* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Biochemistry - Fort Thomas Independent Schools
Survey
Document related concepts
Vectors in gene therapy wikipedia , lookup
Isotopic labeling wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Photosynthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
Genetic code wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Proteolysis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Transcript
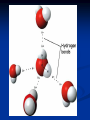
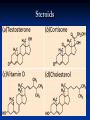
Biochemistry Chapter 3 Water polar compound one end is slightly negative while the other is slightly positive results from unequal sharing of electrons molecule itself is neutral Water hydrogen bonding attraction that holds different water molecules together the slightly negative O is attracted to the slightly positive H of another molecule Water cohesion attractive force of particles of same kind (stick together) adhesion attractive forces between 2 particles of different substances (stick to other things) Water capillary action the movement of water up through small tubes, against the force of gravity Heat sink it takes a lot of energy to change the temperature of water Carbon Compounds Most important element for living things Can make 4 bonds because it has only 4 electrons on the outer shell Carbon Compounds Functional groups clusters of atoms that influence the properties of the molecules they compose molecules with the same functional groups act similar Ex: hydroxyl, amine Carbon Compounds Monomers simple molecules that join together to make large compounds Ex: monosaccharides, amino acids Polymers several monomers joined together to form complex molecules Ex: polysaccharides, proteins Carbon Compounds Macromolecules very large polymers Ex: DNA Condensation reaction Dehydration synthesis Reaction that links monomers Removes a water molecule take an H+ from one molecule & a OH- from another make polymers Hydrolysis reaction Reaction that breaks apart polymers by addition of a water molecule one molecule gets an H+ & one gets OH – It is used to turn ATP into ADP ATP & ADP ATP has 3 phosphates (adenosine triphosphate) ADP has 2 phosphates (adenosine diphosphate) hydrolysis breaks off a phosphate from ATP to ADP this releases a great deal of energy cells use the energy released to function & live make Molecules for Life 1. 2. 3. 4. Carbohydrates Proteins Lipids Nucleic Acids Carbohydrates made of Carbon, Oxygen, & Hydrogen 1. Monosaccharides monomer – it is a single unit 3 types - Glucose, Fructose, & Galactose all 3 have same chemical formula C6H12O6 they have different structural formulas (shape) molecules that have the same chemical formula but different structural formulas are called isomers Carbohydrates 2. Disaccharides are two monosaccharides joined Ex: Sucrose 3. Polysaccharides many monosaccharides are joined Plants store starch Animals store glycogen Proteins contain C H O N made up of Amino Acids (monomers) Peptide bonds join amino acids 20 different amino acids dipeptide – 2 amino acids joined together polypeptide – long chain of amino acids - usually bent or folded - shape will change because of heat, acidity, & other factors Proteins Enzymes special proteins that are biological catalyst lock & key fit with enzyme & substrate substrate – is the substance that binds to enzyme substrate changes enzyme does not Lipids large non-polar molecules that do not dissolve in water fatty acids (monomers) Have a water loving end – hydrophilic Polar end Has a water fearing end – hydrophobic Non-polar end Lipids Triglyceride – 3 fatty acids & a glycerol Saturated – unhealthy Solid at room temp Unsaturated – healthy Liquid at room temp Trigylcerides Saturated vs Unsaturated Lipids Phospholipids – 2 fatty acids & a glycerol Make up cell membranes Lipids Wax – long chain fatty acids Plant leaf covers & ears Steroids – lipids shaped in a ring instead of a chain Ex: cholesterol, testosterone & estrogen Steroids Nucleic Acids large molecules that store genetic information DNA info for cell functions RNA info for making proteins Nucleotides (monomers) made of 1. 2. 3. Phosphate group Sugar Nitrogen Base Neucleotide