* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemical Reactions
Chemical thermodynamics wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Freshwater environmental quality parameters wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Water splitting wikipedia , lookup
Flux (metallurgy) wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Transition state theory wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Sodium bicarbonate wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Surface properties of transition metal oxides wikipedia , lookup
Gaseous signaling molecules wikipedia , lookup
Nitrocellulose wikipedia , lookup
Click chemistry wikipedia , lookup
Geochemistry wikipedia , lookup
Stoichiometry wikipedia , lookup
Electrolysis of water wikipedia , lookup
Sodium hydroxide wikipedia , lookup
Sodium hypochlorite wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Chemical reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Acid–base reaction wikipedia , lookup
Electrochemistry wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Alkaline earth metal wikipedia , lookup
Metalloprotein wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
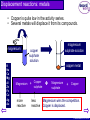
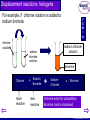
Chemical Reactions © Boardworks Ltd 2003 oxidation and reduction neutralisation precipitation thermal decomposition Types of chemical change reversible reactions exothermic and endothermic displacement reactions: metals displacement reactions: non-metals © Boardworks Ltd 2003 Thermal decomposition Potassium carbonate is not thermally decomposed. Calcium carbonate decomposes on strong heating Silver carbonate decomposes on gentle heating Gets harder • A thermal decomposition is when heat causes a chemical to break down to simpler substances. • Compounds – but not elements - undergo thermal decomposition. • For compounds that contain metals we usually find: the more reactive the metal, the harder it is to decompose its compounds. For example: © Boardworks Ltd 2003 Thermal decomposition Compound How easy to decompose Mercury oxide easy Sodium oxide hard Iron oxide Silver oxide Zinc oxide medium easy medium Potassium sodium calcium magnesium aluminium zinc iron copper mercury silver gold Increasing reactivity Generally, the more reactive the metal, the more difficult it is to decompose its compounds. Fill in the last column: easy, medium or hard. © Boardworks Ltd 2003 Thermal decomposition of carbonates limestone • When carbonates are heated they release carbon dioxide. • This reaction is performed industrially to make calcium oxide (quicklime) from calcium carbonate (limestone). Quicklime is used to make concrete and to make calcium hydroxide (slaked lime). Calcium Carbonate Calcium oxide + waste air and carbon dioxide Hot air 1500°C Carbon dioxide calcium oxide (lime) © Boardworks Ltd 2003 Thermal decomposition of metal oxides • Most metal oxides are thermally stable (i.e. do not decompose when heated). • Oxides of the least reactive metals can be thermally decomposed more easily. • For example, silver oxide begins to break up at about 160oC and mercury oxide decomposes when heated strongly. Mercury Oxide Mercury + mercury metal and oxygen formed O O O O Hg Hg Hg Hg Heat Hg O Hg O O Hg O Hg Hg O Hg O O Hg O Hg Hg O Hg O O Hg O Hg Hg O Hg O O Hg O Hg mercury oxide decomposes oxygen © Boardworks Ltd 2003 Exothermic and endothermic reactions Ex = out (as in exit) En = in (as in entrance) • Exothermic reactions give out heat (gets hot). • Endothermic reactions take in heat (gets cold). • Many chemical reactions need some energy to get them started (activation energy) but then the majority of chemical reactions are exothermic. Shuttle fuel burninghighly exothermic © Boardworks Ltd 2003 Exothermic and endothermic reactions • It is hard to think of examples of endothermic reactions but there are lots of exothermic ones that occur in the laboratory and in everyday life. • List 8 exothermic reactions. Some examples of exothermic reactions Burning wood on a fire Burning petrol in a car Burning butane in a cigarette lighter Burning gas in a gas hob Reacting an acid and alkali together Burning magnesium Rotting compost etc etc © Boardworks Ltd 2003 • These are reactions where two metals are competing to be combined with a non-metal. • The more reactive metal wins the competition and becomes part of a compound. • The less reactive metal is displaced and so is present as the metal at the end of the reaction. Potassium sodium calcium magnesium aluminium zinc iron copper silver gold Increasing reactivity Displacement reactions: metals A more reactive metal (higher in the reactivity series) will displace a less reactive metal from its compound. © Boardworks Ltd 2003 Displacement reactions: metals • Copper is quite low in the activity series. • Several metals will displace it from its compounds. magnesium K Na Ca Mg Al Zn Fe Cu Ag Au magnesium sulphate solution copper sulphate solution copper metal Magnesium more reactive + Copper sulphate less reactive Magnesium sulphate + Copper Magnesium wins the competition. Copper is displaced. © Boardworks Ltd 2003 Displacement reactions: metals Here are some actual photos. The colour changes from blue to red/black as copper metal is displaced. K Na Ca Mg Al Zn Fe Cu Ag Au photograph at start of reaction Magnesium + more reactive photograph at end of reaction Copper sulphate less reactive Magnesium sulphate + Copper Magnesium wins the competition. Copper is displaced © Boardworks Ltd 2003 Displacement reactions: metals The thermit reaction takes place between aluminium and iron oxide. It is so exothermic that molten iron is produced and the reaction is used to repair broken railway tracks. K Na Ca Mg Al Zn Fe Cu Ag Au Aluminium more reactive + Iron Oxide less reactive Aluminium Oxide magnesium fuse iron oxide + aluminium powder + Iron Aluminium wins the competition. Iron is displaced and melts at the high temperatures produced. © Boardworks Ltd 2003 Displacement reactions: metals Here is a photo of the thermit reaction being carried out in a laboratory. magnesium fuse iron oxide + aluminium powder © Boardworks Ltd 2003 Predict which mixtures will result in a reaction. Metal Solution Iron Iron chloride Magnesium nitrate Zinc nitrate Copper sulphate Magnesium Yes No No Yes Yes Yes Zinc Copper Yes No No No No Yes © Boardworks Ltd 2003 Displacement reactions: halogens • Fluorine Chlorine Bromine Iodine Increasing reactivity • These are displacement reactions where two halogens are competing to be combined with a metal. • It is the more reactive halogen that will win and become part of a compound. • The less reactive halogen remains (or becomes) the element. We can often tell which halogen is present from the colour of the solution. © Boardworks Ltd 2003 Displacement reactions: halogens For example, if chlorine solution is added to sodium bromide. chlorine solution F Cl Br I At sodium chloride solution sodium bromide solution bromine Chlorine more reactive + Sodium Bromide less reactive Sodium Chloride + Bromine Chlorine wins the competition. Bromine (red) is displaced. © Boardworks Ltd 2003 Predict what colour these will be after mixing. The compounds of the halogens with Group 1 metals are all colourless. Halogen Halide Chlorine solution Potassium chloride Bromine solution Br2 Potassium bromide Br2 Potassium Iodide I2 Iodine Solution I2 I2 I2 © Boardworks Ltd 2003 Displacement reactions: halogens When writing equations for halogen displacement reactions you must remember that – when in the form of the element – halogens exist in pairs. F Cl Br I At For chlorine and sodium bromide: Chlorine + Sodium sodium bromide chloride + bromine Cl2(aq) + 2NaBr(aq) 2NaCl(aq) + Br2(aq) Cl More reactive Br Less reactive Solution goes yellow/brown as bromine is produced. © Boardworks Ltd 2003 Predict whether or not a chemical reaction will occur. F Cl Br I At • If no reaction - not write “no reaction.” • Where there is a reaction write the names of the products and then write a chemical equation underneath. 1) iodine + sodium bromide solution No reaction 2) bromine + sodium chloride solution No reaction 3) chlorine + sodium iodide solution sodium chloride + iodine Cl2(g) + 2NaI(aq) 2NaCl(aq) + I2(aq) © Boardworks Ltd 2003 Reversible and irreversible reaction • Most chemical reactions are considered irreversible in that the new products are not readily changed back into reactants. For example, once you have reacted magnesium with hydrochloric acid it is very hard to get the magnesium back. • In the equations for irreversible reactions reactants and products are joined by a “one-way arrow.” magnesium + hydrochloric magnesium + hydrogen acid chloride © Boardworks Ltd 2003 Reversible reactions • Although most chemical reactions are difficult to reverse it is possible to find reactions ranging from irreversible through to the fully reversible. • One of the best known reversible processes is heating copper sulphate. Note the double arrow symbol in the chemical equation Heat hydrated copper sulphate anhydrous copper sulphate steam CuSO4.5H20 CuSO4 5H2O these decompose + these combine © Boardworks Ltd 2003 Equilibrium reactions • There are some reactions in which both the “forward and backward” reactions occur to a substantial extent under the same conditions. • These lead to equilibrium mixtures of reactants and products. • One of the most important of these reactions occurs in the Haber Process. N2(g) + 3H2(g) 2 NH3(g) However long you leave the reaction going you still get a mixture of nitrogen, hydrogen and ammonia. © Boardworks Ltd 2003 Getting more product at equilibrium • There are some simple rules that can be used to move the position of an equilibrium towards reactants or products: 1. Exothermic reactions give more product at lower temperatures. (Endothermic – the opposite) 2. Increasing the pressure in gas reactions favours whichever side of the chemical equation has least gas molecules. What conditions will favour formation of more ammonia? 3H2(g) + N2 (g) 2NH3 (g) (exothermic) Low temperature High pressure © Boardworks Ltd 2003 Precipitation reactions • A precipitation reaction is any reaction that produces an insoluble compound when two aqueous solutions are mixed. • It is impossible to predict whether or not we will get precipitation reactions unless we know something about the physical states (especially solubility) of the various reactants and products. Here are the symbols that we use in chemical equations to say what the physical state is: –(s) –(l) –(g) –(aq) solid liquid gas aqueous (dissolved in water) © Boardworks Ltd 2003 Precipitation reactions – first example A precipitation reaction that is often used to measure reaction rates occurs between sodium thiosulphate and hydrochloric acid. Sodium + thiosulphate hydrochloric acid aqueous aqueous Both reactants are colourless and dissolved (aq) sodium + sulphur + water + sulphur chloride dioxide solid aqueous solid liquid gas Sulphur is insoluble and precipitates. This makes the solution go cloudy. © Boardworks Ltd 2003 Precipitation reactions – second example Most metal hydroxides (except sodium, potassium and calcium) are insoluble. Reactions leading to their formation give precipitates. Copper + sulphate aqueous ammonium hydroxide copper hydroxide aqueous solid solid Both reactants are dissolved (aq). Copper sulphate is blue. + ammonium sulphate aqueous Copper hydroxide is insoluble and precipitates. A pale blue solid settles at the bottom of the test tube. © Boardworks Ltd 2003 Precipitation reactions – third example Another metal hydroxide that precipitates is iron(III) hydroxide. Like many transition metals its compounds are coloured. Iron + sodium chloride hydroxide aqueous aqueous Both reactants are dissolved (aq) (iron chloride is yellow). iron hydroxide solid solid + sodium chloride aqueous Iron hydroxide is insoluble and precipitates. A deep brown solid settles at the bottom of the test tube. © Boardworks Ltd 2003 Precipitation and solubility To work out whether a precipitate will be formed we need to know the solubility of the compounds that may be formed. Here are a few general guidelines: Soluble Insoluble All sodium, potassium and ammonium salts All nitrates Most chlorides, bromides and iodides. (halides) Silver and lead halides Most sulphates Lead, barium and calcium sulphates Sodium, potassium and ammonium carbonates Most carbonates Sodium, potassium, ammonium and calcium hydroxide Most hydroxides © Boardworks Ltd 2003 Precipitation and solubility To work out whether a precipitate will be formed when many ionic compounds react there are four stages: 1 Write down the names of the reactants. Sodium chloride & lead nitrate 2 Write down the ions in the reactants. (Ignore numbers) Na+ Cl- Pb2+ NO3- 3 Swap over the + and – ions. Pb2+ Cl- Na+ NO3- 4 Are the products going to be soluble or insoluble? Sodium + chloride aqueous lead nitrate aqueous Lead chloride is insoluble so there will be a precipitate lead chloride solid solid + sodium nitrate aqueous © Boardworks Ltd 2003 Will there be a precipitate if I mix sodium sulphate and magnesium nitrate? 1 Write down the names of the reactants. Sodium nitrate 2 Write down the ions in the reactants. Na+ SO42- Mg2+ NO3- 3 Swap over the + and – ions. Mg2+ SO42- Na+ NO3- 4 Are the products going to be soluble or insoluble? Both the products are soluble there will be no precipitate. Sodium + sulphate aqueous magnesium nitrate aqueous magnesium sulphate aqueous & Magnesium sulphate + sodium nitrate aqueous © Boardworks Ltd 2003 Will there be a precipitate if I mix sodium sulphate and barium nitrate? 1 Write down the names of the reactants. Sodium sulphate & barium nitrate 2 Write down the ions in the reactants. Na+ SO42- Ba2+ NO3Na+ NO3- 3 Swap over the + and – ions. Ba2+ SO42- 4 Are the products going to be soluble or insoluble? Barium sulphate is insoluble so there will be a precipitate. Sodium + sulphate aqueous barium nitrate aqueous barium sulphate solid solid + sodium nitrate aqueous © Boardworks Ltd 2003 Separating Precipitates – reminder! © Boardworks Ltd 2003 Neutralisation reactions • Acids are substances that: • Turn litmus red. • Turn universal indicator yellow, orange or red. • Have a pH below 7. • Form solutions containing H+ ions. • Bases are substances that: • Turn litmus blue. • Turn universal indicator dark green, blue or purple. • React with the H+ ions in acids. • Are called alkalis if they dissolve in water. 1 2 3 4 5 Increasingly acid 6 7 8 9 10 11 12 13 14 Increasingly alkali © Boardworks Ltd 2003 Neutralisation reactions: acids • Common Acids are Name of acid Sulphuric Formula Strong or Weak? H2SO4 strong Hydrochloric HCl strong Nitric HNO3 strong CH3COOH weak Ethanoic (vinegar) • Salts Sulphuric acid Nitric acid Hydrochloric acid Sulphates Nitrates Chlorides © Boardworks Ltd 2003 Neutralisation reactions: bases • Common alkalis are Name of alkali Formula Sodium Hydroxide NaOH strong Potassium Hydroxide KOH strong Ca(OH)2 strong NH4OH weak Calcium Hydroxide Ammonium Hydroxide Strong or Weak? • Common bases (neutralise acids but don’t dissolve) are Type of compound Contain React with acids to give Metal Hydroxides OH- water + a salt Metal Oxides O2- water + a salt CO32- water + a salt + CO2 Metal Carbonates © Boardworks Ltd 2003 Neutralisation reactions: acid + base A neutralisation reaction is where an acid reacts with a base to produce a neutral solution of a salt and water. sodium hydroxide pH 14 neutralisation hydrochloric acid pH 1 1 2 3 sodium chloride pH 7 4 5 Increasingly acid 6 7 8 9 10 11 12 13 14 Increasingly alkali © Boardworks Ltd 2003 Neutralisation - naming salts To name the salt formed in a neutralisation: 1 The first part of the name of the salt comes from the first name of the base So Ammonium hydroxide gives ammonium ………… Magnesium oxide gives magnesium …………... 2 The acid gives the last part of the name of the salt. So Sulphuric acid make sulphates Nitric acid makes nitrates Hydrochloric acid makes chlorides Eg. Sodium hydroxide + nitric acid forms: Calcium carbonate + sulphuric acid forms: Sodium nitrate calcium sulphate © Boardworks Ltd 2003 Name the salt formed in these neutralisations: + Base Acid Calcium hydroxide Hydrochloric acid Magnesium oxide Nitric acid Calcium carbonate Sulphuric acid Aluminium hydroxide Nitric acid Potassium hydroxide Sulphuric acid Salt? Calcium chloride Magnesium nitrate Calcium sulphate Aluminium nitrate Potassium sulphate © Boardworks Ltd 2003 Neutralisation reactions: hydroxides Each OH- ion reacts with one H+ ion. Reaction with hydroxides: H+ + OH- H2O Eg. Potassium +hydrochloric water + hydroxide acid KOH + HCl H2O Eg. Calcium + sulphuric water + hydroxide acid Ca(OH)2 + H2SO4 + potassium chloride KCl calcium sulphate 2H2O + CaSO4 © Boardworks Ltd 2003 Neutralisation Reactions: oxides Neutralisation reactions usually lead to water being formed. Reaction with oxides: 2H+ + O2- H2O Eg. Calcium + hydrochloric water + oxide acid CaO + 2HCl H2O Eg. Sodium + sulphuric water + oxide acid Na2O + H2SO4 H2O calcium chloride + CaCl2 sodium sulphate + Na2SO4 © Boardworks Ltd 2003 Neutralisation Reactions: carbonates Each carbonate ion provides one oxygen to join with two H+ ions. At the same time carbon dioxide is released. 2H+ + CO32- H2O + CO2 Carbonates: Eg. Potassium + hydrochloric water + carbon + potassium carbonate acid dioxide chloride K2CO3 Eg. calcium + carbonate CaCO3 + 2HCl nitric acid + 2HNO3 H2O + CO2 + 2KCl water + carbon + calcium dioxide nitrate H2O + CO2 +Ca(NO3)2 © Boardworks Ltd 2003 Neutralisation equations Complete the word equation Eg. Potassium + hydrochloric hydroxide acid water + Potassium chloride Replace the words with the correct formula KOH Eg. + HCl H2O + KCl Check that it balances (same number of each type of atom each side). Eg. KOH 1*K + HCl Reactants 1*O 2*H H2O + KCl 1*Cl 2*H Products 1*O 1*K 1*Cl © Boardworks Ltd 2003 Neutralisation equations Complete the word equation Eg. Magnesium + nitric oxide acid + water Magnesium nitrate Replace the words with the correct formula Eg. MgO + HNO3 H2O + Mg(NO3)2 Check that it balances (Same number of each type of atom each side. Eg. MgO 1*Mg + 2 HNO3 Reactants 1*O 1*H 2 H 2O 1*NO3 2*H + Mg(NO3)2 Products 1*O 1*Mg 2*NO3 © Boardworks Ltd 2003 Write balanced equations going through the same stages as the previous examples. 1. word equation 2. formulae 3. balance a) sodium hydroxide + hydrochloric acid b) magnesium oxide + hydrochloric acid c) sodium hydroxide + sulphuric acid d) ammonium hydroxide + hydrochloric acid e) calcium hydroxide + nitric acid © Boardworks Ltd 2003 • Insoluble salts can be separated by filtering. • Soluble salts are obtained by evaporating. Put these in the correct order. D A. Check the pH frequently by testing drops of the solution. B. Add the acid slowly to the alkali. C. When neutral pour into the evaporating basin. D. Put on safety specs. E. Allow to cool B A C F E vapour gauze evaporating basin tripod heat-proof mat bunsen burner F. Heat. © Boardworks Ltd 2003 Redox Reactions • Redox is a short way of saying: • Early on in chemistry these words had very straightforward meanings. Oxidation meant adding oxygen to a substance. Rusting (iron becoming iron oxide) is an example of oxidation. Reduction and oxidation Reduction meant taking oxygen away. Extracting iron from iron oxide in the blast furnace is reduction. © Boardworks Ltd 2003 Redox reactions: oxidation and ions • Many redox reactions involve metals and their oxides. • Whenever metals react with oxygen they form ionic compounds and the metal loses electrons to form positively charged ions. • Eg. When magnesium burns to form magnesium oxide magnesium atoms (no charge) become magnesium ions (2+ charge) by losing 2 electrons to oxygen atoms. Mg 2 e- O to give Mg2+ O2- Oxidation involves loss of electrons. © Boardworks Ltd 2003 Redox reactions: electron loss • Think about what has happened to the magnesium when it reacts with oxygen. – It has been oxidised. – It has lost electrons by changing from Mg Mg2+ • Magnesium can also lose electrons to things other than oxygen (e.g. to chlorine or sulphur) and since these also involve Mg Mg2+ these too must be oxidation. O2- Mg2+ O Mg S Mg2+ S2- Cl Oxidation is the loss of electrons. Cl- Mg2+ Cl- © Boardworks Ltd 2003 Redox reactions: electron gain • Exactly the same reasoning applies to reduction. • Reduction can be the removal of oxygen (e.g. from iron oxide to form iron or from aluminium oxide in the electrolysis to extract aluminium.) • When this happens the metal gets back its electrons. – Aluminium has been reduced. – Aluminium has gained electrons O2Al Al3+ O2- Al Al3+ O2- Oxygen removed O 1 ½ O Reduction is the gain of electrons. © Boardworks Ltd 2003 Redox Reactions: oil rig An easy way of remembering this is “Oil Rig”! O oxidation I is L loss of electrons R reduction I is G gain © Boardworks Ltd 2003 Redox Reactions:Two for one! • Whenever something is oxidised, something else is reduced. • This should be obvious if we use the oil rig definition. • If something loses electrons – then something else must have gained them. • For example, when burning magnesium: – Magnesium loses electrons (Mg Mg2+ …..oxidation) – Oxygen gains electrons (O O2- …….reduction) The overall reaction is both Reduction and Oxidation = Redox © Boardworks Ltd 2003 Say whether the substance in red type is oxidised or reduced. Calcium + oxygen calcium oxide zinc + water copper + chlorine iron + aluminium oxide oxidised Zinc oxide + hydrogen reduced Copper chloride reduced Aluminium + iron oxide oxidised © Boardworks Ltd 2003 If the first substance is oxidised, what has been reduced or vice versa (use whichever definition of oxidation and reduction seems easier to apply). Calcium + oxygen oxidised zinc + water copper + chlorine Chlorine is oxidised. It gains an electron Cl- ½Cl2 Aluminium + iron oxide oxidised Hydrogen is oxidised. It gains oxygen. Copper chloride reduced calcium oxide Oxygen is reduced. Each oxygen atom gains 2 e-. Zinc oxide + hydrogen reduced iron + aluminium oxide Iron is reduced. It loses oxygen. © Boardworks Ltd 2003 • Across: 5 tells us whether acid or alkali 11 reaction of an acid with a base • 1 2 3 4 6 7 8 9 10 Down a solid forms in a solution loss of electrons competition reaction gives solutions containing H+ ions to break down into smaller particles removal of oxygen state of balance soluble base ionic compound formed in neutralisations © Boardworks Ltd 2003 Match them up Thermal decomposition Endothermic Dehydrating copper sulphate A solid forms within a solution Metal displacement Reversible reaction Precipitation A salt and water is formed Alkali Reaction in a state of balance Neutralisation Oxidation Reduction Thermit reaction Removal of oxygen Breaking up with heat Soluble base Equilibrium Takes in energy – gets cold Loss of electrons © Boardworks Ltd 2003 When heated the orange powder erupted like a volcano producing a huge pile of green powder that had less mass than the orange material. What type of reaction is this? 1. Neutralisation 2. Thermal decomposition 3. Displacement 4. Precipitation © Boardworks Ltd 2003 When the two colourless solutions mixed a yellow solid formed which sank to the bottom of the test tube. What type of reaction is this? 1. Neutralisation 2. Thermal decomposition 3. Displacement 4. Precipitation © Boardworks Ltd 2003 When the copper was placed in the silver nitrate solution snow-like crystals of silver seemed to grow out from the copper. What type of reaction is this? 1. 2. 3. 4. Equilibrium Thermal decomposition Displacement Precipitation © Boardworks Ltd 2003 When the washing soda was added to the lemon juice it fizzed and the pH rose towards 7. What type of reaction is this? 1. 2. 3. 4. Neutralisation Thermal decomposition Displacement Oxidation © Boardworks Ltd 2003 Which of the oxides shown will thermally decompose most easily? 1. 2. 3. 4. Mercury oxide Potassium oxide Iron oxide Silver oxide © Boardworks Ltd 2003 Which of the salts below might be formed when nitric acid neutralises a metal hydroxide? 1. 2. 3. 4. Potassium hydroxide Potassium nitrate Ammonium nitrate Calcium sulphate © Boardworks Ltd 2003 Which of the mixtures below will result in a metal displacement reaction? 1.Potassium oxide and gold 2.Magnesium and sodium nitrate 3.Copper and silver nitrate 4.Aluminium and calcium sulphate © Boardworks Ltd 2003 Which of the mixtures below will result in a non-metal displacement reaction? 1.Potassium chloride and iodine 2.Potassium bromide and iodine 3.Potassium fluoride and chlorine 4.Potassium iodide and chlorine © Boardworks Ltd 2003 Which of the elements in red (below) is oxidised in the reaction? (Oil Rig!) 1.Ca + 2.2Li + 3.2Al + 4.HNO3 CuO CaO + Cu 2HCl 2LiCl + H2 Fe2O3 Al2O3 + 2Fe + CuO CuNO3 + H2O © Boardworks Ltd 2003 Which compound can you be sure is soluble in water? 1. 2. 3. 4. Manganese nitrate Osmium iodide Thallium chloride Palladium sulphate © Boardworks Ltd 2003