* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download The Chemistry of Life
Catalytic triad wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Microbial metabolism wikipedia , lookup
Genetic code wikipedia , lookup
Western blot wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Protein structure prediction wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Proteolysis wikipedia , lookup
Metalloprotein wikipedia , lookup
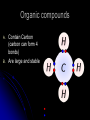
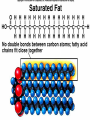
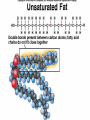
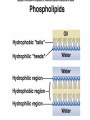
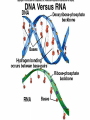
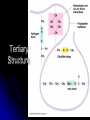
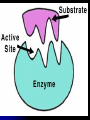
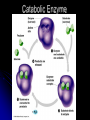
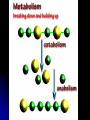
Organic Compounds Organic compounds A. B. Contain Carbon (carbon can form 4 bonds) Are large and stable Organic Compounds C. Examples: Biomolecules Biomolecules are organic compounds produced by living things. There are four groups of biomolecules: 1. Carbohydrates 2. Lipids 3. Nucleic Acids 4. Proteins Carbohydrates Carbohydrates Examples – sugars, glucose, glycogen, starch, cellulose Elements – C,H,O H:O ratio – 2:1 Monomer – monosaccharide Function – Quick energy Storage Carbohydrates Made of carbon, hydrogen, and oxygen 1. Monosaccaride – simple carb a. b. Function: Energy source (ex: glucose, fructose) Form: a “circle” of carbons, also including hydrogen and oxygen Carbohydrates 2. Polysaccaride – complex carb a. b. Function: Energy source (ex: starch in plants and glycogen in animals) and structural (ex: cellulose and chitin) Form: a “string” of monosaccarides Monosaccharides – smallest unit Polysaccharides Simple vs Complex Carbs? Lipids – fats, waxes Lipids Examples – fats, oils, waxes, steroids Elements – C,H,O H:O ratio – >2:1 Monomer – glycerol + fatty acids Function – Long-term energy Storage Lipids Made mostly of carbon and hydrogen. Lipids include fats, oils, waxes, and steroids (such as cholesterol.) Lipids are generally not soluble in water Lipids 1. 2. Function: energy storage and waterproofing membranes Form: Gylcerol attached to a long chain of fatty acids Triglycerides – contain C, H, O (H:O > 2:1) Saturated vs Unsaturated The Good – Omega-3 Fats Lower blood pressure Decrease risk of heart attack Protect against irregular heartbeats Nucleic Acid Nucleic Acids Examples – DNA & RNA Elements – C,H,O,N,P H:O ratio – N/A Monomer – nucleotide Function – Heredity Nucleic Acids Macromolecules containing carbon, hydrogen, oxygen, nitrogen, and phosphorus 1. Function: store and transmit genetic information Form: long chain of nucleotides. Each nucleotide is made of a 5-carbon sugar, a phosphate group, and a nitrogenous base. 2. Proteins Proteins Examples – insulin, keratin, enzymes Elements – C,H,O,N H:O ratio – N/A Monomer – amino acid Function – building and repairing, speeding up reactions Proteins Macromolecules containing carbon, hydrogen, oxygen, and nitrogen 1. Function: a. b. c. 2. Control the rate of reactions and regulate cell processes Form bones and muscles Transport substances into or out of cells or help fight disease Form: long chain of amino acids folded into complex structures Peptide Bonds/Polypeptide Primary Structure – amino acid sequence determines the protein -If you switch 2 amino acids, you get a whole new protein Secondary Structure – alpha helix & beta pleated sheet Tertiary Structure Quaternary Structure – 2 or more polypeptides bond together Protein Folding Inactive Active Enzymes: Bind to substrate (molecule it acts on) Are specific (binds to certain substrate) Has active site (where substrate binds) Are reusable Are affected by temperature and pH Are PROTEINS Metabolism – all chemical reactions in a biological system Catabolism – breakdown of substances Anabolism – formation of new products Catabolic Enzyme Anabolic Enzyme Chemical Reactions/EA Enzyme Lab Conclusion Rate of reaction is determined by measuring the disappearance of substrate or the accumulation of product Rate of reaction is the slope of the linear portion of the graph Reaction rate is affected by pH, substrate conc., enzyme conc., temperature, and ionic con. Conclusions Enzymes have optimum pH, temperature, and salt concentrations that they work in General rules Temp Lower the temp, the slower the molecules collide – slower the reaction rate Higher the temp, the faster the molecules move around – faster the reaction rate Too high a temperature (60-70 C) – protein denatures – reaction doesn’t occur pH – too high or too low the H+ or OH – ions react with the amino acid side chains (R groups) – improper folding occurs – reaction slows Salt conc. – too much or too little causes improper folding of protein Substrate concentration – lower the substrate conc., the slower the reaction Factors Affecting Enzyme Activity Re-Quiz Get out a sheet of notebook paper Label it Re-Quiz # ___; your name, class period DO NOT write on this re-quiz! Once you are finished, turn the notebook paper into the box and place the re-quiz on my podium please. Unit 1 Review