* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download phenols - Gneet`s
Ring-closing metathesis wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
George S. Hammond wikipedia , lookup
Asymmetric induction wikipedia , lookup
Marcus theory wikipedia , lookup
Hydroformylation wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Homoaromaticity wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Aromaticity wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Petasis reaction wikipedia , lookup
Aromatization wikipedia , lookup
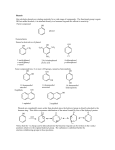
PHENOLS www.gneet.com PHENOL Phenol are the aromatic compounds having hydroxyl group (-OH) directly attached to benzene ring. ELECTRONIC STRUCTURE OF PHENOL In phenols the –OH group is attached to sp2 hybrid carbon of an aromatic ring and the oxygen atom of the hydroxyl group has two lone pairs of electrons and the bond angle in phenol is 109o. The C-O bond length in phenol (136pm) is slightly less than in methanol(412pm) due to resonance in aromatic ring of phenol Phenol has dipole moment 1.54D where as methanol has dipole moment 1.71D. This smaller dipole moment of phenol is due to the electron attracting effect of phenyl group in contrast to the electron releasing effect of methyl ( or alkyl) group in alcohol CLASSIFICATION OF PHENOL Phenols are classified as mono, di and trihydric phenols according to the number of hydroxyl groups attached to the aromatic ring. 1. Monohydric phenols 2. Dihydric phenols 3. Trihydric phenols 1 www.gneet.com PHENOLS www.gneet.com GENERAL METHODS OF PREPARATION OF PHENOLS 1. Alkali fusion of sulphonates Sodium salt of aryl sulphonic acids on fusion with sodium hydroxide at 300-350oC yield phenol The above reaction is laboratory method for preparation of phenol 2. Hydrolysis of diazonium salt 3. Hydrolysis of aryl halides ( Dow’s process) Aryl halides on hydrolysis yield phenol but the process is not so simple because halogen atom attached to benzene ring does not undergo SN2 reaction since C-X bond is resonance stabilized. Hence reaction takes place at high pressure and elevated pressure. 2 www.gneet.com PHENOLS www.gneet.com However if some electron withdrawing group is attached to benzene ring, then reaction conditions get relaxed 4. Decarboxylation of salicylic acid 5. Oxidation of Grignard reagent 3 www.gneet.com PHENOLS www.gneet.com 6. Oxidation of aromatic hydrocarbons Catalyst used in this reaction is cupric salt. 7. Oxidation of iso-propyl benzene (Cumene) Cumene is prepared by Friedel Crafts alkylation of benzene with 2-Chloropropane. 4 www.gneet.com PHENOLS www.gneet.com 8. Rasching’s process PHYSICAL PROPERTIES OF PHENOL 1. Physical state Phenols are colourless liquids or low melting point solids, but they reddish brown due to auto oxidation on exposure to air and light. Phenols are poisonous in nature but act as disinfectant and antiseptic. 2. Solubility Phenols form H-Bonds with water molecule and hence soluble in water, but their solubility is lower than that of alcohols because of large hydrocarbon part Intermolecular H-bonding among water and phenol molecules 3. Boiling point Phenols have much higher boiling point than there corresponding hydrocarbons due to intermolecular hydrogen bonding Amongst the isomeric nitrophenols, O-nitrophenol has much lower melting point and solubility than meta and para isomers of nitrophenol because of intramolecular H-bonding. Thus it does not undergo association with other molecules. In fact ortho-nitrophenol is steam volatile On the other hand meta and para nitrophenol exhibit intermolecular H-bonding with their own molecules as well as with water molecules and hence show comparatively higher melting and solubility. i) Intermolecular H-bonding ( m –nitrophenol) 5 www.gneet.com PHENOLS www.gneet.com ii) H- bonding with water molecules ( m-nitrophenol) iii) Intermolecular H- bonding ( p –nitrophenol) iv) H – bonding with water molecules ( p – nitrophenol) CHEMCIAL PROPERTIES OF PHENOL (A) Reactions involving cleavage of O-H bond 1. Acidic character of phenol (i) Reaction with active metals (ii) Reaction with alkalies 6 www.gneet.com PHENOLS www.gneet.com They do not react with carbonates and bicarbonates. Phenols are weaker acid as compared to carboxylic acid because of polar O-H group in them. The acidic nature of phenol is due to formation of stable phenoxide ion in solution to give H+ ions Phenol behaves as stronger acid than alcohol i. The greater acidity of phenol is due to stability of phenoxide ion is resonance stabilised . ii. Resonance structure of phenol iii. As a result of resonance it weakens the polar O-H bond and thus facilitates the release of proton (H+) to give phenoxide ion which is also stable due to resonance iv. Resonating structure of phenoxide ion v. Both phenol and phenoxide ion are stable due to resonance. But phenoxide ion is more stabilized than phenol because the resonating structure of phenoxide ion carry only negative structure of phenol involves separation of positive and negative charge. This charge delocalization is a stabilizing factor in phenoxide ion and increases acidity of phenol. 7 www.gneet.com PHENOLS www.gneet.com vi. a. Effect of substituent on the acidity of phenols Electron withdrawing group (EWG) 1. Electron withdrawing groups NO2 , -X, -CHO, -COOH, -CN etc stabilizes the phenoxide ion more by dispersing the negative charge relative to phenol ( i.e. proton release become easy) and thus increases the acidic strength of phenols 2. Particular effect is more when the substituent is present on O – and p- positions than in mposition. Thus acidic strength of nitrophenol decreases in the order p-nitrophenol > o-nitrophenol > m-nitrophenol > phenol 3. Further greater the number of electron withdrawing groups at o- and p – position, more acidic the phenol. b. Electron donating group (EDG) (i) Electron donating group –R, -NH2, -OR etc destabilize the phenoxide ion by donating electrons and intensify the negative charge relative to phenol (i.e. proton release become difficult)and thus decreases the acidic strength of phenol. (ii) The effect is more when the substituent is present on o- and p- position than on mposition with respect to –OH group. Thus cresol are less acidic than phenol. m – methoxy and m-amnion phenols are stronger acid than phenols because of –I effect ( of – OCH3 and –NH2 groups) and absence of +R effect. m –methoxy phenol > m-amino phenol > phenol > O-methoxy phenol > p-methoxy phenol vii) Reaction of –OH group i. Reaction with FeCl3 is the test of phenol 8 www.gneet.com PHENOLS www.gneet.com 3C6H5OH + FeCl3 (C6H5O)3Fe + 3HCl (C6H5O)3Fe : Ferric phenoxide ( violet complex) ii. Ester formation Reaction with benzoyl chloride is known as benzoylation Benzoylation of alcohols o phenols in presence of NaOH is called Schotten-Baumann reaction. iii. Ether formation Claisen rearrangement 9 www.gneet.com PHENOLS www.gneet.com (B) Electrophilic aromatic substitution reaction 1. Halogenation Due to steric hinderance at ortho position, para-product predominates 2. Sulphonation 3. Nitration 4. Mercuration 10 www.gneet.com PHENOLS www.gneet.com 5. Friedel crafts alkylation and acylation 6. Nitrosation 7. Kolbe’s reaction 11 www.gneet.com PHENOLS www.gneet.com Mechanism (a) Acetyl salicylic acid ( Asprine) It is a white solid (m.pt. 408K) and is used for relieving pain (analgesic) and to bring down the body temperature (antipyretic during fever) (b) Methyl salicylate 12 www.gneet.com PHENOLS www.gneet.com It is an oily liquid (b.pt. 495K) with pleasant odour (oil of wintergreen) used in perfumery and flavouring agent. It is also used in medicine in the treatment of rheumatic pain and as a remedy for aches, sprains and bruises. (c) phenyl salicylate ( salol) It is white solid (m.pt 316K) and is used as an intestinal antispeptic. 8. Reimer-Tiemann reaction Mechanism The electrophile, dichloromethylene CCl2 is generated from chloroform by action of base OH- + CHCl3 ⇆ HOH + CCl3 →Cl- + CCl2 13 www.gneet.com PHENOLS www.gneet.com Attack of electrophile ( :CCl2) on phenoxide ion Salicyaldehyde If the reaction is carried out with carbon tetrachloride (CCl4) instead of chloroform, o-hydroxy benzoic acid (salicylic acid) is formed. TESTS TO DISTINGUISH BETWEEN ALCOHOLS AND PHENOLS 1. Litmus test Phenol turns blue litmus red being acidic in nature but alcohol do not. 2. Ferric chloride test Phenol react with neutral ferric chloride solution to form violet or green coloured solution where as alcohol do not undergo such reactions. 3. Bromine water test Aqueous solution of phenol forms white precipitate of 2,4,6-tribromo phenol, when treated with bromine water. However alcohol so not give such precipitation. 14 www.gneet.com