* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Proteins
Magnesium transporter wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Protein moonlighting wikipedia , lookup
Protein (nutrient) wikipedia , lookup
Protein domain wikipedia , lookup
List of types of proteins wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Protein folding wikipedia , lookup
Circular dichroism wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
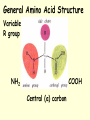
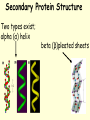
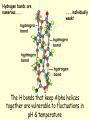
PROTEINS • Have a wide range of functions; haemoglobin, antibodies & enzymes • Amino acids are the monomers • Twenty naturally occurring amino acids • The order of the AA and the way they are folded dictates the function General Amino Acid Structure Variable R group NH2 COOH Central (α) carbon Forming a dipepetide •AA 1 and AA2 line up •Amine next to carboxyl group •Amine Grp ‘donates’ OH •Carb. Grp ‘donates’ H •Water is eliminated •A dipeptide is formed Forming a polypepetide •1000s of AAs link up •Ribosomes convert AA to polypeptides •A string of AA like this is PRIMARY STRUCTURE Secondary Protein Structure Two types exist; alpha (α) helix beta (β)pleated sheets Alpha (α) helix • AA interact with each other • The H on the NH grp is attracted to the O on the CO grp • The H is slightly positive and the O is slightly negative • A H bond forms between these two atoms Hydrogen bonds are numerous…….. ……..individually weak! The H bonds that keep Alpha helices together are vulnerable to fluctuations in pH & temperature Beta (β) pleated sheets Hydrogen bonds hold adjacent primary chains together To go above secondary structure, proteins need more bond types; Disulphide bonds Adjacent cysteine AA Weak bond Broken by reducing agents Ionic bonds Forms between ionised amine & carboxylic grps Broken by pH extreme Hydrophobic Interactions Btwn non polar R-grps TERTIARY PROTEIN STRUCTURE • Classic example – MYOGLOBIN • Folded into a precise 3D shape • H bonds form between tryptophan, arginine & asparagine. • Dulsulphide bonds with cysteine • Ionic bonds & hdrophobic interactions also exist Myoglobin – a tertiary Level protein QUATERNARY PROTEIN STRUCTURE • Classic examples – HAEMOGLOBIN COLLAGEN • Globular proteins are involved in chemical reactions & are soluble molecules; haemoglobin • Fibrous proteins have a structural role in biology; collagen HAEMOGLOBIN • made of 4 polypeptide chains • two alpha chains • two beta chains • Hydrophobic R grps point into the molecule • Hydrophilic R grps point out of the molecule • This makes haemoglobin highly soluble HAEMOGLOBIN • each of the 4 chains has a haem group • the haem group is not made of AA, but is an integral part of the protein – prosthetic grp. • Each haem group contains an ion of iron (Fe2+) • Each haem binds with one molecule of oxygen (O2) • So each haemoglobin can carry O8 Haemoglobin – a quaternary Level protein