* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Background on Protein and Interactions
Extracellular matrix wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Endomembrane system wikipedia , lookup
Signal transduction wikipedia , lookup
Protein moonlighting wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Green fluorescent protein wikipedia , lookup
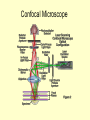
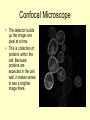
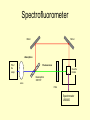
Study of Protein Association by Fluorescence-based Methods Kristin Michalski UWM RET Intern In association with Professor Vali Raicu Role of Proteins • Proteins are complex molecules that play many critical roles in the body. • They are required for the structure, function, and regulation of the body’s tissues and organs. • Proteins are made up of many of smaller units called amino acids, which are attached to one another in long chains. Role of Proteins • Proteins are the catalysts of biological functions. • Also, proteins never act alone. For example, a growth hormone is a messenger protein made by the pituitary gland. It regulates cell growth by binding to a protein called a growth hormone receptor. Why is it important to study protein associations? • Protein associations are known to occur, but what mechanism causes proteins to combine is not always understood. • These associations occur at a distance of ~1 nm. Currently, there is no microscope that can view these small interactions. • How do we see these protein associations? Fluorescent Tagging • Because these structures are so small, proteins of interest are tagged with fluorescent markers in order to study the proteins themselves and their interactions in living cells based on the detection of light from fluorescent tags. What is fluorescence? • Fluorescence is a process in which a molecule is excited by the absorption of light. As the molecule moves to ground state, light is emitted at lower energy (lower wavelength). • This process is known as red-shifting. Energy lost absorb high energy photon Emits lower energy photon Ground State Fluorescence is a process by which molecules absorb light of certain energy or wavelength (e.g., blue), lose some energy internally, and emit a less energetic photon (e.g., green). This emission of light is dependent on the chemical structure of the molecule. Process of tagging proteins • Specific plasmids are chosen and inserted into the cell to knock out the chromosome and produce an altered chromosome. Gene STE 2 Ste 2P Gene responsible for expression of GFP Process of tagging proteins • The letters in the genome that express a specific amino acid on a protein are found. • This amino acid is cut out of the genome using enzymes and replaced with the portion of the gene that expresses GFP (or YFP). • The cells are then left to reproduce with this new coding. Gene Gene STE 2 Ste 2P Gene responsible for expression of GFP STE 2 Ste 2P Fluorescent Emissions • Because we can not view protein associations, we must use a useful tool that can measure the fluorescence of our tagged proteins. • The tool that we use is Förster resonance energy transfer or fluorescence resonance energy transfer (FRET), a process in which an excitation of a fluorescent molecule is transferred non-radiatively to a nearby molecule. How do we see fluorescence? • There are two types of light microscopy, Epifluorescence and Confocal microscopy, used to detect the light emitted by fluorescent molecules tagged to a selected cell structure of interest. • In this project, the epifluorescence microscope was used to locate the cells of interest and the confocal was used for the data collection. Transmission Image • Here is a transmission image of the cells expressing green fluorescence (fluorescence not seen here). • A transmission image was recorded before and after each data collection to locate any shifting of the cells. How does a Confocal Microscope work? • The laser light reflects off of a dichroic mirror and then hits two mirrors which are mounted on motors; these mirrors scan the laser across the sample. • When laser light interacts with fluorophores in the specimen, they emit light which is reflected back through the beam path by the same mirrors that are used to scan the excitation light from the laser. Confocal Microscope • In a confocal laser scanning microscope a laser is used to provide an excitation light in order to get very high intensities that will penetrate into specimens. • One of the major benefits of using the confocal microscope is it allows for optical sectioning. Confocal Microscope Confocal Microscope • The emitted light passes through the dichroic mirror and is focused onto a pinhole. • Because the pinhole diameter is small, very little of the out of focus light emitted from the fluorophores located above and below the focal plane passes through the pinhole aperture. • The light that passes through the pinhole is measured by a detector, a photomultiplier tube. Confocal Microscope • The detector builds up the image one pixel at a time. • This is collection of proteins within the cell. Because proteins are expected in the cell wall, it makes sense to see a brighter image there. Confocal Microscope • In the quantitative analysis, the area of each cell is found. The intensity within that area is measured, determining the total amount of the fluorescence in each cell. FRET Studies 1. FRET can determine the energy-transfer efficiency providing a measure the intermolecular distances inside a protein complex. 2. FRET can determine quantities of the molecular interactions, the ratio of the interacting protein complexes, and finally the size of the oligomers (associated proteins) formed. FRET • A donor protein (GFP) can “lose” energy if an accepter is near. • An accepter protein (YFP) can “gain” energy if a donor is near. GFP YFP FRET • Some donor proteins (GFP) will associate with acceptor proteins (YFP) and transfer energy non-radiantively causing the intensity of the donor fluorescence to be reduced and the accepter’s fluorescence intensity to increase. 476 nm 476 nm FRET • Because the acceptor protein naturally emits fluorescence, photobleaching is used to “bleach” the acceptor. • Once the YFP is bleached, FRET is used as a tool to measure the amount of energy that is emitted from GFP. 514 nm YFP YFP FRET Quantitative Results • Spectral deconvolution of composite fluorescence spectra of cells expressing Ste2p-GFP and STE2p-YFP FRET Quantitative Results • The intensity of the FRET signal will be dependent on the concentration of donor and acceptors present. • In FRET studies, fluorescence intensity of samples is measured at several wavelengths. Studies of fluorescence on a collection of cells • The first part of the presentation explained how fluorescence in measured on individual cells but it would be useful to study a collection of cells. • This will be done using a duel Absorption and Emission Spectrometer. Spectrofluorometers using cell suspended in liquid • Using cells that are suspended in a liquid medium, two things can be obtained. • First, through the use of a absorption spectrometer, the number of cells in the suspension can be measured. • Second, with the use of the emission spectrometer, fluorescence of the cells can be measured. • Because of the dynamic nature of living cells, absorption and emission of cell expression should obtained within minutes of each other. Spectrofluorometer Mirror Mirror Absorption PX-2 Xe Lamp Fluorescence Cuvette Holder Beamsplitter 30R/70T Lens Filter Spectrometer USB200 OOIBase32 Software