Biology 2.3 Carbon Compounds
... oxygen, nitrogen, carbon, and phosphorus Store and transmit genetic information DNA = deoxyribonucleic acid RNA= ribonucleic acid ...
... oxygen, nitrogen, carbon, and phosphorus Store and transmit genetic information DNA = deoxyribonucleic acid RNA= ribonucleic acid ...
Cells and Energy Lesson Quiz B Completion LESSON 4
... Cells and Energy Completion Directions: On each line, write the term that correctly completes each sentence. ...
... Cells and Energy Completion Directions: On each line, write the term that correctly completes each sentence. ...
Macromolecule Expert Sheets
... 3. Draw and label the levels of organization of the protein structure. 4. What types of atoms are found in proteins? 5. Draw the general structure for an amino acid and label its functional groups. 6. Which part of an amino acid determines its specific properties? 7. When 2 amino acids are joined to ...
... 3. Draw and label the levels of organization of the protein structure. 4. What types of atoms are found in proteins? 5. Draw the general structure for an amino acid and label its functional groups. 6. Which part of an amino acid determines its specific properties? 7. When 2 amino acids are joined to ...
carbohydrate metabolism
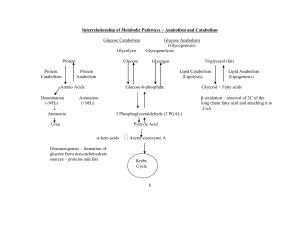
... • Catabolism is a part of metabolism– the breakdown of substances by an organism. Many important catabolic reactions occur in the mitochondria. ...
... • Catabolism is a part of metabolism– the breakdown of substances by an organism. Many important catabolic reactions occur in the mitochondria. ...
Original
... acids bond to form a dipeptide. In this condensation reaction, the two amino acids form a covalent bond (peptide bond) and release a water molecule. ...
... acids bond to form a dipeptide. In this condensation reaction, the two amino acids form a covalent bond (peptide bond) and release a water molecule. ...
Name__________________________________
... Name__________________________________ Date____________ Period____ Practice Questions Enzymes Standards 1b 1h 4e 4f ...
... Name__________________________________ Date____________ Period____ Practice Questions Enzymes Standards 1b 1h 4e 4f ...
Biomolecules
... BIOMOLECULES • Large complex molecules in cells • Formed from repeating subunits • Most biomolecules are formed from a carbon backbone • Six most common elements in living organisms is • CHONPS ...
... BIOMOLECULES • Large complex molecules in cells • Formed from repeating subunits • Most biomolecules are formed from a carbon backbone • Six most common elements in living organisms is • CHONPS ...
1. Which substances are inorganic compounds?
... 6. The reactants represented by letter B in the chart are (1.) glucose and water (2.) ATP and water (3.) proteins and water (4.) fatty acids and glycerol 7. The products represented by letter C in the chart are (1.) glucose and water (2.) ATP and water (3.) carbon dioxide and water (4.) fatty acids ...
... 6. The reactants represented by letter B in the chart are (1.) glucose and water (2.) ATP and water (3.) proteins and water (4.) fatty acids and glycerol 7. The products represented by letter C in the chart are (1.) glucose and water (2.) ATP and water (3.) carbon dioxide and water (4.) fatty acids ...
Hydrolysis and Dehydration Synthesis: How we become what we eat
... Hydrolysis and Dehydration Synthesis: How we become what we eat. Hydrolysis and dehydration synthesis are chemical processes used to break down and rebuild organic compounds. Organic Compounds: o Carbohydrates (Sugars and Starches) o Monosaccharides are simple sugars, i.e., glucose and fructose o Co ...
... Hydrolysis and Dehydration Synthesis: How we become what we eat. Hydrolysis and dehydration synthesis are chemical processes used to break down and rebuild organic compounds. Organic Compounds: o Carbohydrates (Sugars and Starches) o Monosaccharides are simple sugars, i.e., glucose and fructose o Co ...
LECTURE #1 STUDY GUIDE
... How many molecules of ATP become synthesized in the complete combustion of a neutral fat molecule? ...
... How many molecules of ATP become synthesized in the complete combustion of a neutral fat molecule? ...
File
... Polymers consisting of several hundred to several thousand monosaccharide monomers linked together. ...
... Polymers consisting of several hundred to several thousand monosaccharide monomers linked together. ...
What are organic compounds?
... • Lysis = to split apart (or breakdown) • Hydrolysis = process by which complex molecules are broken down to form simple molecules by adding water. ...
... • Lysis = to split apart (or breakdown) • Hydrolysis = process by which complex molecules are broken down to form simple molecules by adding water. ...
Unit Topic: Chemistry of Life
... 4. Describe how the difference in structure between a triglyceride and a phospholipids leads to a difference in function - phospholipids are glycerol and 2 fatty acids - they have a hydrophilic head and hydrophobic tail so a cell membrane that has a bilayer of phospholipids forms a barrier between i ...
... 4. Describe how the difference in structure between a triglyceride and a phospholipids leads to a difference in function - phospholipids are glycerol and 2 fatty acids - they have a hydrophilic head and hydrophobic tail so a cell membrane that has a bilayer of phospholipids forms a barrier between i ...
Week 4:
... We balanced the equation for cellular respiration: 6 O2 + C6H12O6 6 CO2 + 6 H2O (plus energy) and pointed out that cellular respiration allows for inputs other than glucose, including fats, proteins, and other sugars. Introduction of Photosynthesis: Reverse equation of Respiration, and can be divi ...
... We balanced the equation for cellular respiration: 6 O2 + C6H12O6 6 CO2 + 6 H2O (plus energy) and pointed out that cellular respiration allows for inputs other than glucose, including fats, proteins, and other sugars. Introduction of Photosynthesis: Reverse equation of Respiration, and can be divi ...
ABC
... question: What are 4 common types of lipids and what are their main properties/functions? ...
... question: What are 4 common types of lipids and what are their main properties/functions? ...
Cell Processes vocabulary 11/1/16
... to release stored energy in food molecules Process by which oxygen-lacking cells and some one-celled organisms release small amounts of energy from glucose molecules ...
... to release stored energy in food molecules Process by which oxygen-lacking cells and some one-celled organisms release small amounts of energy from glucose molecules ...
Document
... (I) Micronutrients not for energy, no Calories. Unique functions. Deficiencies and excesses can be life threatening. -1- minerals: inorganic. Most abundant is calcium. Includes electrolytes, iron, iodine. -2- vitamins: organic. a. water soluble = Bs and C. Many are in coenzymes. b. fat soluble = A, ...
... (I) Micronutrients not for energy, no Calories. Unique functions. Deficiencies and excesses can be life threatening. -1- minerals: inorganic. Most abundant is calcium. Includes electrolytes, iron, iodine. -2- vitamins: organic. a. water soluble = Bs and C. Many are in coenzymes. b. fat soluble = A, ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.