Identifying On the lines provided, identify each
... 10. Circle the letter of each sentence that is true about carbohydrates. a. Starches and sugars are examples of carbohydrates. b. Living things use them as their main source of energy. c. The monomers in sugar polymers are starch molecules. d. The monomers of proteins are amino acids 11. Circle the ...
... 10. Circle the letter of each sentence that is true about carbohydrates. a. Starches and sugars are examples of carbohydrates. b. Living things use them as their main source of energy. c. The monomers in sugar polymers are starch molecules. d. The monomers of proteins are amino acids 11. Circle the ...
02 Chemistry b - Crestwood Local Schools
... Most are globular proteins that act as biological catalysts Holoenzymes consist of an apoenzyme (protein) and a cofactor (usually an ion) Enzymes are chemically specific ...
... Most are globular proteins that act as biological catalysts Holoenzymes consist of an apoenzyme (protein) and a cofactor (usually an ion) Enzymes are chemically specific ...
EOC Macromolecules
... brinogen is made of chains of amino acids, it is an example of which type of organic molecule? A. ...
... brinogen is made of chains of amino acids, it is an example of which type of organic molecule? A. ...
Zoology – The Chemical Basis of Animal Life
... 3. Fats and oils are produced from one molecule of glycerol & 3 fatty acid molecules. a.Most fatty acids consist of a chain with 16 to 18 carbon atoms. 1) Saturated fats - with hydrogen atoms attached to every available bonding site on carbons of fatty acid, and with no ...
... 3. Fats and oils are produced from one molecule of glycerol & 3 fatty acid molecules. a.Most fatty acids consist of a chain with 16 to 18 carbon atoms. 1) Saturated fats - with hydrogen atoms attached to every available bonding site on carbons of fatty acid, and with no ...
BIOCHEMISTRY STUDY GUIDE Look over Chapter 3 Review on
... What are organic molecules that catalyze reactions in living systems? What is the function of ATP? What are isomers? What are the structural differences between monosaccharides, disaccharides, and polysaccharides? How many PAIRS of electrons are shared in a single bond? Double bond? Triple bond? Wha ...
... What are organic molecules that catalyze reactions in living systems? What is the function of ATP? What are isomers? What are the structural differences between monosaccharides, disaccharides, and polysaccharides? How many PAIRS of electrons are shared in a single bond? Double bond? Triple bond? Wha ...
Chapter 4 - Cellular Metabolism 4.1 Introduction (p. 74) A. A living
... Energy is stored while converting ADP to ATP; when energy is released, ATP becomes ADP, ready to be regenerated into ATP. 4.5 Metabolic Pathways (p. 78; Fig. 4.7) A. The enzymes controlling either an anabolic or catabolic sequence of reactions must act in a specific order. B. A sequence of enzyme-co ...
... Energy is stored while converting ADP to ATP; when energy is released, ATP becomes ADP, ready to be regenerated into ATP. 4.5 Metabolic Pathways (p. 78; Fig. 4.7) A. The enzymes controlling either an anabolic or catabolic sequence of reactions must act in a specific order. B. A sequence of enzyme-co ...
Biomolecules Unit Review File
... 10. Is a starch a carbohydrate? If so is it a monosaccharide, disaccharide, or polysaccharide? What is the function of starch? 11. What is glycogen and what type of storage is it? Where is glycogen stored? 12. Draw a single nucleotide. Draw a chain of nucleic acid. How many strands does DNA have? Ho ...
... 10. Is a starch a carbohydrate? If so is it a monosaccharide, disaccharide, or polysaccharide? What is the function of starch? 11. What is glycogen and what type of storage is it? Where is glycogen stored? 12. Draw a single nucleotide. Draw a chain of nucleic acid. How many strands does DNA have? Ho ...
Chapter 3 Review Questions
... 1. DNA and RNA are examples of _________________. 2. Carbohydrates and lipids both contain the elements ______________, _____________, and _________________. 3. Proteins are made of ___________________ 4. ______________ are made of nucleotides. 5. Examples of lipids include _________________________ ...
... 1. DNA and RNA are examples of _________________. 2. Carbohydrates and lipids both contain the elements ______________, _____________, and _________________. 3. Proteins are made of ___________________ 4. ______________ are made of nucleotides. 5. Examples of lipids include _________________________ ...
Chapter 7 Study Guide
... Carbohydrates, such as glucose, are energy-rich because when catabolized they can yield a large number of electrons per molecule. Glycolysis is a pathway that degrades glucose to pyruvic acid without requiring oxygen. Pyruvic acid is processed in aerobic respiration via the Krebs cycle and its assoc ...
... Carbohydrates, such as glucose, are energy-rich because when catabolized they can yield a large number of electrons per molecule. Glycolysis is a pathway that degrades glucose to pyruvic acid without requiring oxygen. Pyruvic acid is processed in aerobic respiration via the Krebs cycle and its assoc ...
Fatty oxidation, Amino acid degradation and energy metabolism
... 3. What are the ultimate catabolic products of Amino acids in mammalians? 4. What are the carriers of amino gp (ammonia) in blood? 5. During heavy exercise muscles produce excess of pyruvate due to accelerated glycolysis and lower oxygen availability. This pyruvate is used up for gluconeo genesis. H ...
... 3. What are the ultimate catabolic products of Amino acids in mammalians? 4. What are the carriers of amino gp (ammonia) in blood? 5. During heavy exercise muscles produce excess of pyruvate due to accelerated glycolysis and lower oxygen availability. This pyruvate is used up for gluconeo genesis. H ...
File
... monomers called amino acids. • There are 20 different types identified by their R group. • Dipeptides: Two amino acids are joined by peptide bonds to form a dipeptide. • Polypeptides: A long chain of amino acids is called a polypeptide. Usually 1000’s of amino acids joined together by • peptide bond ...
... monomers called amino acids. • There are 20 different types identified by their R group. • Dipeptides: Two amino acids are joined by peptide bonds to form a dipeptide. • Polypeptides: A long chain of amino acids is called a polypeptide. Usually 1000’s of amino acids joined together by • peptide bond ...
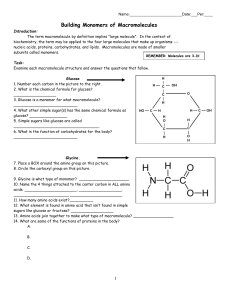
Building Monomers of Macromolecules
... 11. How many amino acids exist?__________ 12. What element is found in amino acid that isn’t found in simple sugars like glucose or fructose? __________________ 13. Amino acids join together to make what type of macromolecule? _________________ 14. What are some of the functions of proteins in the b ...
... 11. How many amino acids exist?__________ 12. What element is found in amino acid that isn’t found in simple sugars like glucose or fructose? __________________ 13. Amino acids join together to make what type of macromolecule? _________________ 14. What are some of the functions of proteins in the b ...
3. Related Pathways
... Once broken down, these monomers are able to feed into various parts of glycolysis or the Krebs cycle (Fig.1, p.117) ...
... Once broken down, these monomers are able to feed into various parts of glycolysis or the Krebs cycle (Fig.1, p.117) ...
Food Utilization
... – safe estimate of daily intake for standard needs • Essential nutrients can not be synthesized – minerals, vitamins, 8 amino acids and 1-3 fatty acids ...
... – safe estimate of daily intake for standard needs • Essential nutrients can not be synthesized – minerals, vitamins, 8 amino acids and 1-3 fatty acids ...
2.3: Carbon-Based Molecules
... • Blood contains enzyme carbonic anhydrase – Catalyzes reaction in your blood where carbon dioxide combines with water to form carbonic acid – Makes reaction one million times faster so that carbon dioxide does not build up in your blood, ...
... • Blood contains enzyme carbonic anhydrase – Catalyzes reaction in your blood where carbon dioxide combines with water to form carbonic acid – Makes reaction one million times faster so that carbon dioxide does not build up in your blood, ...
amino acids - 11 College Biology
... “KINKS” made in the fatty acid chains allow for space. Liquid at room temperature. Example: olive oil ...
... “KINKS” made in the fatty acid chains allow for space. Liquid at room temperature. Example: olive oil ...
Powerpoint Slides for Chapter Seven
... Fatty acids move from blood into cell (lipoprotein lipase) and into cytoplasm. Progressively broken into two carbon fragments (lipolysis or beta oxidation) Two carbon fragments move into mitochondria as acetyl Co A and are metabolized. Glycerol moves from blood to cell. As 3C molecule is made into p ...
... Fatty acids move from blood into cell (lipoprotein lipase) and into cytoplasm. Progressively broken into two carbon fragments (lipolysis or beta oxidation) Two carbon fragments move into mitochondria as acetyl Co A and are metabolized. Glycerol moves from blood to cell. As 3C molecule is made into p ...
Chapter 3 Chemistry of Life Modern Biology Textbook Holt
... • Adenosine triphosphate (ATP) stores and releases energy during cell processes, enabling organisms to function. • ATP is the fuel/energy for life! ...
... • Adenosine triphosphate (ATP) stores and releases energy during cell processes, enabling organisms to function. • ATP is the fuel/energy for life! ...
Notes handout for Basic Biochemistry
... Carbon can form long chains, branched structures, or rings. Adjacent carbon atoms can also form Double and Triple bonds. There are four basic classes of organic molecules: carbohydrates, lipids, proteins and nucleic acids. Most organic molecules are large polymers made up of many similar subunits, c ...
... Carbon can form long chains, branched structures, or rings. Adjacent carbon atoms can also form Double and Triple bonds. There are four basic classes of organic molecules: carbohydrates, lipids, proteins and nucleic acids. Most organic molecules are large polymers made up of many similar subunits, c ...
The Chemistry of Living Things
... • A sugar called glucose is the most important monosaccharide on Earth. It is also the simplest sugar. Glucose is used in cellular respiration and created by photosynthesis. When you think of table sugar, like the kind in candy, it is actually a disaccharide. The sugar on your dinner table is made o ...
... • A sugar called glucose is the most important monosaccharide on Earth. It is also the simplest sugar. Glucose is used in cellular respiration and created by photosynthesis. When you think of table sugar, like the kind in candy, it is actually a disaccharide. The sugar on your dinner table is made o ...
Final Review
... 1. What is the cellular organelle associated with energy production? Mitochondrion 2. The ultimate source of energy for all but a very few biochemical reactions is The sun – through the process of photosynthesis 3. The biochemical process in which simple molecules are combined to make larger ones is ...
... 1. What is the cellular organelle associated with energy production? Mitochondrion 2. The ultimate source of energy for all but a very few biochemical reactions is The sun – through the process of photosynthesis 3. The biochemical process in which simple molecules are combined to make larger ones is ...
Chem 2B
... 1. What is the cellular organelle associated with energy production? Mitochondrion 2. The ultimate source of energy for all but a very few biochemical reactions is The sun – through the process of photosynthesis 3. The biochemical process in which simple molecules are combined to make larger ones is ...
... 1. What is the cellular organelle associated with energy production? Mitochondrion 2. The ultimate source of energy for all but a very few biochemical reactions is The sun – through the process of photosynthesis 3. The biochemical process in which simple molecules are combined to make larger ones is ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.