* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter I Overview of Immunology
Duffy antigen system wikipedia , lookup
Complement system wikipedia , lookup
Immunocontraception wikipedia , lookup
DNA vaccination wikipedia , lookup
Lymphopoiesis wikipedia , lookup
Immune system wikipedia , lookup
Molecular mimicry wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Adaptive immune system wikipedia , lookup
Innate immune system wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
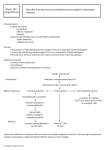
Immunology Concept, Functions and Types of Immunity Concept, Functions and Types of Immunity Introduction Concept and functions of Immunity Types of Immunity Part I Concept and functions of Immunity I Concept of Immunity 1)Tranditional concept----Immunity refers to protection against infectious diseases. 2)Modern concept---- Immunity is a function of which an individual recognizes and excludes antigenic foreign substances. It is normally beneficial,but sometimes,it is injurious. II Concept of Immunology 1) Tranditional concept----anti-infection immunity to different types of pathogenic microorganisms. 2) Modern concept----Immunology is an independent subject about composition,functions of immune system; mechanism of immune response and the disease associated with immunity. III Functions of Immunity Immune defense Immune homeostasis Immune surveillance Functions and Manifestation of Immunity Functions Normal Manifestation Abnormal Manifestation Immune Hypersensitivity Defense Immunodeficiency Immune Homeostasis Immune Surveillance infection Eliminate injured and senile cells Tolerate to self components Anti-infection immune dismodulation Autoimmune disease destroy transformed cells (anti-tumor ) Prevent from persistent infection Tumor or Persistent virus Part II Types of Immunity I. Innate Immunity (or native immunity/ non-specific immunity /congenital immunity) II. Adaptive Immunity (or acquired immunity/specific immunity) I. Innate immunity ( natural immunity/ non-specific immunity ) Innate immunity: Protection against infection that relies on mechanisms that exist before infection,are capable of a rapid response to microbes,and react in essentially the same way to repeated infections. Exists at birth Be the first line of defense against infection Innate immunity 1. Characteristics Exists naturally Non–specific No immune memory (innate immunity can’t be enhanced by the second stimulation of the same antigen) Immune memory: Exposure of the immune system to a foreign antigen enhances its ability to respond again to that antigen. Hereditable No racial difference 2. Composition (1) Barriers Physical barrier : skin and mucosa Chemical barrier: antimicrobial substances in secretion of skin and mucosa Biotic barrier: normal flora existing on the surface of skin and mucosa Anatomic barrier . blood- brain barrier . blood- placental barrier . blood- tymus barrier (2) Humoral factors Complement Lysozyme Interferons(IFN) C-reactive protein (3)Cells participating in innate immunity Phagocyte: endocytosis and phagocytosis mononuclear phagocytes ----Monocytes,Macrophages (M Φ)----PRR Neutrophils Nature killer cells (NK)—KAR/KIR,IgG receptor Dentritic cells(DC) γδ T cells B1 cells Other cells participating in innate immunity Fc Macrophages NK cells Neutrophils Macrophages Macrophages excluding the pathogen Over view What are the main types of white blood cells? Name the two main types of immunity? What are the main distinctions between these two categories? What cells are involved in which aspects of the immune system? Lymphocytes Many types; important in both humoral and cellmediated immunity B-cells produce antibodies (APC cell) T- cells Cytotoxic T cells Helper T cells Memory cells Lymphocytes Natural Killer cells Large granular lymphocytes (not B or T) Kills tumor cells Kills cells infected with certain viruses (intracellular pathogens) Monocytes/Macrophage Phagocytosis and killing of microorganisms Activation of T cells and initiation of immune response Monocyte is a young macrophage in blood There are tissue-specific macrophages APC cells Dendritic Cells Phagocytosis and killing of microorganisms Function as antigen presenting cells (APC) In the blood and tissues – mature and migrate to the lymph nodes Neutrophil Granulocyte Cytoplasmic granules Polymorphonuclear Phagocytosis Short life span (hours) Very important at “clearing” bacterial infections Innate Immunity Eosinophils Kills Ab-coated parasites through degranulation Involved in allergic inflammation A granulocyte Double Lobed nucleus Orange granules contain toxic compounds Basophils Might be “blood Mast cells’ A cell-killing cells Blue granules contain toxic and inflammatory compounds Important in allergic reactions Antigen-presenting cells (APC) Highly specialized Process antigen and display peptide fragments on cell surface Involved in T-cell activation Macrophages, dendritic cells and B-cells Immune system divisions Innate immunity First line of defense Adaptive (acquired) immunity Takes time to develop Humoral immunity (antibody–mediated specific immunity) Cell-mediated immunity (The aspect of the adaptive immune response where antigen-specific T cell have a main role) Active immunity Passive or maternal immunity Injection of Immunoglobulin Absorption of maternal antibodies Innate vs. adaptive immunity Innate immunity First line of defense (present in all individuals at all times) Immediate (0 – 4 hours) Non-specific Does not generate lasting protective immunity Adaptive immune response (late: > 96 hours) Is initiated if innate immune response is not adequate (> 4 days) Antigen-specific immunity Generates lasting protective immunity (e.g. Antibodies, memory T-cells) Immune system cells Innate immunity Granulocytes (i.e. neutrophils) Macrophages Dendritic cells Natural killer (NK) cells Adaptive immunity Lymphocyte B cells T cells Cytotoxic T cells (CTLs) Helper T cells (Th) Memory cells Innate immune system The first line of defense: Penetration of the epithelial surface of the body by microorganism (e.g. bacteria) Engulfment of microorganism by macrophages, neutrophils, and dendritic cells Release of cytokines and chemokines Inflammation Killing by granulocytes Macrophages and neutrophils recognize pathogen by means of cell-surface receptors Example: mannose receptor, CD14 receptor, scavenger receptors, glucan receptor etc. Binding of MØ/neutrophils with pathogen leads to phagocytosis Bound pathogen is surrounded by phagocyte membrane Internalized (phagosome) Killing of pathogen (Phagolysosome*) Oxidative burst (synthesis of hydrogen peroxide (H2O2)or free oxygen radicals) Acidification Antimicrobial peptides (e.g. defensins) * Phagolysosome = lysosome +phagosome Phagocytosis Lipid mediators Mannose receptor Lysosome Phagosome Scavenger receptor LPS receptor (CD14) The macrophage expresses receptors for many bacterial constituents Cytokines Bacteria binding to macrophage receptors initiate the release of cytokines and small lipid mediators of inflammation Phagolysosome Macrophages engulf and digest bacteria to which they bind Phagocytosis (Immunology animation: Janeway) http://www.blink.biz/immunoanimations/# Immune response (IV) 9.1 - Phagocytosis Humoral immune response Cell-surface immunoglobulin receptors (BCR) detect extracellular pathogens Once activated, secrete immunoglobulins as soluble antibodies Antibodies Variable region (2 identical antigen-binding sites) Constant region (determines how antibody disposes of the pathogen once it is bound) V region; At binding Fc region Cell killing – NK cells NK cells do not require prior immunization or activation They attach to ‘target’ cells (ADCC) Cytotoxic granules are released onto surface of cell Effector proteins penetrate cell membrane and induce programmed cell death Inflammation Inflammatory cells migrate into tissue, releasing inflammatory mediators that cause pain Chemokines Cytokines Bacteria trigger macrophages to release cytokines and chemokines Proteins Fluids Vasodilation and increased vascular premeability cause redness, heat, and swelling Cytokines Low molecular weight, soluble proteins that are produced in response to an antigen and function as chemical messengers for regulating the innate and adaptive immune system Innate immune system Macrophages and Dendritic cells Tumor necrosis factor-alpha (TNF-) Interleukin-1 (IL-1) Interleukin-12 (IL-12) Adaptive immune system T-lymphocytes Interleukin-2 (IL-2) Interleukin-4 (IL-4) II.Adaptive immunity ( acquired immunity/specific immunity) Adaptive immunity: The form of immunity that is mediated by T or B lymphocytes and stimulated by exposure to infectious agents. Take effects after innate immune response Be the second line of defense against infection 1.Characteristics Specificity Acquired (set up after birth ) Immune memory (Adaptive immunity can be enhanced by the second stimulation of the same antigen) Transferable Self-limitation 2.Composition T cell : Cell-mediated immunity (CMI) B cell : Humoral immunity(HI) or antibody-mediated immunity 3.The process of immune response in adaptive immunity Recognition of antigens Activation,proliferation and differenciation of T or B lymphocytes Effector phase of immune response ----Elimination of antigens Comparison of Adaptive and Innate Immunity Innate immunity Adaptive immunity Characteristics Exists naturally Acquired by antigen stimulation after birth Responds rapidly in the early develops slowly stage of infection No antigen specificity Has antigen specificity No immune memory Has immune memory Participates in natural defence Participates in specific immune response Cells Neutrophil,Phagocytes,NK cell et al. T cell, B cell, APC Molecules Complement.lysozyme,cytokines et al Antibody,cytokines Immunology Terms • Antigen – Any molecule that binds to immunoglobulin or T cell receptor • Pathogen – Microorganism that can cause disease • Antibody (Ab) – Secreted immunoglobulin • Immunoglobulin (Ig) – Antigen binding molecules of B cells • Vaccination – Deliberate induction of protective immunity to a pathogen • Immunization – The ability to resist infection Types of Immunity • Humoral immunity – Immunity that is mediated by antibodies – Can be transferred by to a non-immune recipient by serum • Cell Mediated Immunity – Immune response in which antigen specific T cells dominate Cells of the Immune system • Many cells of the immune system derived from the bone marrow • Hematopoetic stem cell differentiation The Pathway of Specific Immune Response Step 1 Pathogens eaten by Macrophage Step 2 Displays portion of Pathogen on surface Step 3 Pathogens Helper-T cell recognizes Pathogen Activates B- Cell Activates Cytotoxic T- Cell Memory T-Cell Memory B-Cell Antibodies Kills Infected Cells Cellular Immunity .vs. Antibody Immunity Cellular Immunity • Carried out by T-Cells • Infected cells are killed by Cytotoxic T –Cells. Antibody or Humoral Immunity • Carried out by B-cells • Antibodies are produced and dumped into blood stream. • Antibodies bind to antigens and deactivate them. Immune Response Explained 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Antigen infects cells. Macrophage ingests antigen and displays portion on its surface. Helper T- Cell recognizes antigen on the surface of the macrophage and becomes active. Active Helper T-Cell activates Cytotoxic T-Cells and B-Cells. Cytotoxic T-Cells divide into Active Cytotoxic T-cells and Memory T – Cells. Active Cytotoxic T-Cells kill infected cells. At the same time, B-Cells divide into Plasma Cells and Memory B- Cells. Plasma cells produce antibodies that deactivate pathogen. Memory T and Memory B cells remain in the body to speed up the response if the same antigen reappears. Supressor T-Cells stop the immune response when all antigens have been destroyed. Immune Response Summary Displays copy of antigen on surface of cell Antigen Macrophage Antibody Immunity Helper T - Cell Cellular Immunity Active Cytotoxic T-Cell Kills Infected Cells Memory T- Cell Active B - Cell Plasma Cell Antibodies Deactivates Antigens Memory B-Cell Primary .vs. Secondary Immune Response • Primary Immune Response – This is a response to an invader the First time the invader infects the body. • No measurable immune response for first few days. • Next 10 – 15 days antibody production grows steadily • Secondary Immune Response – A more rapid response to an invader the 2nd time it invades the body. • Antibody production increases dramatically and in a much shorter time period.. Primary .vs. Secondary Immune Response Passive .vs. Active Immunity 1. Active Immunity This is immunity where the body is “actively” producing antibodies to fight infection. Ex: You have a throat infection and you are actively creating antibodies to fight it. Vaccination: An injection of a weakened strain of an infectious microbe (pathogen) that causes the body to undergo active immunity (produce antibodies). 2. Passive Immunity This is immunity where antibodies are given to a person from the blood of another person or animal. This immunity only lasts for a short period of time. ex: Breastfeeding mothers pass antibodies to their children through the milk. Antibodies – structure, classes and function Objectives By the end of the session you should be able to: • illustrate and describe the basic structure of antibodies in terms of heavy and light chains • explain the structure in relation to function of the molecules • define the terms ‘isotype’, ‘allotype’ and ‘idiotype’ • describe the structure of each of the five main classes of antibody • describe the location and function of each class of antibody • define the terms ‘affinity’ and avidity’ of antibodies The fragment antigen binding (Fab fragment) is a region on an antibody which binds to antigens. It is composed of one constant and one variable domain of each of the heavy and the light chain. These domains shape the paratope—the antigen binding site—at the amino terminal end of the monomer. The two variable domains bind the epitope on their specific antigens. An epitope, also know as antigenic determinant, is the part of a macromolecule that is recognized by the immune system, specifically by antibodies, B cells, or T cells. The part of an antibody that recognizes the epitope is called a paratope. Although epitopes are usually thought to be derived from nonself proteins, sequences derived from the host that can be recognized are also classified as epitopes. Antibody heterogeneity Isotypes Two light chain isotypes – k and l : Five heavy chain isotypes - m,g,d, and e Allotypes Genetic markers- (rather like blood groups and usually the result of minor amino acid differences) on immunoglobulins that segregated within the species and inherited in Mendelian fashion eg. Km (on k light chains) and Gm (on IgG heavy chains) Idiotypes Each antibody varies in its amino acid sequences in the variable regions of both heavy and light chains to create different specificities Ig class Heavy chain Light chain IgM m (mu) k (kappa) or l (lambda) IgG g (gamma) k (kappa) or l (lambda) IgA (alpha) k (kappa) or l (lambda) IgE e (epsilon) k (kappa) or l (lambda) IgD d (delta) k (kappa) or l (lambda) Summary of different classes and subclasses of antibodies Physical forces holding antibodies and antigen together Affinity and avidity of antibodies Affinity The tightness with which the antigen binding site attaches to an antigen determinant (epitope) Avidity The tightness of binding when several antigen binding sites attach to several antigenic determinants Immunoglobulin Element of adaptive immune mechanism Better known as antibody It recognize the foreign objects How they work (examples) Animation1 Animation2 Structure of immunoglobulin Two identical heavy (H) chains and two identical light (L) chains combine to form this Y-shaped antibody molecule Disulfide bonds Bonds between two amino acids result of the SH (sulfhydral) group of one amino acid covalently bonding to the SH group of another amino acid Stronger than hydrogen bonds Eg. Hair proteins are held together by disulfide bonds Heavy chains The heavy chains each have four domains Variable domains (VH) Constant domains (CH1,2,3) Light chain The light chains are constructed of two domains Variable (VL) Constant (CL) Structure of immunoglobulin The fragment antigen binding (Fab fragment) The fragment crystallizable region (Fc region) Antibodies bind to antigens by reversible, noncovalent interactions, including hydrogen bonds and charge interactions Antigen binding some pictures Antigen binding some pictures Structure of immunoglobulin Functional consequences: (VH) and (VL) are positioned to stereochemically react with antigen The stem is good for mediate effector functions Hinge Two disulfide bonds in the hinge region unite the two heavy chains The hinge allows the two antigen-binding Fab regions of each antibody molecule to move Conclusion Changes in the antigen binding site conformation are vital for antigen recognition Herewith the variety of antibody conformation is vital for our health Humoral Immune Response Cellular Events Antigen is “processed” by T lymphocytes and macrophages. Possess special receptors on surface. Termed “antigen presenter cell” APC. Antigen presented to B cell Cellular Immune Response Important in defending against: fungi, parasites, bacteria. Responsible for hypersensitivity, transplant rejection, tumor surveillance. Thymus derived (T) lymphocytes Cell Mediated Reaction Helper T cells – turn on immune response Suppressor T cells – turn off immune response Cytotoxic T cells directly attack antigen Cell Mediated Immunity Basic Antibody Structure Figure 21.12a, b Antibody Structure Antibodies responding to different antigens have different V regions but the C region is the same for all antibodies in a given class C regions form the stem of the Y-shaped antibody and: Determine the class of the antibody Serve common functions in all antibodies Dictate the cells and chemicals that the antibody can bind to Determine how the antibody class will function in elimination of antigens Mechanisms of Antibody Diversity Plasma cells make over a billion different types of antibodies Each cell, however, only contains 100,000 genes that code for these polypeptides To code for this many antibodies, somatic recombination takes place Gene segments are shuffled and combined in different ways by each B cell as it becomes immunocompetent Information of the newly assembled genes is expressed as B cell receptors and as antibodies Antibody Diversity Random mixing of gene segments makes unique antibody genes that: Code for H and L chains Account for part of the variability in antibodies V gene segments, called hypervariable regions, mutate and increase antibody variation Plasma cells can switch H chains, making two or more classes with the same V region Antibody Targets Antibodies themselves do not destroy antigen; they inactivate and tag it for destruction All antibodies form an antigen-antibody (immune) complex Defensive mechanisms used by antibodies are neutralization, agglutination, precipitation, and complement fixation Complement Fixation and Activation Complement fixation is the main mechanism used against cellular antigens Antibodies bound to cells change shape and expose complement binding sites This triggers complement fixation and cell lysis Complement activation: Enhances the inflammatory response Uses a positive feedback cycle to promote phagocytosis Enlists more and more defensive elements Other Mechanisms of Antibody Action Neutralization – antibodies bind to and block specific sites on viruses or exotoxins, thus preventing these antigens from binding to receptors on tissue cells Other Mechanisms of Antibody Action Agglutination – antibodies bind the same determinant on more than one antigen Makes antigen-antibody complexes that are crosslinked into large lattices Cell-bound antigens are cross-linked, causing clumping (agglutination) Precipitation – soluble molecules are cross-linked into large insoluble complexes Mechanisms of Antibody Action Figure 21.13 Cell-Mediated Immune Response Since antibodies are useless against intracellular antigens, cell-mediated immunity is needed Two major populations of T cells mediate cellular immunity CD4 cells (T4 cells) are primarily helper T cells (TH) CD8 cells (T8 cells) are cytotoxic T cells (TC) that destroy cells harboring foreign antigens Other types of T cells are: Suppressor T cells (TS) Memory T cells Major Types of T Cells Figure 21.14 Importance of Humoral Response Soluble antibodies The simplest ammunition of the immune response Interact in extracellular environments such as body secretions, tissue fluid, blood, and lymph The Humoral Immune Response (HIR) is the aspect of immunity that is mediated by secreted antibodies (as opposed to cell-mediated immunity which involves T lymphocytes) produced in the cells of the B lymphocyte lineage (B cell). Secreted antibodies bind to antigens on the surfaces of invading microbes (such as viruses or bacteria), which flags them for destruction.[1] Humoral immunity is called as such, because it involves substances found in the humours, or body fluids. Importance of Cellular Response T cells recognize and respond only to processed fragments of antigen displayed on the surface of body cells T cells are best suited for cell-to-cell interactions, and target: Cells infected with viruses, bacteria, or intracellular parasites Abnormal or cancerous cells Cells of infused or transplanted foreign tissue Antigen Recognition and MHC Restriction Immunocompetent T cells are activated when the V regions of their surface receptors bind to a recognized antigen T cells must simultaneously recognize: Nonself (the antigen) Self (a MHC protein of a body cell) The major histocompatibility complex (MHC) is a large genomic region or gene family found in most vertebrates. It is the most gene-dense region of the mammalian genome and plays an important role in the immune system, autoimmunity, and reproductive success. The proteins encoded by the MHC are expressed on the surface of cells in all jawed vertebrates, and display both self antigens (peptide fragments from the cell itself) and nonself antigens (e.g. fragments of invading microorganisms) to a type of white blood cell called a T cell that has the capacity to kill or co-ordinate the killing of pathogens, infected or malfunctioning cells. MHC Proteins Both types of MHC proteins are important to T cell activation Class I MHC proteins Always recognized by CD8 T cells Display peptides from endogenous antigens Class I MHC Proteins Endogenous antigens are: Degraded by proteases and enter the endoplasmic reticulum Transported via TAP (transporter associated with antigen processing) Loaded onto class I MHC molecules Displayed on the cell surface in association with a class I MHC molecule Class I MHC Proteins Figure 21.15a Class II MHC Proteins Class II MHC proteins are found only on mature B cells, some T cells, and antigen-presenting cells A phagosome containing pathogens (with exogenous antigens) merges with a lysosome Invariant protein prevents class II MHC proteins from binding to peptides in the endoplasmic reticulum Class II MHC Proteins Class II MHC proteins migrate into the phagosomes where the antigen is degraded and the invariant chain is removed for peptide loading Loaded Class II MHC molecules then migrate to the cell membrane and display antigenic peptide for recognition by CD4 cells Class II MHC Proteins Figure 21.15b Antigen Recognition Provides the key for the immune system to recognize the presence of intracellular microorganisms MHC proteins are ignored by T cells if they are complexed with self protein fragments Antigen Recognition If MHC proteins are complexed with endogenous or exogenous antigenic peptides, they: Indicate the presence of intracellular infectious microorganisms Act as antigen holders Form the self part of the self-antiself complexes recognized by T cells T Cell Activation: Step One – Antigen Binding T cell antigen receptors (TCRs): Bind to an antigen-MHC protein complex Have variable and constant regions consisting of two chains (alpha and beta) T Cell Activation: Step One – Antigen Binding MHC restriction – TH and TC bind to different classes of MHC proteins TH cells bind to antigen linked to class II MHC proteins Mobile APCs (Langerhans’ cells) quickly alert the body to the presence of antigen by migrating to the lymph nodes and presenting antigen T Cell Activation: Step One – Antigen Binding TC cells are activated by antigen fragments complexed with class I MHC proteins APCs produce co-stimulatory molecules that are required for TC activation TCR that acts to recognize the self-antiself complex is linked to multiple intracellular signaling pathways Other T cell surface proteins are involved in antigen binding (e.g., CD4 and CD8 help maintain coupling during antigen recognition) T Cell Activation: Step One – Antigen Binding Figure 21.16 T Cell Activation: Step Two – Co-stimulation Before a T cell can undergo clonal expansion, it must recognize one or more co-stimulatory signals This recognition may require binding to other surface receptors on an APC Macrophages produce surface B7 proteins when nonspecific defenses are mobilized B7 binding with the CD28 receptor on the surface of T cells is a crucial co-stimulatory signal Other co-stimulatory signals include cytokines and interleukin 1 and 2 T Cell Activation: Step Two – Co-stimulation Depending on receptor type, co-stimulators can cause T cells to complete their activation or abort activation Without co-stimulation, T cells: Become tolerant to that antigen Are unable to divide Do not secrete cytokines T Cell Activation: Step Two – Co-stimulation T cells that are activated: Enlarge, proliferate, and form clones Differentiate and perform functions according to their T cell class T Cell Activation: Step Two – Co-stimulation Primary T cell response peaks within a week after signal exposure T cells then undergo apoptosis between days 7 and 30 Effector activity wanes as the amount of antigen declines The disposal of activated effector cells is a protective mechanism for the body Memory T cells remain and mediate secondary responses to the same antigen Cytokines Mediators involved in cellular immunity, including hormonelike glycoproteins released by activated T cells and macrophages Some are co-stimulators of T cells and T cell proliferation Interleukin 1 (IL-1) released by macrophages costimulates bound T cells to: Release interleukin 2 (IL-2) Synthesize more IL-2 receptors Cytokines IL-2 is a key growth factor, which sets up a positive feedback cycle that encourages activated T cells to divide It is used therapeutically to enhance the body’s defenses against cancer Other cytokines amplify and regulate immune and nonspecific responses Cytokines Examples include: Perforin and lymphotoxin – cell toxins Gamma interferon – enhances the killing power of macrophages Inflammatory factors Helper T Cells (TH) Regulatory cells that play a central role in the immune response Once primed by APC presentation of antigen, they: Chemically or directly stimulate proliferation of other T cells Stimulate B cells that have already become bound to antigen Without TH, there is no immune response Helper T Cells (TH) Figure 21.17a Helper T Cell TH cells interact directly with B cells that have antigen fragments on their surfaces bound to MHC II receptors TH cells stimulate B cells to divide more rapidly and begin antibody formation B cells may be activated without TH cells by binding to T cell–independent antigens Most antigens, however, require TH co-stimulation to activate B cells Cytokines released by TH amplify nonspecific defenses Helper T Cells Figure 21.17b Cytotoxic T Cell (Tc) TC cells, or killer T cells, are the only T cells that can directly attack and kill other cells They circulate throughout the body in search of body cells that display the antigen to which they have been sensitized Their targets include: Virus-infected cells Cells with intracellular bacteria or parasites Cancer cells Foreign cells from blood transfusions or transplants Cytotoxic T Cells Bind to self-antiself complexes on all body cells Infected or abnormal cells can be destroyed as long as appropriate antigen and co-stimulatory stimuli (e.g., IL-2) are present Natural killer cells activate their killing machinery when they bind to MICA receptor MICA receptor – MHC-related cell surface protein in cancer cells, virus-infected cells, and cells of transplanted organs Mechanisms of Tc Action In some cases, TC cells: Bind to the target cell and release perforin into its membrane In the presence of Ca2+ perforin causes cell lysis by creating transmembrane pores Other TC cells induce cell death by: Secreting lymphotoxin, which fragments the target cell’s DNA Secreting gamma interferon, which stimulates phagocytosis by macrophages Mechanisms of Tc Action Figure 21.18a, b Other T Cells Suppressor T cells (TS) – regulatory cells that release cytokines, which suppress the activity of both T cells and B cells Gamma delta T cells (Tgd) – 10% of all T cells found in the intestines that are triggered by binding to MICA receptors