* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Unit
Cell nucleus wikipedia , lookup
Endomembrane system wikipedia , lookup
Programmed cell death wikipedia , lookup
Extracellular matrix wikipedia , lookup
Tissue engineering wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cell growth wikipedia , lookup
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cytokinesis wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
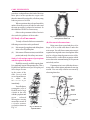
Core Experiments : Unit - 3 In this unit experiments to study animal and plant cells will be performed. For this purpose you will make temporary slides of various plant and animal material to study them. According to curriculum, in the following pages instruction are given for the following excercises. 1. To make a temporary mount of root apex to study the steps of mitosis. 2. To make a slide of flower bud to study the steps of meiosis. 3. To study cyclosis in Hydrilla and Paramaecium. 4. To make a slide of onion peel to study structure of plant cell. 5. To make a slide of cheek epithelial cell of frog / man to study structure of animal cell. 6. To study the presence of cell wall components cellulose, lignin, suberin, mucilage in plant cell. Since to study the above phenomena, temporary slides have to be made, therefore the procedure of making temporary slides will be explained first. Temporary Slide Mounting Temporary slides are made to study microscopic organisms and the cells and tissues of plants and animal since these slides cannot be preserved for long, they are called temporary slides. (3) Stain the given tissue by the given method and place it on the centre of slide. (4) To find the centre of the slide place the slide on a paper, and draw its outline. Now join the opposite corners of the slide by drawing a line. Where the two lines intersect, it is the centre of the slide. Place the slide back on the figure and since the glass slide is transparent the centre point will be easily visible. (5) Now put a drop of glycerine on the tissue placed on the centre of the slide and with the help of a needle slowly lower the cover slip on the material. Avoid entry of air bubbles (fig 3.2). (6) Blot the excess glycerine with cloth or blotting paper . To make any temporary slide the following materials are required : Microslide Cover slip Needles with handles Forceps & dropper Watch glass glycerine Necessary stains such as eocin, methylene blue saffranine, haemotoxylin acetocarmine etc. Instructions for making a temporary slide: (1) Dip the given slide in water and then clean it with a cotton cloth followed by a silk cloth. (2) Clean the cover-slip lightly with a silk cloth only Fig. 3.1 To find centre of slide 35 36 (7) PRACTICAL BIOLOGY - XI Stuck a lebel on one slide of the slide bearing name of tissue and your name. 4. Press the cover slip lightly and observe the slide under the microscope. nuclear membrane Aster nucleolus Chromatid chromatin thread centriole cellmembrane Fig. 3.2 Placing cover slip (a) Interphase centromere (b) Prophase (c) Metaphase centriole nucleolus Cell Division chromatin thread nuclear membrane 6. Mitosis Aim of Exeperiment : To study various steps of mitotic cell division in the root tip. Materials required : Root tips of onion, FAA solution, IN HCl, spirit lamp, acetocarmine stain, needle, slide, cover slip, watch glass, foceps, droppers etc. To perform this experiment, first take 23 onion and place them on a glass of water without dipping in the water. Place the glass in slightly dark place and study it daily when the young roots are about 2-3 mm long, then in the morning at around 8.00 a.m. cut 2-3 mm of the root apex and transfer in a small bottle in which FAA solution is present. FAA solution is made from formaline, acetic acid and 90% alcohol are mixed in equal proportions FAA solution is ready for use. Procedure : 1. Take out 2-3 piece of root tips kept in FAA, place them in watch glass and wash them with water. 2. Now place the root tips on a slide and add few drops of IN HCl on them. Heat the slide on a low flame with a needle, it can also be teased. 3. Now put a few drops of acetocarmine stain on the material and heat the slide. After sometime cover the material with a cover slip. (d) Anaphase (e) Telophase Fig. 3.3 Different stages of Mitotic Cell Division Observation : Observe the slide first under low power and then under high power, Find the stages of prophase, metaphase, anaphase and telophase and compare them with the given figures. Note : Observe permanent slides of the various phases also. (7) Meiosis For observing meiotic cell division the above N membrane Pair of homologous chromosomes Prophase A Leptotene B Zygotene Tetrad C Pachytene Crossing over D Diplotene E Dikinesis Spindle fibres Centromere F Metaphase-I G Anaphase-I H Telophase-I I Interphase-I 4 daughter cells J Prophase-11 K Metaphase-11 L Anaphase-1 M Telophase-1 Fig. 3.4 Different stages of Meiotic Cell Division CORE EXPERIMENTS 37 Chloroplast procedure is adopted but as this kind of division takes place in the reproductive organ cells therefore instead of root tip cells, cells from young buds are preserved in FAA When experiment has to be performed the anthers from the preserved buds are taken and the same procedure is adopted as in mitosis. In this addition of IN HCl is not necessary. Observe the permanent slides of meiosis also under the guidance of the teacher. (8) Study of cell movement : To study cells and cellular movements the following two activities can be performed: (a) Movement of peotoplasm and chloroplasts in the cells of Hydrilla plant. (b) Movement of Paramaecium (a unicellular protist) and study of its ciliary movement. Study of cells and movement of protoplasm and chloroplast in Hydrilla : Hydrilla is an easily available aquatic plant. It is commonly kept in fish aquarium also. Take a Hydrilla leaf and take out its peel. Keep the peel in few drops of water in the centre of the slide. Cover it with cover slip and abserve it under the m i c r o s c o p e immediately. You will observe rectangular cells of Hydrilla and a large vacuole in the cells with the cytoplasm and green coloured chloroplast moving around the vacuole in a circular manner (cyclosis) as shown in Fig. 3.6 Fig. 3.6 Movement of protoplasm and chloroplant in Plant Cell (B) Movement in Paramaecium : Bring water from a pond and place a few drops of it on a slide and observe under the microscope. You will observe many microscopic organism moving in the water. Among them there may be a slipper shaped paramaecuim visible. Paramaecuim is a unicellular organism which moves due to the constant beating of cilia present on its body surface. If paramaecuim is not visible then observe many other cellular plants and animals moving around. You can study cellular movement by observing them also. Upper edge Upper contractile vacuole Micro Nucleus Macro nucleus Mouth Notch Protoplasm Post end Cell Mouth lower contractile vacuole Cilia Fig. 3.5 Branch of a Hydrilla Plant Fig. 3.7 Paramaecium 38 (9) PRACTICAL BIOLOGY - XI Slide of Human / Frog cheek epithelial cells : Materials required : Slide, cover slip, tooth pick, dropper, water, watch glass, methylene blue, needle, glycerine etc. Cell group Nucleus Fig. 3.8 Method of removal of onion peel cyto plasm Fig. 3.10 Epithelial cell of cheek Procedure : peel and place it on the centre of the slide (4) Put a drop of iodine solution on the peel and then wash it gently with a few drops of water. (5) Now put a drop of glycerine on the peel and carefully place a cover slip on it so that air bubble don’t enter. (6) Observe the slide under the microscope. Wash the mouth throughtly and with the help of tooth pick scrape the inner side of your cheek. Take the scraping on a slide and put a few drops of water on it. Add two to three drops of methylene blue on it. Cover it with a cover slip after a few minutes. Observation : Observe the slide under the microscope. Try to locate the nucleus in the cell. The same experiment can be done using a chloroformed frog to get scraping from the cheek. (i) In the simple microscope rectangular cells are clearly visible. Nucleus is also visible. (ii) Each cell has a nucleus and large vacuoles are visible. (iii) Each cell is surrounded by a cell wall. (11) Tests for various components in cell wall of plant cells : Nucleus (10) Study of plant cell in an onion peel Materials required : An onion, Blade or knife, watch glass, water, forceps dropper, iodine solution, slide and cover slip. Rectangular cells Procedure : (1) Cut long pieces of onion with the help of knife or blade (2) Take a fleshy scale leaf from one of the pieces and fold it on the concave surface. On doing so a then peel will come out. Keep the peel in water in a watch glass. (3) Now cut a 0.5 cm square piece of the Fig. 3.9 Cells in Onion peel As you know the plant cells are surrounded by a rigid cell wall there are many chemical components present in the cell wall - the chief among them being cellulose, lignin, suberin and mucilage. Carry out the follow tests to study them. (A) Test for Cellulose : CORE EXPERIMENTS Materials required : Microscope, glass slide, cover slip, iodine solution, 75% conc. H2SO4, water paper or cotton fibres. Procedure : Place a few fibres of cotton and study them under the microscope. Now add a few drops on the fibres and put a few drops of iodine solution. The fibres turn brown in colour. Now add drop of 75% H2SO4 and a drop of water. Observation : The fibres turn yellow in colour, which shows the presence of cellulose (The presence of cellulose can be observed in a section of the stem of tomato plant. For that place a thin T.S. of stem in iodine solution. After a minute blot out the iodine solution with a filter paper and add 1 drop of 60-70% H2SO4. Place a coverslip and observe under the microscope immediately. The cell wall of the cells swell and become blue in colour. (B) Test for Lignin : This can be tested by any of the following two methods : (A) Materials required : Microscope, stem of tomato / sunflower, match stick, 1% alcoholic phloroglucin, conc. HCl etc. Procedure : Cut thin section of stem of tomato / sunflower or take small segments of match stick on a slide. Put a drop 1% alcoholic phloroglucin on it. Cover with cover slip and add drops of conc. HCl along the sides of the coverslip with the help of a dropper. Observation : Red purple colour in the cell walls indicate the presence of lignin in them. (b) Material required : Stem of tomato / sunflower, match stick, 1% potassium permanganate solution in water, dil 2% HCl and sodium bicarbonate. Procedure : Cut thin sections of the stem or match stick and place them in 1% KMnO4 solution in a watch glass for 15-20 mins. After that remove the KMnO4 solution and wash the sections in dil 2% 39 HCl. Now trnasfer the sections in another watch glass and wash them several times with water. After that add a few drops of Sodium bicarbonate on them. Observation : Deep red colour in the sections indicate the presence of lignin in their cell walls. (C) Tests for Suberin : Material required : Cork, Sudan IV (aloholic) 50% alcohol, slide, cover slip, water, glycerine etc. Prodecure : Cut thin sections of cork and place them in alcohol Sudan IV solution for 20-25 minutes in a watch glass. When the sections are well stained, then wash the sections in 50% alcohol, two to three times to remove the excess stain. Finally wash them in water and place them on a slide in a drop of glycerine. Place a cover slip on it and observe under the microscope. Observation : Red coloured suberin is observed in the cell walls of the cork cells. (D) Tests for Mucilage : Materials required : Testa of linseed 10% CuSO4 solution, 10% NaOH, water, slide, cover slip, glycerine etc. Procedure : Take thin sections of linseed and place them for about 20 minutes in 10% CuSO4 solution. After 20 minutes wash them with water and place a section on a slide. Wash the slide once again with two drops of 10% CuSO4 and put glycerine and cover with a cover slip. Observe under the microscope. Observation : Cells having mucilage give a shiny appearance.