* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Changing Climate Changing Understanding
Global warming hiatus wikipedia , lookup
Climate change and agriculture wikipedia , lookup
Citizens' Climate Lobby wikipedia , lookup
Climate sensitivity wikipedia , lookup
Surveys of scientists' views on climate change wikipedia , lookup
Climate change, industry and society wikipedia , lookup
Effects of global warming on humans wikipedia , lookup
Public opinion on global warming wikipedia , lookup
Climate engineering wikipedia , lookup
Climate-friendly gardening wikipedia , lookup
Climate change mitigation wikipedia , lookup
Scientific opinion on climate change wikipedia , lookup
Economics of global warming wikipedia , lookup
Fred Singer wikipedia , lookup
General circulation model wikipedia , lookup
Climate change and poverty wikipedia , lookup
Low-carbon economy wikipedia , lookup
Climate change in Canada wikipedia , lookup
Physical impacts of climate change wikipedia , lookup
Instrumental temperature record wikipedia , lookup
Climate change in the United States wikipedia , lookup
Carbon Pollution Reduction Scheme wikipedia , lookup
United Nations Climate Change conference wikipedia , lookup
Mitigation of global warming in Australia wikipedia , lookup
Global Energy and Water Cycle Experiment wikipedia , lookup
Global warming wikipedia , lookup
Attribution of recent climate change wikipedia , lookup
Years of Living Dangerously wikipedia , lookup
Solar radiation management wikipedia , lookup
Business action on climate change wikipedia , lookup
Politics of global warming wikipedia , lookup
Climate change feedback wikipedia , lookup
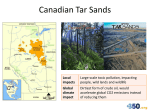
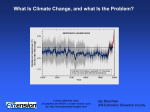
Changing Climate Changing Understanding … a drama in six acts Peter Mahaffy Brian Martin Alberta Environment December 8, 2010 ‘An understanding of chemistry is essential as the basis for medicine and public health, in addressing challenges such as global climate change, in providing sustainable sources of clean water, food and energy, and in maintaining a wholesome environment for the well-being of all people. UN International Year of Chemistry (2011) Visualizing and Understanding the Science of Climate Change IUPAC Project 2008-043-1-050 The Prologue… Act One – Carbon Today International Geophysical Year 1957 Scripps Institute of Oceanography http://rammb.cira.colostate.edu/dev/hillger/IGY.htm C02 (ppm) Perhaps one of the most important graphs in history! Date Where are you? Your grandchildren? IGY 1957 IYC 2011 CDIAC, 2010 Go to NASA/Oak Ridge Video Act Two – View From “Deep” History History shows quite clearly that climate change is natural and inevitable – there is nothing different about our current observed change is there? – Doesn’t this show that it is mostly “natural”? IPCC 2007, Climate Change: The Physical Science Basis, Working Group 1, 4th Assessment Report I don’t get it! We weren't around 100 000 years ago but there was clearly big climate change happening then – again isn’t this arguing that’s it’s natural? Act Three – Carbon Dioxide and How to Live a Balanced Life Learning to see our planet in new ways NASA radiometer images Wavelength Distribution of Solar Radiation C. Middlecamp et. al, Chemistry in Context, McGraw Hill Not to scale - IR intensity is MUCH less than visible! Greenhouse gases – A New Idea? 1827: Fourier – theorized that greenhouse 1896: Arrhenius - proposed that changes in gases warm the planet atmospheric CO2 concentrations due to volcanic eruptions and the combustion of coal can cause climate change 1938: Callendar – first noted that human emissions of CO2 may add significantly to natural concentrations in the atmosphere 1957: Revelle et al. – first warned that human emissions have started a global scale geophysical experiment and initiated an atmospheric CO2 concentration monitoring program Environment Canada Radiation balance How can greenhouse gases heat the atmosphere? But isn’t the absorption of CO2 saturated in the atmosphere? How can increasing the CO2 concentration affect temperature? Besides – isn’t water vapour more important anyway? How significant are small changes in Earth’s radiation balance? Act Four – Spectral Windows, Methane Clathrate Hydrates and Other Sleeping Giants Key Players - A Closer Look Carbon Dioxide Methane Nitrous Oxide CFCs and HCFCs Water ! Solar activity Aerosols CO2, CH4, N2O – over 2000 years Mahaffy, et. al., Chemistry: Human Activity, Chemical Reactivity, 2010, Nelson, p 86 Adapted from IPCC, 2007, 4th Assessment Report Greenhouse Gases to Focus on? The effect of a ‘greenhouse’ gas on climate depends on (a) its concentration, (b) how strongly and where in the infrared region it absorbs energy and (c) its atmospheric lifetime. Global warming potential, the ability of a gas to cause changes to earth’s climate relative to CO2, which is arbitrarily assigned a value of 1. Some Key Players Gas Global Warming Potential Atmospheric Lifetime (years) Time Horizon 20 years 100 years 500 years Variable 1 1 1 Methane (CH4) 12 72 25 7.6 CFC-12 (CF2Cl2) 100 11,000 10,900 5200 Nitrous Oxide (N2O) 114 289 298 153 Perfluoropropane (C3F8) 2600 6,310 8,830 12,500 Carbon dioxide (CO2) IR Spectroscopy helps us sort out which atmospheric gases to pay the most attention to! Clathrates (methane hydrates) Ken Jordan, U Pittsburgh Methane Clathrate Hydrates: Positive Feedback of Unknown Impact But History may Have Some Lessons Northern regions of Russia/Canada contain peatland-permfrost with huge stores of methane (CH4), 25X the GWP of CO2 over 100 years. Courtesy of Geoff Strong 32 N2(g) + 3H2(g) 2NH3(g) As atmospheric concentrations of CO2 have increased by 30%, the deposition into the atmosphere of reactive nitrogen species has gone up by 300%. Human activity is now responsible for about one-half of the entire global cycle of nitrogen Reactive Nitrogen Production Nitrogen atom efficiency N f ertilizer produced N f ertilizer applied 100 94 -6 N f ertilizer produced 47 - 47 N f ertilizer applied 100 94 -6 N in crop - 47 N harvested 31 26 -5 - 16 N in crop N in f eed 47 31 - 16 N in f ood 14 vegetarian diet - 12 N in store 7 - 24 N consumed N consumed 4 -3 J. N. Galloway & E. B. Cowling, 31, Ambio, March 2002 carnivorous diet Terraform Mars with Super Greenhouse Gases? Proceedings of Natl Academy of Sciences, Feb 2001 IPCC 2007 Act Five – Terra Less Firma and Pteropods Our Fragile Oceans Positive feedback loops and arctic ice changes Hoegh-Guldberg, O., Bruno, J. Science, 328, p 1523, 18 June 2010 The physics and chemistry of adding an acid to the ocean are so well understood, so inexorable, that there cannot be an iota of doubt—gigatons of acid are lowering the pH of the world ocean, humans are totally responsible, and the more carbon dioxide we emit, the worse it’s going to get. Ocean pH is lower than it’s been for 20 million years Models predict drop from a pre-industrial pH of 8.2 to about 7.8 by the end of this century. That would increase the surface ocean’s acidity by about 150% on average. Doney, S. Science, 328 18 June 2010 Predicted 2100 pH 7.8 Pre-industrial pH 8.2 Mahaffy, et. al., Chemistry: Human Activity, Chemical Reactivity, 2010, Nelson, p 552 Sea Surface pH change since Industrial Revolution Sea Surface CO32- change since Industrial Revolution Hoegh-Guldberg, O., Bruno, J. Science, 328, p 1523, 18 June 2010 Act Six – The Future? http://www.gapminder.org/world/#;example=6 Fossil Fuel Emissions: Actual vs. IPCC Scenarios 10 International Energy Agency -1 Fossil Fuel Emission (GtC y ) Carbon Dioxide Information Analysis Center 9 Averages A1B 8 A1FI A1T A2 7 B1 B2 6 Full range of IPCC individual scenarios 5 1990 1995 2000 2005 2010 2015 Raupach et al. 2007, PNAS, updated; Le Quéré et al. 2009, Nature Geoscience; International Monetary Fund 2009 “What’s the use of having developed a science well enough to make predictions if, in the end, all we’re willing to do is stand around and wait for them to come true?” F. Sherwood Roland Nobel Laureate Chemistry Ozone Depletion Thank You! Any Questions? Mass of the Atmosphere Basic Facts: •Atmospheric pressure 100 kPa •1 Pa = 1N/m2 •Radius of Earth 6.38 X 106m Every square m of the Earth’s surface supports 104 kg of air SA 4 R 2 What mass of a column of air 1m2 at the base will exert a force of 100 kN ? Mass 4 R 2 (104 kg / m 2 ) 4 (6.38 106 m )2 (10 4 kg / m 2 ) 5 1018 kg Ans: mg = 100 000 N m = 104 kg How many molecules are there in the atmosphere? Basic Facts: •Atmospheric is mostly N2 and O2 •“molar mass” approximately 30 g/mol •Mass of atmosphere 5 X 1018 kg Number of mols mass of atmosphere molar mass 5 1021 g 1.7 1020 mol 30 g / mol How Much CO2 in ppm Does a Barrel Basic Facts: of Oil Produce? 1 barrel releases 425 kg of CO2; in moles this is 425 kg 104 mol 0.044 kg / mol •Carbon-based fuel releases 3.15 times its mass in CO2 •Mass of a barrel of oil is about 135 kg or •1 barrel releases 425 kg CO2 •CO2 has a molar mass of 44g/mol Since the atmosphere contains 1.7 X 1020 mol one barrel will release 104 17 6 10 1.7 1020 This is the fraction of CO2 relative to the entire atmosphere – multiply by 1 million to get the parts-per-million or ppm. So, 1 barrel releases an additional 6 1011 ppm Is the observed increase in CO2 “natural” or Basic Facts: … (30 109 bbl/a)(6 1011 ppm/bbl ) 1.8 ppm/a •1 barrel of oil releases 6 X 10-11 ppm of new CO2 into the atmosphere •30 billion barrels of oil are consumed annually Slope = 1.8 ppm/a 46 ppm 25 a Basic Facts: A Bit Closer to home… what is the annual Carbon footprint of the Alberta Oil Sands in ppm? •Fort Mac produces 1.5 million barrels of oil per day •Annual Carbon footprint is 40 million tonnes of carbon dioxide •1 barrel of oil releases 6 X 10-11 ppm of new CO2 into the atmosphere 40 Mt CO2 9.4 107 bbl 425 kg / bbl (9.4 107 bbl/a)(6 1011 ppm/bbl ) 0.006 ppm/a …but – that’s not the end of the story! Components of Fossil Fuel Emissions Le Quéré et al. 2009, Nature Geoscience How about Coal-Generated Power? Basic Facts: •The Sundance Coal-fired Power Generation Plant on Lake Wabamum produces 2126 MWH •Annual Carbon footprint is 17.5 million tonnes of carbon dioxide The Sundance plant produces roughly 7.5/40 times as much CO2 as The Alberta Oil Sands In other words – Sundance adds (17.5 / 40)(0.006 ppm/a)= 0.003 ppm/a (or about “half-a-Fort Mac”) Let’s Re-run the Numbers… Basic Facts: •CO2 sources by percent: •Coal 40% •Oil 36% •Natural Gas 20% •Other 4% If the burning of oil accounts for only 36% of the total CO2 loading then the total (anthropogenic) loading is … 1.8 ppm / a 5 ppm / a 0.36 So – where is the rest going? How much energy do we receive from the Sun? Basic Facts: •Average Solar Constant is 1.366 kW/m² Solar constant is “spread” over the top of atmosphere of the Earth so each square metre receives R2e (1.336 kW / m2 ) 4 R 2e 334 W / m2 Thank Goodness for Greenhouse Gases! Basic Facts: •Earth’s albedo is about 0.35 •Solar constant So is about 1366 W/m2 • = 5.67 X 10-8 Wm-2K-4 Energy Absorbed from Sun Esun So (1 ) R 2e Teff Energy emitted by Earth 4 EEarth 4 R 2e Teff 4 So (1 ) 4 Earth’s effective surface temperature should be – 15 C! Watt – Does it Matter? At 500 ppm CO2, Earth radiates 277.702 W/m2 back into space At the current 380 ppm CO2, Earth radiates 279.146 W/m2 back into space This is “only” a difference of 1.44 W/m2 Ways to see the “difference” Basic Facts: W/m2 over This is 1.44 the surface of the Earth so multiplying by the surface area of the earth gives us a better picture: •Surface area of Earth is 5.1 X 1014 m2 •Mass of hydrosphere is 1.4 X 1021 kg •This represents is 7.4 X 1014 W of “extra” power that is being absorbed by the environment •What if you “paid” the bill – in 1 hour this would be 2.7 X 1015 kWh; @ $0.05/kWh you are on the hook for $1.3 X 1014 or 130 trillion dollars – per hour! •Over 1 year this difference is 2.3 X 1022 Joules of extra energy •In one century this could heat the oceans by U thermal mc T 2.3 1024 J T (1.4 1021 kg)(4186 0.4 C J kgC o ) How Much Will a Change in CO2 Concentration Affect Global Temperature Basic Facts: •CO2 has increased from 318 ppm to 390 ppm in the last 50 years •Temperature increase is 0.5 C during this time T2 x T ln(2) ln( o ) First-order Temp vs ρCO2 Relationship ln( T T2 x T2 x (0.5 C ) o ) ln(2) ln(2) 1.70 C 390 ln( ) 318 The CO2 doubling temperature is 1.70 C (low if feedback effects are considered) How will this affect future temperature? If CO2 concentration grows from 390 ppm to 500 ppm: T 1.70 C ln(500 ) 390 0.61 C ln(2) (This is a gross estimate – much higher in polar regions) Contrasting “Historical” Changes with the Change we now observe • Go to the climate trends applet and select two different data regions: – Past region showing significant change – Last 100 years of data • Compare both Temperature and CO2 trends Why CO2 “Saturation” is a Red Herring… Shortly after Arrhenius suggested that increasing CO2 could raise atmospheric temperature, Angstrom measured the absorption in a gas cell as a function of increasing CO2 concentration and found no significant Increase – Conclusion – CO2 was saturated and increasing CO2 cannot block more IR. This is wrong for a number of fundamental reasons Some Key Points… • The atmosphere is NOT a gas cell! • At low concentration photons can escape to space from lower in the atmosphere • As CO2 concentration increases the optical depth increases and photon escape occurs at higher locations in the atmosphere • Photon “absorption” is really a process of absorption – scattering-emission - the Equation of Transfer describes this process • There is never a point at which adding more CO2 does not decrease the outward IR flux Some Simulations using MODTRAN3 R Animation showing CO2 absorption as concentration increases from 1 ppm to 1000 ppm in 50 ppm steps Graphs show outward flux at 70 km in atmosphere A Curve of Growth… That’s all fine BUT isn’t Water Vapour more important anyway? Climate Forcings – The Milankovitch Hypothesis • Variations in: – Orbital eccentricity – Obliquity These are very large forcings acting over “astronomical” time scales – ie “deep time” These forcings produce large changes in insolation •Graphic depicts variation over 500 thousand years – 250 ka in the past; 250 ka in the future Global average RF estimates, 2005 IPCC 2007 4th AR