* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cellular Respiration
Biosynthesis wikipedia , lookup
Mitochondrion wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthesis wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Electron transport chain wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Citric acid cycle wikipedia , lookup
Biochemistry wikipedia , lookup
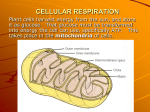
Cellular Respiration Core 3.7 & Option C3 3.7.1 Define cellular respiration. Introduction: • • • • All forms of life depend directly or indirectly on light energy captured during photosynthesis Photosynthesis is done by plants to make glucose In cellular respiration, glucose molecules are broken down back into carbon dioxide and water (molecules the plant started with) Cell respiration is the controlled release of energy from organic compounds in cells to form ATP. ATP – Adenosine triphosphate most common energy carrier in cells nucleotide composed of adenine, the sugar ribose, and three phosphate groups synthesized from adenosine diphosphate (ADP) and inorganic phosphate – process is called phosphorylation during glucose breakdown, energy is release and stored in bonds of ATP Adenine Ribose Summary of complete glucose metabolism: Photosynthesis: 6CO2 + 6H2O + sunlight energy C6H12O6 + 6O2 Complete glucose metabolism: C6H12O6 + 6O2 6CO2 + 6H2O + chemical and heat energies 3.7.2 State that, in cellular respiration, glucose in the cytoplasm is broken down by glycolysis into pyruvate, with a small yield of ATP. Glycolysis First stage of aerobic respiration Does not require O2 (anaerobic) and proceeds in exactly the same way under both aerobic (with oxygen) and anaerobic (without oxygen) conditions Splits apart a single glucose molecule (6 carbon) into two molecules of pyruvate (3 carbon). 2 ATP are yielded. Occurs in cytoplasm Under anaerobic conditions, pyruvate is converted by fermentation to lactic acid or ethanol Under aerobic conditions, pyruvate may enter the mitochondria – breaks pyruvate down completely to CO2 and water generating an additional 34 to 36 ATP – aerobic respiration Each step (reaction) is catalyzed by an enzyme Products of glycolysis are 2 molecules of ATP and 2 molecules of NADH – Nicotinamide adenine dinucleotide (NAD) – an electron carrier that transports energy in the form of energetic electrons – It is a coenzyme – electrons are held in high-energy outer electron shells – NAD+ NADH – donates the electrons and their energy to other molecules – hydrogen ions are often picked up simultaneously NAD+ + 2H+ +2e- NADH + H+ (NADH2) C.3.1 State that oxidation involves the loss of electrons from an element, whereas reduction involves a gain of electrons; and that oxidation frequently involves gaining oxygen or losing hydrogen, whereas reduction frequently involves losing oxygen or gaining hydrogen. Products are formed through oxidation/reduction reactions (redox) – involves two complementary reactions – Oxidation – liberates (releases) energy from the oxidation substance; results from the removal of one or more electrons, alone or with H+ – In biological oxidation, oxygen is frequently added to the oxidized compound Reduction – stores energy in a reduced compound; reduction results from addition of one or more electrons, alone or with H+ In biological reduction, compounds frequently lose oxygen OIL RIG = Oxidation Is Loss of electrons; Reduction Is Gain of electrons OIL RIG = Oxidation Is Loss of electrons; Reduction Is Gain of electrons C.3.2 Outline the process of glycolysis, including phosphorylation, lysis, oxidation and ATP formation. Glycolysis consists of two major sets of reactions: Step 1 - glucose activation (phosphorylation) – 2 ATP are used to convert stable glucose into highly unstable fructose bisphosphate (6 C) Step 2 – Energy harvest (lysis & oxidation) – – – – fructose bisphosphate splits into two 3 C molecules of glyceraldehyde 3-phosphate (G3P aka PGAL) *lysis each G3P molecule goes through series of reactions that convert it into pyruvate (pyruvic acid) During these reactions, 2 high energy electrons and a H+ are added to NAD+ to form “energized” carrier NADH – 2 NADH are made (one from each PGAL) *oxidation 2 ATPs are made per G3P for a total of 4 – however, net gain is only 2 ATPs 3.7.3 Explain that, anaerobic cellular respiration, pyruvate can be converted in the cytoplasm into lactate, or ethanol and carbon dioxide, with no further yield of ATP. Fermentation In the absence of oxygen, pyruvate acts as electron acceptor from NADH producing ethanol or lactic acid – this process is called fermentation NADH production is not used as a method to capture energy – used to get rid of hydrogen ions and electrons made when glucose is broken down NAD+ is regenerated by pyruvate acting as final electron acceptor - pyruvate may be converted to lactic acid (lactic acid fermentation – occurs in human muscles during strenuous exercise) or ethanol and CO2 (alcoholic fermentation – occurs in plants, many yeasts & some bacteria) The only ATP produced from anaerobic respiration are the 2 yielded in glycolysis, none is made in fermentation In anaerobic conditions: Without fermentation, NADH would not be able to be converted back to NAD+ and no more pyruvate would be made. In other words, without fermentation, glycolysis could not continue and ATP production would stop. 3.7.4 Explain that, during aerobic cell respiration, pyruvate can be broken down in the mitochondria into carbon dioxide and water with a large yield of ATP. Aerobic Respiration In the presence of oxygen, oxygen is the electron acceptor (in the electron transport system) allowing pyruvate to be fully broken down (back into CO2 and water) to make even more ATP Aerobic Cellular Respiration – – – series of reactions, occurring under aerobic conditions, in which large amounts of ATP are produced pyruvate is broken down into carbon dioxide and water – oxygen serves as final electron acceptor each step is catalyzed by enzymes C.3.6 Explain the relationship between the structure of the mitochondria and its function. Aerobic Respiration Occurs in the mitochondria – – – – double membrane – inner folds are called cristae inner compartment contains fluid matrix intermembrane compartment separates the two membranes Mitochondria have their own DNA (circular chromosome) and ribosomes (70s – smaller than eukaryotic ribosomes) C.3.3 Draw and label a diagram showing the structure of a mitochondria as seen in electron micrographs. C.3.4 Explain aerobic respiration, including the link reaction, the Kreb’s cycle, the role of NADH + H+, the electron transport chain and the role of oxygen. Aerobic Respiration Step 1 – Glycolysis Step 2 – Oxidative Decarboxylation (Link Reaction) – two molecules of pyruvate produced by glycolysis are transported across both mitochondrial membranes into matrix – each pyruvate is split into CO2 and a 2 C acetyl group which immediately attaches to coenzyme A to form acetyl CoA – during this reaction NADH is produced (oxidative piece) *Decarboxylation = loss of CO2 x – – Step 3 – Krebs Cycle (Citric Acid Cycle) the acetyl CoA enter Krebs cycle by briefly combining with oxaloacetate to form citrate – coenzyme A is released to be reused Kreb’s cycle rearranges citrate to regenerate oxaloacetate giving off 2 CO2, 1 ATP and four electron carriers (1 FADH2 and 3 NADH) per pyruvate molecule (x2 per glucose molecule) Electron Transport System (Oxidative Phosphorylation) energetic electrons from NADH and FADH2 are used to generate more ATP (3 ATP are generated per NADH and 2 ATP per FADH2) located in inner mitochondrial membrane electrons move from molecule to molecule along transport system – energy released by electrons is used to pump hydrogen ions from the matrix across the inner membrane into the intermembrane compartment (used for chemiosmosis) at the end of the ETS, oxygen and hydrogen ions accept the electrons to form water – clears out transport system for more electrons to run through C.3.5 Explain oxidative phosphorylation in terms of chemiosmosis. Chemiosmosis hydrogen ions pumped across the inner membrane generate a large H+ concentration gradient (high concentration in intermembrane compartment and low concentration in matrix) inner membrane is impermeable to hydrogen ions except at protein channels that are part of ATPsynthesizing enzymes (ATP synthase) - whole thing is called the F1 complex during chemiosmosis, hydrogen ions move down the concentration gradient from intermembrane compartment to matrix by means of the F1 complex the flow of hydrogen ions provides energy to synthesize 32 – 34 ATPs from ADP Metabolism of Fats and Proteins Cells can also extract energy from fats and proteins Breakdown of fat and proteins creates products that can be fed into the enzyme pathways of respiration Fats – starts with hydrolysis into glycerol and fatty acids – glycerol is converted into G3P and enters pathway – fatty acids are converted into acetyl-CoA and enter pathway Proteins – amino acids are broken down in a number of ways the amino group is first removed (deamination) some amino acids are converted into pyruvic acid, some into acetyl-CoA, and some into other compounds in the Krebs cycle Mammals and birds (and some other organisms) make use of heat produced during metabolism – have evolved mechanisms to conserve heat (insulation with fat, hair, feathers) – – – Endothermic (“internally heated”) – have fairly high body temps (higher than environment) Homeothermic – tend to maintain fairly constant body temp even when environmental temp fluctuates metabolic rate stays fairly constant