominant bbcc F1 Gray body, straight wings Black body, curved wings Testcross b b c c b b c c bbcc bbcc F2 Gray body, straight wings Black body, curved wings Gray body, curved wings Black body, straight wings Total Number observed 371 359 133 137 1,000 Number expected based on independent assortment 250 250 250 250 1,000 b b b b c c c c bbcc b b c c bbcc bbcc Nonrecombinants b b c c bbcc Recombinants Figure 17.4 Linkage and recombination of alleles. An experimenter crossed bbcc+ and bbcc flies to produce F1 heterozygotes. F1 females were then testcrossed to bbcc males. The large number of parental phenotypes in the F2 generation suggests that the two genes are linked on the same chromosome. F2 recombinant phenotypes occur because the alleles can be rearranged by crossing over. As shown below, four different egg cells are possible: b c Crossover b b b b c c c c Homologs in F1 female b c Meiosis b b c c Nonrecombinant chromosomes Recombinant chromosomes Due to crossing over, two of the four egg cells produced by meiosis have recombinant chromosomes. What happens when eggs containing such chromosomes are fertilized in the testcross? Each of the male fly’s sperm cells carries a chromosome with the two recessive alleles. If the egg contains the recombinant chromosome carrying the b and c alleles, the testcross will produce an F2 offspring with a gray body and curved wings. If the egg contains the recombinant chromosome carrying the b and c alleles, F2 offspring will have a black body and straight wings. Therefore, crossing over in the F1 female can explain the occurrence of both types of F2 recombinant offspring. Brooker−Widmaier−Graham−Stiling: Biology 356 III. Nucleic Acid Structure and DNA Replication 103 © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance UNIT III – CHAPTER 17 Morgan’s ideas about linkage and crossing over were based on similar data, derived from his studies of genes on the X chromosome. The idea that linked genes tend to be inherited together explained the high frequency of parental combinations of traits in certain crosses. The suggestion that crossing over produces chromosomes with new allele combinations accounted for the occurrence of recombinant phenotypes. Morgan’s third idea regarding linkage was that the frequency of crossing over between linked genes depends on the distance between them. This suggested a method for determining the relative positions of genes on a chromosome, as we will see next. Recombination Frequencies Provide a Method for Mapping Genes Along Chromosomes The oldest approach to studying the arrangement of genes in a species’ genome is called genetic linkage mapping (also known as gene mapping or chromosome mapping). This experimental method is used to determine the linear order of genes that are linked to each other along the same chromosome. As depicted in Figure 17.5, this linear arrangement is shown in a chart known as a genetic linkage map. Each gene has its own unique locus at a particular site within a chromosome. For example, the gene for black body color (b) that we discussed earlier is located near the middle of the chromosome, while the gene for curved wings (c) is closer to one end. The first genetic linkage map, showing five genes on the Drosophila X chromosome, was constructed in 1911 by Alfred Sturtevant, an undergraduate who spent time in Morgan’s laboratory. Genetic linkage mapping allows us to estimate the relative distances between linked genes based on the likelihood that a crossover will occur between them. This likelihood is proportional to the distance between the genes, as Morgan first proposed. If the genes are very close together, a crossover is unlikely to begin in the region between them. However, if the genes are very far apart, a crossover is more likely to be initiated between them and thereby recombine their alleles. Therefore, in a cross involving two genes on the same chromosome, the percentage of recombinant offspring is correlated with the distance between the genes. This correlation provides the experimental basis for gene mapping. If a two-factor testcross produces many recombinant offspring, the experimenter concludes that the genes are far apart. If very few recombinant offspring are observed, the genes must be close together. To find the distance between two genes, the experimenter must determine the frequency of crossing over between them, called their recombination frequency. This is accomplished by conducting a testcross. As an example, let’s refer back to the Drosophila testcross described in Figure 17.4. As we discussed, the genes for body color and wing shape are on the same chromosome; the recombinant offspring are the result of crossing Mutant phenotype Wild-type phenotype Aristaless, al Long aristae 13.0 Dumpy wings, dp Long wings 48.5 Black body, b Gray body 54.5 Purple eyes, pr Red eyes 67.0 Vestigial wings, vg Long wings 75.5 Curved wings, c Straight wings 104.5 Brown eyes, bw Red eyes Map units 0.0 Figure 17.5 A simplified genetic linkage map. This map shows the relative locations of a few genes along a chromosome in Drosophila melanogaster. The name of each gene is based on the mutant phenotype. The numbers on the left are map units (mu). The distance between two genes, in map units, corresponds to their recombination frequency in testcrosses. over during egg formation in the F1 female. We can use the data from the testcross shown in Figure 17.4 to estimate the distance between these two genes. The map distance between two linked genes is defined as the number of recombinant offspring divided by the total number of offspring times 100. Map distance Number of recombinant offspring Total number of offspring 133 137 371 359 133 137 100 100 27.0 map units The units of distance are called map units (mu), or sometimes centiMorgans (cM) in honor of Thomas Hunt Morgan. One map unit is equivalent to a 1% recombination frequency. In this example, 270 out of 1,000 offspring are recombinants, so the recombination frequency is 27% and the two genes are 27.0 mu apart. Genetic linkage mapping has been useful for analyzing the genes of organisms that are easily crossed and produce many offspring in a short time. It has been used to map the genes of several plant species and of certain species of animals, such as 104 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication 17. Complex Patterns of Inheritance © The McGraw−Hill Companies, 2008 COMPLEX PATTERNS OF INHERITANCE Drosophila. However, for most organisms, including humans, linkage mapping is impractical due to long generation times or the inability to carry out experimental crosses. Fortunately, many alternative methods of gene mapping have been developed in the past few decades that are faster and do not depend on crosses. These newer cytological and molecular approaches, which we will discuss in Chapter 20, are now used to map genes in a wide variety of organisms. 17.3 Extranuclear Inheritance: Organelle Genomes In the previous section, we examined the inheritance patterns of linked genes that violate the law of independent assortment. In this section, we will explore inheritance patterns that violate the law of segregation. Gene transmission may defy this law because some genes are not found on the chromosomes in the cell nucleus. The segregation of genes is explained by the pairing and segregation of homologous chromosomes during meiosis; genes found elsewhere in the cell do not segregate in the same way. The transmission of genes that are located outside the cell nucleus is called extranuclear inheritance. Two important types of extranuclear inheritance patterns involve genes that are found in mitochondria and chloroplasts (Figure 17.6). Extranuclear inheritance is also called cytoplasmic inheritance because these organelles are in the cytoplasm of the cell. As we discussed in Chapter 6, mitochondria and chloroplasts are found in eukaryotic cells because of an ancient endosymbiotic relationship. They contain their own genetic material, or genomes. Although these organelle genomes are much smaller than nuclear genomes, researchers have discovered that they are critically important in the phenotypes of organisms. In plants, for example, the chloroplast genome carries many genes that are vital for photosynthesis. Mitochondrial genes are critical for respiration. In humans, mutations in the mitochondrial genome may cause inherited diseases. In this section, we will examine the transmission patterns observed for genes found in the chloroplast and mitochondrial genomes and consider how mutations in these genes may affect an individual’s traits. Chloroplast Genomes Are Often Maternally Inherited One of the first experiments showing an extranuclear inheritance pattern was carried out by Carl Correns in 1909. Correns discovered that leaf pigmentation in the four-o’clock plant (Mirabilis jalapa) follows a pattern of inheritance that does not obey Mendel’s law of segregation. Four-o’clock leaves may be green, white, or variegated, as shown in Figure 17.7. Correns observed that the pigmentation of the offspring depended solely on the pigmentation of the maternal parent, a phenomenon called maternal inheritance. If the female parent had white leaves, 357 Nuclear genome Mitochondrial genome (a) An animal cell Nuclear genome Chloroplast genome Mitochondrial genome (b) A plant cell Figure 17.6 The locations of genetic material in animal and plant cells. The chromosomes in the cell nucleus are collectively known as the nuclear genome. Mitochondria and chloroplasts have small circular chromosomes, which are called the mitochondrial and chloroplast genomes. all of the offspring had white leaves. Similarly, if the female was green, so were all of the offspring. The offspring of a variegated female parent could be green, white, or variegated. At the time, Correns did not understand that chloroplasts contain some genes. We now know that the pigmentation of four-o’clock leaves can be explained by the occurrence of genetically different types of chloroplasts in the leaf cells. As discussed in Chapter 8, chloroplasts are the site of photosynthesis, and their green color is due to the presence of the pigment called chlorophyll. Certain genes required for chlorophyll synthesis are found within the chloroplast DNA. The green phenotype is due to the presence of chloroplasts that have normal genes and synthesize the usual quantity of chlorophyll. The white phenotype is caused by a mutation in a gene within the chloroplast DNA that prevents the synthesis of most of the chlorophyll. Brooker−Widmaier−Graham−Stiling: Biology 358 III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 105 UNIT III – CHAPTER 17 Correns’ crosses Cross 1 All white offspring Reciprocal cross of cross 1 All green offspring Cross 2 (Enough chlorophyll is made for the plant to survive.) The variegated phenotype occurs in leaves that have a mixture of the two types of chloroplasts. Leaf pigmentation follows a maternal inheritance pattern because the chloroplasts in four o’clocks are inherited only through the cytoplasm of the egg (Figure 17.8). During plant fertilization, a sperm cell from a pollen grain fertilizes an egg cell to create a zygote, which eventually develops into a plant. In four o’clocks, the egg cell contains several proplastids that are inherited by the offspring, while the sperm cell does not contribute any proplastids. As discussed in Chapter 4, proplastids develop into various types of plastids, including chloroplasts. Thus, the phenotype of a four-o’clock plant depends on the types of proplastids it inherits from the maternal parent. If the maternal parent transmits only normal proplastids, all offspring will have green leaves (Figure 17.8a). Alternatively, if the maternal parent transmits only mutant proplastids, all offspring will have white leaves (Figure 17.8b). The genetic composition of the paternal parent does not affect the outcome. Because an egg cell contains several proplastids, an offspring from a variegated maternal parent may inherit only normal proplastids, only Normal proplastid will produce chloroplasts with a normal amount of green pigment. Mutant proplastid will produce chloroplasts with very little pigment. Egg cell Green, white, or variegated offspring Reciprocal cross of cross 2 (a) Egg cell from a maternal parent with green leaves (b) Egg cell from a maternal parent with white leaves All green offspring Figure 17.7 Maternal inheritance in the four-o’clock plant. The genes for green pigment synthesis in plants are part of the chloroplast genome. The white phenotype in four o’clocks is due to chloroplasts with a mutant allele that greatly reduces green pigment production. The variegated phenotype is due to a mixture of normal and mutant chloroplasts. In four o’clocks, the egg contains all of the plastids that are inherited by the offspring, so the phenotype of the offspring is determined by the female parent. Biological inquiry: In this example, where is the gene located that causes the green color of four-o’clock leaves? How is this gene transmitted from parent to offspring? (c) Possible egg cells from a maternal parent with variegated leaves Figure 17.8 Plastid composition of egg cells from green, white, and variegated four-o’clock plants. In this drawing of four-o’clock egg cells, normal proplastids are represented as green and mutant proplastids as white. Proplastids do not differentiate into chloroplasts in egg cells, and they are not actually green. (a) A green plant produces eggs carrying normal proplastids. (b) A white plant produces eggs carrying mutant proplastids. (c) A variegated plant produces eggs that may contain either or both types of proplastids. 106 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance COMPLEX PATTERNS OF INHERITANCE mutant proplastids, or a mixture of normal and mutant proplastids. Consequently, the offspring of a variegated maternal parent can be green, white, or variegated individuals (Figure 17.8c). The variegated phenotype is due to segregation events that occur after fertilization. As a zygote containing both types of chloroplasts divides to produce a multicellular plant, some cells may receive mostly normal chloroplasts. Further division of these cells gives rise to a patch of green tissue. Alternatively, as a matter of chance, other cells may receive mostly mutant chloroplasts that are defective in chlorophyll synthesis. This results in a patch of tissue that is white. In most species of plants, the egg cell provides most of the zygote’s cytoplasm, while the much smaller male gamete often provides little more than a nucleus. Therefore, chloroplasts are most often inherited via the egg. In seed-bearing plants, maternal inheritance of chloroplasts is the most common transmission pattern. However, certain species exhibit a pattern called biparental inheritance, in which both the pollen and the egg contribute chloroplasts to the offspring. Others exhibit paternal inheritance, in which only the pollen contributes these organelles. For example, most types of pine trees show paternal inheritance of chloroplasts. Mitochondrial Genomes Are Maternally Inherited in Humans and Most Other Species Mitochondria are found in nearly all eukaryotic species. Similar to the transmission of chloroplasts in plants, maternal inheritance is the most common pattern of mitochondrial transmission in eukaryotic species, although some species do exhibit biparental or paternal inheritance. The mitochondrial genome of many mammalian species has been analyzed and usually contains a total of 37 genes. Twenty-four genes encode tRNAs and rRNAs, which are needed for translation inside the mitochondrion. Thirteen genes encode proteins that are involved in oxidative phosphorylation. As discussed in Chapter 7, the primary function of the mitochondrion is the synthesis of ATP via oxidative phosphorylation. In humans, as in most species, mitochondria are maternally inherited. Researchers have discovered that mutations in human mitochondrial genes can cause a variety of rare diseases (Table 17.2). These are usually chronic degenerative disorders that affect the brain, eyes, heart, muscle, kidney, and endocrine glands. For example, Leber’s hereditary optic neuropathy (LHON) affects the optic nerve. It may lead to the progressive loss of vision in one or both eyes. LHON can be caused by a mutation in one of several different mitochondrial genes. 17.4 X Inactivation, Genomic Imprinting, and Maternal Effect We will end our discussion of complex inheritance patterns by considering examples in which the timing and control of gene expression create inheritance patterns that are determined by Table 17.2 359 Examples of Human Mitochondrial Diseases Disease Description Leber’s hereditary optic neuropathy Caused by a mutation in one of several mitochondrial genes that encode electrontransport proteins. The main symptom is loss of vision. Neurogenic muscle weakness Caused by a mutation in a mitochondrial gene that encodes a subunit of mitochondrial ATP synthase, which is required for ATP synthesis. Symptoms involve abnormalities in the nervous system that affect the muscles and eyes. Mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes Mutations in mitochondrial genes that encode tRNAs for leucine and lysine. Symptoms include strokelike episodes, secretion of lactic acid into the bloodstream, seizures, migraine headaches, and lack of coordination. Maternal myopathy and cardiomyopathy A mutation in a mitochondrial gene that encodes a tRNA for leucine. The primary symptoms involve muscle abnormalities, most notably in the heart. Myoclonic epilepsy and ragged-red muscle fibers A mutation in a mitochondrial gene that encodes a tRNA for lysine. Symptoms include epilepsy, dementia, blindness, deafness, and heart and kidney malfunctions. the sex of the individual or by the sex of the parents. The first two patterns, called X inactivation and genomic imprinting, are types of epigenetic inheritance. In epigenetic inheritance, modification of a gene or chromosome during egg formation, sperm formation, or early stages of embryo growth alters gene expression in a way that is fixed during an individual’s lifetime. Epigenetic changes permanently affect the phenotype of the individual, but they are not permanent over the course of many generations and they do not change the actual DNA sequence. For example, a gene may undergo an epigenetic change that inactivates it for an individual’s entire life, so it is never expressed in that individual. However, when the same individual makes gametes, the gene may become activated and remain active during the lifetime of an offspring that inherits the gene. At the end of this section, we will also consider genes that exhibit a bizarre inheritance pattern called the maternal effect, in which the genotype of the mother directly determines the phenotype of her offspring. Surprisingly, for maternal effect genes, the genotypes of the father and of the offspring themselves do not affect the offspring’s phenotype. As you will learn, this phenomenon is explained by the accumulation of gene products that the mother provides to her developing eggs. In Female Mammals, One X Chromosome Is Inactivated in Each Somatic Cell In 1961, the British geneticist Mary Lyon proposed the phenomenon of X inactivation, in which one X chromosome in the somatic cells of female mammals is inactivated, meaning that its genes Brooker−Widmaier−Graham−Stiling: Biology 360 III. Nucleic Acid Structure and DNA Replication 107 © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance UNIT III – CHAPTER 17 Orange fur allele Barr body O (a) Calico cat 1 In the early embryo, all X chromosomes are initially active. Black fur allele O B B O O B O B O B O B O O B B B Figure 17.9 X-chromosome inactivation in female mammals. This light micrograph shows the nucleus of a human female cell. The label shows the Barr body, a condensed, inactivated X chromosome found just inside the nuclear envelope in the somatic cells of female mammals. are not expressed. The Lyon hypothesis, as X inactivation also came to be known, was based on two lines of evidence. The first evidence came from microscopic studies of mammalian cells. In 1949, Murray Barr and Ewart Bertram identified a highly condensed structure in the cells of female cats that was not found in the cells of male cats. This structure was named a Barr body after one of its discoverers (Figure 17.9). In 1960, Susumu Ohno correctly proposed that a Barr body is a highly condensed X chromosome. Lyon’s second line of evidence was the inheritance pattern of variegated coat colors in certain mammals. A classic case is the calico cat, which has randomly distributed patches of black and orange fur (Figure 17.10a). According to the Lyon hypothesis, the calico pattern is explained by the permanent inactivation of one X chromosome in each cell that forms a patch of the cat’s skin, as shown in Figure 17.10b. The gene involved is an X-linked gene that occurs as an orange allele, XO, and a black allele, XB. A female cat that is heterozygous for this gene will be calico. (The white underside is due to a dominant allele of a different autosomal gene.) At an early stage of embryonic development, one of the two X chromosomes is randomly inactivated in each of the cat’s somatic cells, including those that will give rise to the hairproducing skin cells. As the embryo grows and matures, the pattern of X inactivation is maintained during subsequent cell divisions. For example, skin cells derived from a single embryonic cell in which the XB-carrying chromosome has been inactivated will produce a patch of orange fur, because they express only the XO allele that is carried on the active chromosome. Alternatively, a group of skin cells in which the chromosome carrying XO has been inactivated will express only the XB allele, producing a patch of black fur. Because the primary event of X inactivation is a random process that occurs at an early stage of development, the result is an animal with randomly distributed patches of black and orange fur. B O 2 In each embryonic cell, random inactivation occurs for one of the X chromosomes, which becomes a Barr body. B O O B B O O Barr bodies 3 As development proceeds, the pattern of X inactivation is maintained during cell division. (b) Process of X inactivation Figure 17.10 Random X-chromosome inactivation in a calico cat. (a) A calico cat. (b) X inactivation during embryonic development. The calico pattern is due to random X-chromosome inactivation in a female that is heterozygous for the X-linked gene with black and orange alleles. The cells at the top of this figure represent a small mass of cells making up the very early embryo. In these cells, both X chromosomes are active. At an early stage of embryonic development, one X chromosome is randomly inactivated in each cell. The initial inactivation pattern is maintained in the descendents of each cell as the embryo matures into an adult. The pattern of orange and black fur in the adult cat reflects the pattern of X inactivation in the embryo. Biological inquiry: If a female cat is homozygous for the orange allele, would it show a calico phenotype? In female mammals that are heterozygous for X-linked genes, approximately half of their somatic cells will express one allele, while the rest of their somatic cells will express the other allele. These heterozygotes are called mosaics because they are composed of two types of cells, analogous to the differentcolored pieces in the pictures called mosaics. The phenomenon 108 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 361 COMPLEX PATTERNS OF INHERITANCE of mosaicism is readily apparent in calico cats, in which the alleles affect fur color. Likewise, human females who are heterozygous for X-linked genes are mosaics, with one allele expressed in some cells and the alternative allele in other cells. Women who are heterozygous for recessive X-linked alleles usually show the dominant trait because the expression of the dominant allele in 50% of their cells is sufficient to produce the dominant phenotype. On rare occasions, a female who is heterozygous for a recessive X-linked disease-causing allele may show mild or even severe disease symptoms. Because the pattern of X-chromosome inactivation is random, there will be a small percentage of heterozygous women who happen to inactivate the X chromosome carrying the normal allele in a large percentage of their cells, as a matter of bad luck. As an example, let’s consider the recessive X-linked form of hemophilia that we discussed in Chapter 16. This type of hemophilia is caused by a defect in a gene that encodes a blood-clotting factor, called factor VIII, that is made by cells in the liver and secreted into the bloodstream. X inactivation in humans occurs when an embryo is 10 days old. At this stage, the liver contains only about a dozen cells. In most females who are heterozygous for the normal and hemophilia alleles, roughly half of their liver cells will express the normal allele. However, on rare occasions, all or most of the dozen embryonic liver cells might happen to inactivate the X chromosome carrying the dominant normal allele. Following growth and development, such a female will have a very low level of factor VIII and as a result will show symptoms of hemophilia. At this point, you may be wondering why X inactivation occurs. Researchers have proposed that X inactivation achieves dosage compensation between male and female mammals. The X chromosome carries many genes, while the Y chromosome has only a few. The inactivation of one X chromosome in the female reduces the number of expressed copies (doses) of X-linked genes from two to one. As a result, the expression of X-linked genes in females and males is roughly equal. The X Chromosome Has an X Inactivation Center That Controls Compaction into a Barr Body After the Lyon hypothesis was confirmed, researchers became interested in the genetic control of X inactivation. The cells of humans and other mammals have the ability to count their X chromosomes and allow only one of them to remain active. Additional X chromosomes are converted to Barr bodies. In normal females, two X chromosomes are counted and one is inactivated. In normal males, one X chromosome is counted and none inactivated. On occasion, however, people are born with abnormalities in the number of their sex chromosomes. In these disorders, known as Turner syndrome, Triple X syndrome, and Klinefelter syndrome, the cells inactivate the number of X chromosomes necessary to leave a single active chromosome. Chromosome Composition Number of Barr Bodies Normal female XX 1 Normal male XY 0 Turner syndrome (female) XO 0 Triple X syndrome (female) XXX 2 Klinefelter syndrome (male) XXY 1 Phenotype Although the genetic control of inactivation is not entirely understood at the molecular level, a short region on the X chromosome called the X inactivation center (Xic) is known to play a critical role. Eeva Therman and Klaus Patau identified Xic from its key role in X inactivation. The counting of human X chromosomes is accomplished by counting the number of Xics. The Xic on each X chromosome is necessary for inactivation to occur. Therman and Patau found that in cells with two X chromosomes, if one of them is missing its Xic due to a chromosome mutation, neither X chromosome will be inactivated. This is a lethal condition for a human female embryo. The expression of a specific gene within the X inactivation center is required for compaction of the X chromosome into a Barr body. This gene, discovered in 1991, is named Xist (for X inactive specific transcript). The Xist gene product is a long RNA molecule that does not encode a protein. Instead, the role of Xist RNA is to coat one of the two X chromosomes during the process of X inactivation. The Xist gene on the inactivated X chromosome continues to be expressed after other genes on this chromosome have been silenced. The process of X inactivation can be divided into three phases: initiation, spreading, and maintenance (Figure 17.11). During initiation, one of the X chromosomes is targeted for inactivation. This chromosome is inactivated during the spreading phase, so called because inactivation begins near the X inactivation center and spreads in both directions along the chromosome. Spreading requires the transcription of the Xist gene and coating of the X chromosome with Xist RNA. After coating, proteins associate with the Xist RNA and promote compaction of the chromosome into a Barr body. Maintenance refers to replication of the compacted chromosome during subsequent cell divisions. While initiation and spreading occur only during embryonic development, maintenance occurs throughout the individual’s life. Continued activity of the Xist gene on an inactivated X chromosome maintains this chromosome as a Barr body during cell division. Whenever a somatic cell divides in a female mammal, the Barr body is replicated to produce two Barr bodies. The Transcription of an Imprinted Gene Depends on the Sex of the Parent As we have seen, X inactivation is a type of epigenetic inheritance in which a chromosome is modified in the early embryo, permanently altering gene expression in that individual. Other types of epigenetic inheritance occur in which genes or chromosomes Brooker−Widmaier−Graham−Stiling: Biology 362 III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 109 UNIT III – CHAPTER 17 Barr body Coating by Xist RNA and additional proteins To be inactivated Further spreading Xic 1 Initiation: Occurs during embryonic development. The X inactivation centers (Xics) are counted and one of the X chromosomes is targeted for inactivation. Figure 17.11 Xic Xic 2 Xic Spreading: Occurs during embryonic development. It begins at the Xic and progresses toward both ends until the entire chromosome is inactivated. The Xist gene, located within the Xic, encodes an RNA that coats the X chromosome and promotes its compaction into a Barr body. Igf-2m Igf-2m (homozygous dwarf) Offspring genotype Maintenance: Occurs from embryonic development through adult life. The inactivated X chromosome is maintained as a Barr body during subsequent cell divisions. The process of X inactivation. are modified in the gametes of a parent, permanently altering gene expression in the offspring. Genomic imprinting refers to a phenomenon in which a segment of DNA is imprinted, or marked, in a way that affects gene expression throughout the life of the individual who inherits that DNA. Genomic imprinting occurs in numerous species, including insects, plants, and mammals. Imprinting may involve a single gene, a part of a chromosome, an entire chromosome, or even all of the chromosomes inherited from one parent. It is permanent in the somatic cells of a given individual, but the marking of the DNA is altered from generation to generation. Imprinted genes do not follow a Mendelian pattern of inheritance because imprinting causes the offspring to distinguish between maternally and paternally inherited alleles. Depending on how a par- Parents 3 Igf-2 Igf-2 (homozygous normal) Igf-2 Igf-2m (heterozygous) ticular gene is marked by each parent, the offspring will express either the maternal or the paternal allele, but not both. Let’s consider a specific example of imprinting that involves a gene called Igf-2 that is found in mice and other mammals. This gene encodes a growth hormone called insulin-like growth factor 2 that is needed for proper growth. If a normal copy of this gene is not expressed, a mouse will be dwarf. The Igf-2 gene is known to be located on an autosome, not on a sex chromosome. Because mice are diploid, they have two copies of this gene, one from each parent. Researchers have discovered that mutations can occur in the Igf-2 gene that block the function of the Igf-2 hormone. When mice carrying normal or mutant alleles are crossed to each other, a bizarre result is obtained (Figure 17.12). If the male parent Igf-2 Igf-2 (homozygous normal) Igf-2m Igf-2m (homozygous dwarf) Igf-2 Igf-2m (heterozygous) Allele that is transcribed in offspring Igf-2 (normal) Igf-2m (nonfunctional) Phenotype Normal Dwarf Igf-2 Igf-2m normal allele mutant allele silenced allele (from female parent) expressed allele (from male parent) Figure 17.12 An example of genomic imprinting in the mouse. In the cross on the left, a homozygous male with the normal Igf-2 allele is crossed to a homozygous female carrying a defective allele, Igf-2m. An offspring is phenotypically normal because the paternal allele is expressed. In the cross on the right, a homozygous male carrying the defective allele is crossed to a homozygous normal female. In this case, an offspring is dwarf because the paternal allele is defective and the maternal allele is not expressed. 110 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 363 COMPLEX PATTERNS OF INHERITANCE is homozygous for the normal allele and the female is homozygous for the mutant allele, all the offspring grow to a normal size. In contrast, if the male is homozygous for the mutant allele and the female is homozygous for the normal allele, all the offspring are dwarf. The reason this result is so surprising is that the normal and dwarf offspring have the same genotype but different phenotypes! These phenotypes are not the result of any external influence on the offspring’s development. Rather, the allele that is expressed in their somatic cells depends on which parent contributed which allele. In mice, the Igf-2 gene inherited from the mother is imprinted in such a way that it cannot be transcribed into mRNA. Therefore, only the paternal gene is expressed. The mouse on the left side of Figure 17.12 is normal because it expresses a functional paternal gene. In contrast, the mouse on the right is dwarf because the paternal gene is a mutant allele that results in a nonfunctional hormone. In both cases, the maternal gene is inactive due to imprinting. Why is the maternal gene not transcribed into mRNA? To answer this question we need to consider the molecular function of genes. As discussed in Chapter 13, the attachment of methyl (¬ CH3) groups to the bases of DNA can alter gene transcription. For most genes, methylation silences gene expression by causing the DNA to become more compact. For a few genes, methylation may enhance gene expression by attracting activator proteins to the promoter. Researchers have discovered that DNA methylation is the marking process that occurs during the imprinting of certain genes, including the Igf-2 gene. Figure 17.13 shows the imprinting process in which a maternal gene is methylated. The left side of the figure follows the marking process during the life of a female individual; the right side follows the same process in a male. Both individuals received a methylated gene from their mother and a nonmethylated copy of the same gene from their father. Via cell division, the zygote develops into a multicellular organism. Each time a somatic cell divides, enzymes in the cell maintain the methylation of the maternal gene, while the paternal gene remains unmethylated. If the methylation inhibits transcription of this gene, only the paternal copy will be expressed in the somatic cells of both the male and female offspring. The methylation state of an imprinted gene may be altered when individuals make gametes. First, as shown in Figure 17.13, the methylation is erased. Next, the gene may be methylated again, but that depends on whether the individual is a female or male. In females making eggs, both copies of the gene are methylated; in males making sperm, neither copy is methylated. When we consider the effects of methylation over the course of two or more generations, we can see how this phenomenon creates an epigenetic transmission pattern. The male in Figure 17.13 has inherited a methylated gene from his mother that is transcriptionally silenced in his somatic cells. Although he does not express this gene during his lifetime, he can pass on an active, nonmethylated copy of this exact same gene to his offspring. Genomic imprinting is a recently discovered phenomenon that has been shown to occur for a few genes in mammals. For some genes, such as Igf-2, the maternal allele is silenced, while for other genes the paternal allele is silenced. Biologists are still trying to understand the reason for this curious marking process. Male offspring Female offspring After fertilization, somatic cells retain the methylation pattern inherited from the parents. 1 Maternal Paternal chromosome chromosome Paternal chromosome All somatic cells All somatic cells – CH3 – CH3 – CH3 – CH3 – CH3 – CH3 Erasure 2 Male gameteproducing cell No methylation During egg formation, the gene is always methylated, while during sperm formation it is not. – CH3 – CH3 – CH3 – CH3 – CH3 – CH3 Formation of sperm Formation of eggs Figure 17.13 – CH3 – CH3 – CH3 During gamete formation, methylation is erased. New methylation – CH3 – CH3 – CH3 – CH3 – CH3 – CH3 Erasure Female gameteproducing cell 3 Maternal chromosome – CH3 – CH3 – CH3 Genomic imprinting via DNA methylation. The cells at the top of this figure have a methylated gene inherited from the mother and a nonmethylated version of the same gene inherited from the father. This pattern of methylation is the same in male and female offspring and is maintained in their somatic cells. The methylation is erased during gamete formation, but in females the gene is methylated again at a later stage in the formation of eggs. Therefore, females always transmit a methylated, transcriptionally silent copy of this gene, while males transmit a nonmethylated, active copy. Brooker−Widmaier−Graham−Stiling: Biology 364 III. Nucleic Acid Structure and DNA Replication 111 © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance UNIT III – CHAPTER 17 For Maternal Effect Genes, the Genotype of the Mother Determines the Phenotype of the Offspring In epigenetic inheritance, genes are altered in ways that affect their expression in an individual or the individual’s offspring. As we have seen, some of these alterations produce strange inheritance patterns, in which organisms with the same genotype have different phenotypes. Another strange inheritance pattern, with a very different explanation, involves a category of genes called maternal effect genes. Inheritance patterns due to maternal effect genes were first identified in the 1920s by A. E. Boycott, in his studies of the freshwater snail Lymnaea peregra. In this species, the shell and internal organs can be arranged in either a right-handed (dextral) or a left-handed (sinistral) direction. The dextral orientation is more common and is dominant to the sinistral orientation. Whether a snail’s body curves in a dextral or a sinistral direction depends on the pattern of cell division immediately following fertilization. Figure 17.14 shows the results of Boycott’s crosses of truebreeding strains of snails with either a dextral or a sinistral orientation. When a dextral female (DD) was crossed to a sinistral male (dd), all of the offspring were dextral. However, crossing a sinistral female (dd) to a dextral male (DD) produced the opposite result: all of the offspring were sinistral. These seemingly contradictory outcomes could not be explained in terms of Mendelian inheritance. Alfred Sturtevant later suggested that snail coiling is due to a maternal effect gene that exists as a dextral (D) and a sinistral (d) allele. In the cross shown on the left, the P generation female is dextral (DD) and the male is sinistral (dd). In the cross on the right, the female is sinistral (dd) and the male is dextral (DD). In either case, the F1 offspring are Dd. When the F1 individuals from these two crosses are mated to each other, a genotypic ratio of 1 DD : 2 Dd : 1 dd is predicted for the F2 generation. Because the D allele is dominant to the d allele, a Mendelian inheritance pattern would produce a 3:1 phenotypic ratio of dextral to sinistral snails. Instead, the snails of the F2 generation were all dextral. To explain this observed result, Sturtevant proposed that the phenotype of the F2 offspring depended solely on the genotype of the F1 mother. Because the F1 mothers were Dd, and the D allele is dominant, the F2 offspring were dextral even if their genotype was dd! Sturtevant’s hypothesis is supported by the ratio of phenotypes seen in the F3 generation. When members of the F2 generation were crossed, the F3 generation exhibited a 3:1 ratio of dextral to sinistral snails. These F3 phenotypes reflect the genotypes of the F2 mothers. The ratio of genotypes for the F2 females was 1 DD : 2 Dd : 1 dd. The DD and Dd females produced dextral offspring, while the dd females produced sinistral offspring. This is consistent with the 3:1 phenotypic ratio in the F3 generation. The peculiar inheritance pattern of maternal effect genes can be explained by the process of egg maturation in female animals (Figure 17.15). Maternal cells called nurse cells surround a de- P DD dd dd DD F1 Dd All sinistral Dd All dextral F2 Males and females 1 DD : 2 Dd : 1 dd All dextral Cross to each other F3 Males and females 3 dextral : 1 sinistral Figure 17.14 The inheritance of snail coiling direction as an example of a maternal effect gene. In the snails shown in this experiment, the direction of body coiling is controlled by a single pair of genes. D (dextral, or right-handed) is dominant to d (sinistral, or left-handed). The genotype of the mother determines the phenotype of the offspring. A DD or Dd mother will produce dextral offspring and a dd mother will produce sinistral offspring, regardless of the genotypes of the father and of the offspring themselves. Biological inquiry: An offspring has a genotype of Dd and coils to the left. What is the genotype of its mother? veloping egg cell and provide it with nutrients. Within these diploid nurse cells, both copies of a maternal effect gene are activated to produce their gene products. The gene products are transported into the egg, where they persist for a significant time during embryonic development. The D and d gene products influence the pattern of cell division during the early stages of the snail’s embryonic development. If an egg receives only the D gene product, the snail will develop a dextral orientation, while an egg that receives only the d gene product will produce a 112 Brooker−Widmaier−Graham−Stiling: Biology III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance COMPLEX PATTERNS OF INHERITANCE Mother is DD. DD Nurse cells DD D gene products DD DD Mother is Dd. 365 Mother is dd. Dd dd Dd DD Dd DD dd D and d gene products Dd Dd Dd d gene products dd dd dd dd Egg All offspring are dextral because the egg received the gene products of the D allele. All offspring are dextral because the egg received the gene products of the D and d alleles, but the D gene products are dominant. All offspring are sinistral because the egg received the gene products of the d allele. Figure 17.15 The mechanism of maternal effect in snail coiling. In this simplified diagram, the mother’s diploid nurse cells transfer gene products to the egg as it matures. These gene products persist after fertilization, affecting development of the early embryo. If the nurse cells are DD or Dd, they will transfer the dominant D gene product to the egg, causing the offspring to be dextral. If the nurse cells are dd, only the d gene product will be transferred to the egg and the offspring will be sinistral. snail with a sinistral orientation. If an egg receives both D and d gene products, the snail will be dextral because the D gene product is dominant over d. In this way, the gene products of nurse cells, which are determined by the mother’s genotype, influence the development of the offspring. Several dozen maternal effect genes have been identified in experimental organisms, such as Drosophila. Recently, they have also been found in mice and humans. As we will discuss in Chapter 19, the products of maternal effect genes are critically important in the early stages of animal development. 17.3 • 17.1 A variety of inheritance patterns are more complex than Mendel had realized. Many of these do not obey one or both of his laws of inheritance. (Table 17.1) When the alleles of one gene mask the effects of the alleles of a different gene, this type of gene interaction is called epistasis. (Figure 17.1) • Quantitative traits such as height and weight are polygenic, which means that several genes govern the trait. Often, the alleles of such genes contribute in an additive way to the phenotype. This produces continuous variation in the trait, which is graphed as a bell curve. (Figure 17.2) 17.2 Mitochondria and chloroplasts carry a small number of genes. The inheritance of such genes is called extranuclear inheritance. (Figure 17.6) • Chloroplasts in the four-o’clock plant are transmitted via the egg, a pattern called maternal inheritance. (Figures 17.7, 17.8) • Several human diseases are known to be caused by mutations in mitochondrial genes, which follow a maternal inheritance pattern. (Table 17.2) Gene Interactions • 17.4 When two different genes are on the same chromosome, they are said to be linked. Linked genes tend to be inherited as a unit, unless crossing over separates them. (Figures 17.3, 17.4) • The percentage of offspring produced in a two-factor testcross can be used to map the relative locations of genes along a chromosome. (Figure 17.5) X Inactivation, Genomic Imprinting, and Maternal Effect • Epigenetic inheritance refers to patterns in which a gene is inactivated during the life of an organism, but not over the course of many generations. • X inactivation in mammals occurs when one X chromosome is randomly inactivated in females. If the female is heterozygous for an X-linked gene, this can lead to a variegated phenotype. (Figures 17.9, 17.10) • X inactivation occurs in three phases: initiation, spreading, and maintenance. (Figure 17.11) • Imprinted genes are inactivated by one parent but not both. The offspring expresses only one of the two alleles. (Figure 17.12) • During gamete formation, methylation of a gene from one parent is a mechanism to achieve imprinting. (Figure 17.13) Genes on the Same Chromosome: Linkage, Recombination, and Mapping • Extranuclear Inheritance: Organelle Genomes • Brooker−Widmaier−Graham−Stiling: Biology 366 III. Nucleic Acid Structure and DNA Replication © The McGraw−Hill Companies, 2008 17. Complex Patterns of Inheritance 113 UNIT III – CHAPTER 17 • For maternal effect genes, the genotype of the mother determines the phenotype of the offspring. This is explained by the phenomenon that the mother’s nurse cells contribute gene products to egg cells that are needed for early stages of development. (Figures 17.14, 17.15) 1. Quantitative traits such as height and weight are governed by several genes that usually contribute in an additive way to the trait. This is called a. independent assortment. b. discontinuous inheritance. c. maternal inheritance. d. linkage. e. polygenic inheritance. 2. When two genes are located on the same chromosome they are said to be a. homologous. d. linked. b. allelic. e. polygenic. c. epistatic. 8. When a gene is inactivated during gamete formation and that gene is maintained in an inactivated state in the somatic cells of offspring, such an inheritance pattern is called a. linkage. b. X inactivation. c. maternal effect. d. genomic imprinting. e. polygenic inheritance. 9. Calico coat pattern in cats is the result of a. X inactivation. d. genomic imprinting. b. epistasis. e. maternal inheritance. c. organelle heredity. 10. Maternal effect inheritance can be explained by a. gene products that are given to an egg by the nurse cells. b. the methylation of genes during gamete formation. c. the spreading of X inactivation from the Xic locus. d. the inheritance of alleles that contribute additively to a trait. e. none of the above. 3. Based on the ideas proposed by Morgan, which of the following statements concerning linkage is not true? a. Traits determined by genes located on the same chromosome are likely to be inherited together. b. Crossing over between homologous chromosomes can create new gene combinations. c. Crossing over is more likely to occur between genes that are closer together. d. The probability of crossing over depends on the distance between the genes. e. All but one of the above statements are correct. 1. Define linkage and linkage group. 4. In genetic linkage mapping, 1 map unit is equivalent to a. 100 base pairs. b. 1 base pair. c. 10% recombination frequency. d. 1% recombination frequency. e. 1% the length of the chromosome. 2. What were the expected results of Bateson and Punnett’s cross? 5. Organelle heredity is possible because a. gene products may be stored in organelles. b. mRNA may be stored in organelles. c. some organelles contain genetic information. d. conjugation of nuclei occurs before cellular division. e. both a and c. 6. In many organisms, organelles such as the mitochondria are contributed by only the egg. This phenomenon is known as a. biparental inheritance. b. paternal inheritance. c. maternal effect. d. maternal inheritance. e. both c and d. 7. Modification of a gene during gamete formation or early development that alters the way the gene is expressed during the individual’s lifetime is called a. maternal inheritance. b. epigenetic inheritance. c. epistasis. d. multiple allelism. e. alternative splicing. 2. Explain extranuclear inheritance and give two examples. 3. Define genomic imprinting. 1. What hypothesis were Bateson and Punnett testing when conducting the crosses in the sweet pea? 3. How did the observed results differ from the predicted results? How did Bateson and Punnett explain the results of this particular cross? 1. Discuss two types of gene interactions. 2. Discuss the concept of linkage. www.brookerbiology.com This website includes answers to the Biological Inquiry questions found in the figure legends and all end-of-chapter questions.