* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemical Equilibrium
Work (thermodynamics) wikipedia , lookup
Statistical mechanics wikipedia , lookup
Acid–base reaction wikipedia , lookup
Vapor–liquid equilibrium wikipedia , lookup
Detailed balance wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Thermodynamic equilibrium wikipedia , lookup
Ionic compound wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Thermodynamics wikipedia , lookup
Electrochemistry wikipedia , lookup
Marcus theory wikipedia , lookup
Industrial catalysts wikipedia , lookup
Enzyme catalysis wikipedia , lookup
Countercurrent exchange wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Ultraviolet–visible spectroscopy wikipedia , lookup
Reaction progress kinetic analysis wikipedia , lookup
George S. Hammond wikipedia , lookup
Rate equation wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Transition state theory wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
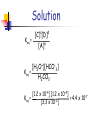
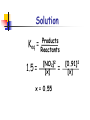
Chapter 14 Chemical Equilibrium Reaction Types There are 2 Types of Reactions 1.Completion Reactions and 2. Reversible Reactions Completion Reactions A + B C + D Completion reactions go in one direction. “All” ?? reactants are converted into products and then the reactions stops. Usually involve to formation, and escape, of a gas….you lose products to the environment Sulfur + Oxygen S8(s) + 8O2(g) Sulfur Dioxide SO2(g) Reversible Reactions will reach Equilibrium A “reversible” reaction is a reaction in which the products re-form the original reactants. NaCl(s) Na1+(aq ) + Cl1-(aq) 5 Facts about Reversible Reactions 1. Reversible reactions go in both directions. 2. Opposite reactions occur at the same rate. Rate of Dissolving = Rate of Crystallization Sodium Chloride NaCl(s) Sodium + Chlorine Na(aq) + Cl(aq) 3.Once Equilibrium is established the forward and reverse reactions will continue. Reversible Reactions 4. The Amounts of the reactants and the products will remain constant as the reaction continues. 5. Even though the rates of reactant and product production are equal, it is rare for the concentrations of both the reactant or product to be equal. There’s usual more of one than the other. Reading the Symbols Completion Reaction Reversible Reaction Reversible Reaction with a higher concentration of products Reversible Reaction with a higher concentration of reactants At Equilibrium Both forward and reverse reactions will continue to occur. The forward reaction rate is equal to the reverse reaction rate. “Dynamic” equilibrium means there is no net change! Examples of Equilibrium 1. Homeostasis… Living Cell Chemistry 2. Biogeochemical Cycles * The Earth’s “Recycling” Program - The Nitrogen Cycle - The Carbon Cycle - The Oxygen Cycle Complex Ions Complex ions can increase the solubility of certain elements. Some Reversible Reactions require “Complex Ions” to establish Equilibrium. Complex Ions have + or – charges and usually are produced from transition metals. Complex Ions Complex Ions are produced by combining cations and “Ligands”. Ligands are molecules, like NH3, or ions that bond to central atoms to form Complex ions. These complex ions improve solubility. H2O is a common ligand. NH3 are the Ligands The Equilibrium Constant The equilibrium constant is the ratio of products to reactants when the reaction has reached equilibrium. The Equilibrium Constant Keq At equilibrium the concentration of reactants and products can be determined mathematically, only if the temperature is constant. The ratio of concentration of products to reactants is raised to its coefficient as an exponent. aA + bB Keq = [C]c[D]d [A]a[B]b cC + dD [ ] means Molarity Keq is called the “equilibrium constant” expression **Solids and liquids do not show up in the expression. Size of Keq [C]c[D]d = [A]a[B]b At equilibrium: aA + bB cC + dD If Keq is about 1 the concentration of reactants and products is about equal. If Keq is greater than 1 there is a higher concentration of products than reactants at equilibrium. (The higher value K = more products less reactants.) If Keq is less than 1 there is a higher concentration of reactants than products at equilibrium. (The lower the value K = more reactants less products.) Value of Keq Values of Keq can only be determined by experiment. 2 types of math problems involve Keq 1 - given [M] of all reactants and products calculate Keq 2 - given Keq and [M] of all reactants and products but one, solve for its [M] Keq does not indicate a reaction’s rate. Determination of Keq Keq is used to determine product and reactant concentrations at equilibrium. Keq = Products Reactants Changes in Keq *Changes in concentrations DO NOT change Keq adding or removing reactant or product [R] and [P] change but Keq remains the same Changing the pressure of the system DOES NOT change Keq Placing it in a bigger or smaller container Changes in temp DO cause Keq to change [R] gets bigger and [P] gets smaller Or the other way around Determination of Keq H2CO3(aq) + H2O(l) <--> H30+(aq) + HC03-(aq) Determine the value of Keq for the reaction above if H2CO3(aq) = [3.3 x 10-2] H30+(aq) = [1.2 x 10-4] HC03-(aq) = [1.2 x 10-4] Solution Keq = [C]c[D]d [A]a [H3O+][HCO-3] Keq = H2CO3 [1.2 x 10-4] [1.2 x 10-4] -7 4.4 x 10 = Keq = [3.3 x 10-2] Given Keq:Determination of Concentration The reaction; N2O4(g) 2NO2(g) Has a value of K is 1.50. Calculate the concentration of the N2O4(g) present in the system when [NO2] = 0.91 M Solution Keq = 1.5 = Products Reactants [NO2]2 [x] x = 0.55 = [0.91]2 [x] The Solubility product constant...Ksp Three parts to this section: Learn how to write the solubility constant expression Ksp. Learn how to determine solubility from Ksp. Learn how to determine Ksp from solubility. The Solubility Product Constant, Ksp You can dissolve salt in water until the solution becomes saturated. When a solution reaches its saturation point it starts to reform solid reactants. A saturated solution is at “Dynamic Equilibrium”. Many ionic compounds are only “slightly” soluble in water. Example of Using Ksp If you try to dissolve AgI in water you’ll find it’s only “slightly” soluble and you’ll end up with virtually no Ag+ or I- ions. So…how can you compare the solubility of AgI(s) to (Ag+(aq) or I-(aq) )? Easy! Eliminate the reactant and determine the Solubility of the Products. Using Ksp You can compare the solubility of “slightly” soluble salts using Ksp. The solubility product constant, Ksp, is the product of the concentration of the ions produced, each raised to their coefficient as an exponent. The Higher the Ksp, the more soluble the salt will be! The Lower the Ksp, the less soluble the salt will be! Solubility Product Constant Written for the equilibrium of the solution of slightly soluble salts. AaBb(s) ksp = [A+]a [B-]b CaCO3(s) Ca2+(aq ) + CO32-(aq) ksp = [Ca2+] [CO32-] Ag2CO3(s) aA+(aq) + bB-(aq) 2Ag1+(aq) + CO32-(aq) ksp = [Ag+1]2 [CO32-] Solid and liquids do not show up in the ksp expression This will make you or Break You! For every NaF that breaks apart 1 Na+ and 1 Fare formed NaF(s) Na+(aq) + F-(aq) s s Ksp = [Na+][F-] or Ksp = [s][s] or Ksp = s2 For every Ag2CO3 that breaks apart 2 Ag+ and 1 CO32- are formed Ag2CO3(s) 2Ag+(aq) + CO32-(aq) 2s s Ksp = [Ag+]2[CO32-] or Ksp = [2s]2[s] or Ksp = 4s3 Write ksp expression for each of the following: AgI(s) Ag2S(s) PbI2(s) MgCO3(s) Ca3(PO4)2(s) Write ksp expression for each of the following: AgI(s) Ag1+ (aq) + I1- (aq) Ag2S(s) 2Ag1+ (aq) + S2- (aq) PbI2(s) Pb2+ (aq) + 2I1- (aq) MgCO3(s) Mg2+(aq) + CO32-(aq) Ca3(PO4)2(s) 3Ca2+(aq) + 2PO43- (aq) Solubility Product Constant = ksp Ksp shows the solubility of the salt High ksp = more soluble Low ksp = less soluble Table 14-3 pg 508 3 Types of problems 1. Calculating ksp given molar concentrations of the ions. 2. Calculating Molar Solubility given the ksp. 3. Calculating ksp from the Molar Solubility. Values For Ksp At 25 oC Large Ksp = more soluble Small Ksp = less soluble 1. Calculating ksp given molar concentrations of the ions. Calculate the Solubility Product Constant, ksp for Copper(I) Bromide. The concentration of Cu1+ ions in solution is 7.9 x 10-5 mol/L. CuBr(s) Cu1+(aq) + Br1-(aq) Ksp = Cu1+(aq) + Br1-(aq) Ksp = [7.9 x 10-5 mol/L] [7.9 x 10-5 mol/L] = 6.2 x 10-9 2. Calculating Molar Solubility given the ksp. Calculate the solubility of AgI in water if Ksp = 1.8 x 10-10 AgI Ag+ + I[s] [s] Ksp = [Ag+][I-] = s2 1.8 x 10-10 = s2 √ s = 1.8 x 10-10 = 1.3 x 10-5 3. Calculating ksp from the Molar Solubility. It is found that 1.2 x 10-3 mol of lead (II) iodide, PbI2, dissolves in 1.0 L of aqueous solution at 25oC. What is the Ksp at this temperature? PbI2(s) Pb2+(aq) + 2I1-(aq) [s] Ksp = [Pb2+][2I-]2 = 4s3 Ksp = 4(1.2 x 10-3)3 Ksp = 6.9 x 10-9 [2s] LeChatelier’s Principle When an system at equilibrium is disturbed, the system adjusts in a way to relieve the stress…. It will “shift” to the right or to the left. Equililibrium shifts toward the reactants or the products. 3 types of changes (stresses) affect equilibrium D [concentration] [ ] means molarity (mol/L) Increase or decrease D temperature D pressure – for gas systems only Using La Chatelier’s Principle Systems at equilibrium can be forced to completion by changing concentrations, pressure or temperature. Increase in Concentration Increase in the amount of reactants in number of collision between reactants. Forward rx occurs more than the reverse rx…..there’s a shift to the right. Concentration of all r and p change K remains the same An increase in concentration of a substance pushes the equilibrium away from the side of the increase. Same thought process for an increase in the concentration of the products. Decrease in Concentration Decrease in the amount of reactants in number of collision between reactants Reverse rx occurs more than the forward rx….there’s a shift to the left. Decreases in concentration of a substance pulls the equilibrium towards the side of the decrease. Same thought process for a decrease in the concentration of the products. Concentration of all r and p change K remains the same Examples 2NO2(g) N2O4(g) What happens to k if the concentration of NO2 is increased? What happens to concentration of N2O4 if the concentration of NO2 is decreased? What happens to k if the concentration of N2O4 is increased? What happens to concentration of NO2 if the concentration of N2O4 is increased? LeChatelier’s Principle Changing the pressure on the container Increasing the pressure... The reaction will shift toward the side with the fewer moles of gas. Decreasing the pressure... The reaction will shift toward the side with more moles of gas. Changing the pressure will make no difference if there is an equal number of moles of gas on each side. k does NOT change LeChat Pressure N2(g) + 3H2(g) <---> 2NH3(g) Which way will the equilibrium shift if the pressure is increased? Which direction will the equilibrium shift if the container size is increased? Change in Temperature Do you know the forms for endothermic and exothermic reactions??? A + B heat + A + B C + D + heat C +D Heat, energy, or a number of KJ on reactant side = endothermic Heat, energy, or a number of KJ on product side = exothermic Changes in Temperature When a temperature change occurs you write “heat”, or the number of KJ into the equation as a reactant or product. Which side is heat written on? Based on whether reaction is endothermic or exothermic Treat “heat” as a reactant or product in heat, push reaction away from the side “heat” is located in heat, pull reaction towards the side “heat” is located K will get bigger or smaller depending on which direction the equilibrium reaction shifts.. If the shift is to the right, K get bigger.. If the shift is to the left, K gets smaller. Change in Temperature The reaction 2NO2(g) N2O4(g) gives off 57.2 KJ of energy. What happens to the concentration of N2O4 if the equilibrium system is heated? What happens to the value of K? The reaction is exothermic therefore: 2NO2(g) N2O4(g) + heat (57.2 KJ) The decrease in heat will pull reaction to the right. Common Ion Effect The Common Ion Effect explains why there is a reduction in the solubility of a salt due to the presence of a common ion. Example: Think about the solubility of CaF2(s) in NaF(aq)…. The CaF2(s) can’t breakdown as easily because there is already F- ions in solution.