* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Synthesis of an Ester: Fischer Esterification The ester is synthesised
Kinetic resolution wikipedia , lookup
Asymmetric induction wikipedia , lookup
George S. Hammond wikipedia , lookup
Ene reaction wikipedia , lookup
Sulfuric acid wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Petasis reaction wikipedia , lookup
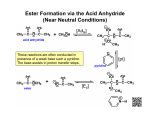
Synthesis of an Ester: Fischer Esterification O R O cat. H2SO4 + R' OH R OH + H2O O R' The ester is synthesised via a Fischer esterification – the reaction of a carboxylic acid with an alcohol in the presence of a mineral acid catalyst Using butyric acid? ⇒ The carboxylic acid is the limiting reagent Using acetic acid? ⇒ The alcohol is the limiting reagent Weigh compounds directly into the reaction flask Be careful with butyric acid – rancid odour! Don’t forget to add the sulfuric acid! Reaction Mechanism Every step of the reaction is reversible. In order to shift the equilibrium position to the right, favouring the ester product, a two-fold excess of either the carboxylic acid or alcohol is used. The acid catalyst (H2SO4) has two functions - It speeds up the reaction rate, allowing equilibrium to be reached more quickly - It acts as a dehydrating agent, helping to shift the equilibrium towards products (alcohols are weak nucleophiles – protonation of the carbonyl group enhances the reactivity of the acid, meaning it is more readily attacked) Reaction Work-Up Wash with 2/3 portions of D.I. water This removes the bulk of the water soluble compounds from the organic layer. Namely, excess alcohol (assuming it is water soluble) and/or excess carboxylic acid, along with the sulfuric acid catalyst. Wash with 2 portions of NaHCO3 Despite washing with water, the organic layer will inevitably remain acidic. Washing with NaHCO3, a weak base, neutralises all acidic compounds in the organic layer, giving water soluble salts: O R O + NaHCO3 + CO2 + H2 O + 2CO2 + 2H2O O Na OH H2SO4 R + 2NaHCO3 Na2SO4 Care has to be taken when performing this extraction, since carbon dioxide gas is liberated during the neutralisation. Reaction Work-Up Wash with 1 portion of saturated aqueous NaCl Since the organic layer was previously washed with NaHCO3 and, in some cases, the ester product has appreciable water solubility, an additional H2O wash is omitted. Instead, the organic layer is washed a final time with NaCl solution. This serves to remove the bulk of the water from the organic layer. Dry over Na2SO4 Remove drying agent by micro-filtration Record the infrared spectrum IR Interpretation Evidence of product: C=O and C–O stretches of ester Presence of impurities: O–H (water, acid, alcohol) Reaction Yield The percent yield for the reaction is highly dependent on the identity of the ester synthesised Esters possessing six or fewer carbons have moderate water solubility. As a result, they are partially extracted into the aqueous layer during work-up, reducing the amount recovered. O O O propyl acetate O methyl butanoate < 6 carbons, one hydrophilic functional group The yield of the reaction is also dependent on the position of the equilibrium (i.e. it not lying completely in favour of products) A number of methods can be employed to drive the reaction completely to product, including: - Removing one of the products (ester or water) from the reaction, as it is formed. - Using an even larger excess of one of the reagents. - Employing a carboxylic acid derivative, such as an acid chloride, which makes the reaction irreversible.