Ternary nucleation of inorganic acids, ammonia, and water
... rest of the paper, the H2 SO4 concentration refers to the total concentration of H2 SO4 共including H2 O–H2 SO4 hydrates兲. Vapors containing nitric acid or HCl are most affected by ammonia. The nucleation rate increases over ten orders of magnitude if the ammonia concentration increases just one orde ...
... rest of the paper, the H2 SO4 concentration refers to the total concentration of H2 SO4 共including H2 O–H2 SO4 hydrates兲. Vapors containing nitric acid or HCl are most affected by ammonia. The nucleation rate increases over ten orders of magnitude if the ammonia concentration increases just one orde ...
2 - equations
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
CHEMICAL AND PROCESS DESIGN HANDBOOK
... of the various chemicals. Each item shows the chemical reaction by which that particular chemical can be manufactured. Equations are kept simple so that they can be understood by people in the many scientific and engineering disciplines involved in the chemical manufacturing industry. Indeed, it is ...
... of the various chemicals. Each item shows the chemical reaction by which that particular chemical can be manufactured. Equations are kept simple so that they can be understood by people in the many scientific and engineering disciplines involved in the chemical manufacturing industry. Indeed, it is ...
Chemsheets AS 1027
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
A high-field solid-state Cl NMR and quantum chemical
... X-Ray crystallography continues to provide much information on many biologically important molecules, but this technique is generally limited to materials that can be crystallized and, in some situations, is unable to probe the exact nature of the interactions between proteins and other biological m ...
... X-Ray crystallography continues to provide much information on many biologically important molecules, but this technique is generally limited to materials that can be crystallized and, in some situations, is unable to probe the exact nature of the interactions between proteins and other biological m ...
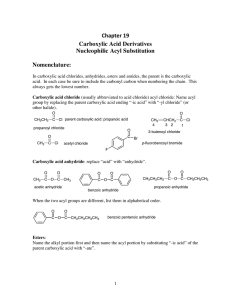
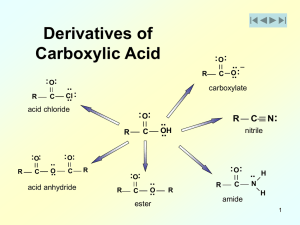
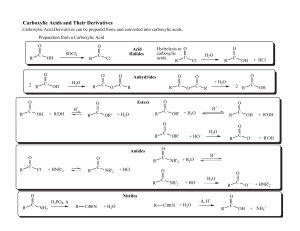
Chapter 19 Carboxylic Acid Derivatives Nucleophilic Acyl
... As we will see, there are two opposing trends: the X-group in each of our carboxylic acid derivatives has a lone pair that can donate electrons to the carbonyl carbon and make the carbonyl carbon LESS electron rich and all of the X-groups have an atom that is MORE electronegative than the carbonyl c ...
... As we will see, there are two opposing trends: the X-group in each of our carboxylic acid derivatives has a lone pair that can donate electrons to the carbonyl carbon and make the carbonyl carbon LESS electron rich and all of the X-groups have an atom that is MORE electronegative than the carbonyl c ...
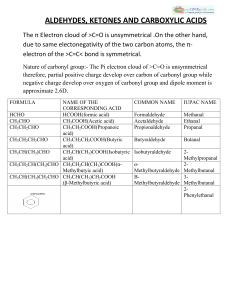
ALDEHYDES, KETONES AND CARBOXYLIC ACIDS
... :-Aldehydes, Ketones and Carboxylic acids are important classes of organic compounds containing carbonyl groups. :-They are highly polar molecules. :-They boil at higher temperatures than the corresponding hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water b ...
... :-Aldehydes, Ketones and Carboxylic acids are important classes of organic compounds containing carbonyl groups. :-They are highly polar molecules. :-They boil at higher temperatures than the corresponding hydrocarbons and weakly polar compounds such as ethers. :-Lower members are soluble in water b ...
The role of formic acid pretreatment in improving the carboxyl
... cellulose fiber along their principal axis, are high aspect ratio fibers with diameters ranging from 3 to 100 nm, depending on their origin. Cellulose nanofibers or NFC have gained much attention for their potential uses as coatings, coating layers, and films. The most relevant and important propert ...
... cellulose fiber along their principal axis, are high aspect ratio fibers with diameters ranging from 3 to 100 nm, depending on their origin. Cellulose nanofibers or NFC have gained much attention for their potential uses as coatings, coating layers, and films. The most relevant and important propert ...
Biological Buffers
... A basic principle is that a buffer that has a pH value of one pH unit above or below the pKa value loses so much buffer capacity that it no longer has any real buffer function. Based on the Henderson-Hasselbalch equation pH = pKa + log [A–]/[HA] for the calculation of the pH of a weak acid or alkal ...
... A basic principle is that a buffer that has a pH value of one pH unit above or below the pKa value loses so much buffer capacity that it no longer has any real buffer function. Based on the Henderson-Hasselbalch equation pH = pKa + log [A–]/[HA] for the calculation of the pH of a weak acid or alkal ...
Crystal engineering of binary compounds containing pharmaceutical
... such as a DNA base pair or the well-known carboxylic acid-pyridyl interaction10). The latter brings with it the opportunity to build multiple component superstructures and is the main focus of this study. ...
... such as a DNA base pair or the well-known carboxylic acid-pyridyl interaction10). The latter brings with it the opportunity to build multiple component superstructures and is the main focus of this study. ...
Full-Text PDF
... decomposition of organo-metallic compounds in supercritical fluids and aerosol methods [7]. Many of these techniques produce nanometer-sized particles that are either amorphous or in γ-phase. In some techniques, the alumina loses its nano-crystalline nature upon repetitive calcinations because as γ- ...
... decomposition of organo-metallic compounds in supercritical fluids and aerosol methods [7]. Many of these techniques produce nanometer-sized particles that are either amorphous or in γ-phase. In some techniques, the alumina loses its nano-crystalline nature upon repetitive calcinations because as γ- ...
Amount of substance
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
... a) reaction of hydrochloric acid (aq) with potassium hydroxide (aq) b) precipitation of silver iodide from reaction between silver nitrate (aq) and potassium iodide (aq) c) reaction of potassium carbonate (aq) with nitric acid (aq) d) precipitation of calcium hydroxide from reaction between sodium h ...
Synthesis and physicochemical study of novel amino acid based
... dosage of detergents which is required to achieve the best cleaning effect has been reduced to one-half or even one-third compared with ten years ago. Concentrated detergent attained almost 28% market share of usual products in Europe. The amount of energy required for washing is further reduced as ...
... dosage of detergents which is required to achieve the best cleaning effect has been reduced to one-half or even one-third compared with ten years ago. Concentrated detergent attained almost 28% market share of usual products in Europe. The amount of energy required for washing is further reduced as ...
The Acidic Environment #2
... pollution. This led to regulations to control emissions from factories, power stations and motor cars. The annual average concentration of SO2 and NO2 in most large cities around the world is 0.01 ppm for each gas. This is about 10 times the value for clean air, though a concentration of 0.01 ppm ...
... pollution. This led to regulations to control emissions from factories, power stations and motor cars. The annual average concentration of SO2 and NO2 in most large cities around the world is 0.01 ppm for each gas. This is about 10 times the value for clean air, though a concentration of 0.01 ppm ...
A Dictionary of the New Chymical Nomenclature
... Gas, oxygenated muriatic acid Dephlogisticated marine acid, of Scheele Gas Prussic acid ...
... Gas, oxygenated muriatic acid Dephlogisticated marine acid, of Scheele Gas Prussic acid ...
Acid-Base Equilibria - Riverside Local Schools
... acid has donated a proton. Similarly, the conjugate acid of a Br0nstedLowry base is the species that results when the base accepts a proton. Two such species that differ from each other only by the presence or absence of a proton together are known as a conjugate acid-base pair. Autoionization of wa ...
... acid has donated a proton. Similarly, the conjugate acid of a Br0nstedLowry base is the species that results when the base accepts a proton. Two such species that differ from each other only by the presence or absence of a proton together are known as a conjugate acid-base pair. Autoionization of wa ...
Answer Key
... reaction proceeds from left to right in order to regain eq. Addition of more solid BaSO4 or BaO will have no effect on the eq. because concentrations of pure solids are not included in the eq. constant expression. If total pressure is altered, the numerical value of K is not changed. The only factor ...
... reaction proceeds from left to right in order to regain eq. Addition of more solid BaSO4 or BaO will have no effect on the eq. because concentrations of pure solids are not included in the eq. constant expression. If total pressure is altered, the numerical value of K is not changed. The only factor ...
Thermodynamics and kinetics of the hydrolysis of atmospherically
... ensure solution homogeneity, an aliquot of the reaction mixture was loaded into an NMR tube and NMR spectral collection was started. The reaction time was recorded as the ending time of each 30 s NMR data collection period. Depending on the rate of reaction for the various species, the solutions wer ...
... ensure solution homogeneity, an aliquot of the reaction mixture was loaded into an NMR tube and NMR spectral collection was started. The reaction time was recorded as the ending time of each 30 s NMR data collection period. Depending on the rate of reaction for the various species, the solutions wer ...
Mechanisms of volatile production from non
... Non-sulfur amino acid monomers were used to study the mechanisms of volatile production in meat by irradiation. Irradiation not only produced many volatiles but also increased the amounts of volatiles from non-sulfur amino acid monomers. The major reaction mechanisms involved in volatile production ...
... Non-sulfur amino acid monomers were used to study the mechanisms of volatile production in meat by irradiation. Irradiation not only produced many volatiles but also increased the amounts of volatiles from non-sulfur amino acid monomers. The major reaction mechanisms involved in volatile production ...
Unit 8 Acids and Bases Notes (answers)
... pH Scale: - a logarithmic scale to measure the acidity (relative [H3O+]) of a solution. - the lower the pH, the more acidic (less basic) is the solution (more [H3O+] and less [OH−]). - the higher the pH, the more basic (less acidic) is the solution (less [H3O+] and more [OH−]). - it is normally repo ...
... pH Scale: - a logarithmic scale to measure the acidity (relative [H3O+]) of a solution. - the lower the pH, the more acidic (less basic) is the solution (more [H3O+] and less [OH−]). - the higher the pH, the more basic (less acidic) is the solution (less [H3O+] and more [OH−]). - it is normally repo ...
Slide 1
... Recall that electron donors (Nu: -’s) add to the electrophilic carbonyl C in aldehydes and ketones. The C=O p bond breaks and the pair of electrons are stabilized on the electronegative O atom. R (alkyl groups) and hydrogens (H) bonded to the C=O carbon remain in place. R- and H- are too reactive (p ...
... Recall that electron donors (Nu: -’s) add to the electrophilic carbonyl C in aldehydes and ketones. The C=O p bond breaks and the pair of electrons are stabilized on the electronegative O atom. R (alkyl groups) and hydrogens (H) bonded to the C=O carbon remain in place. R- and H- are too reactive (p ...
Chapter 19 - U of L Class Index
... 2 CH3CO2H C C H3C O CH3 Esters from alcohols Another good method of making phenyl esters (esters of phenol). ...
... 2 CH3CO2H C C H3C O CH3 Esters from alcohols Another good method of making phenyl esters (esters of phenol). ...
Acid-Base
... 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1.00 litre with pure water, the pH does not change. Discuss why the pH remains constant on dilution. (c) A ...
... 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1.00 litre with pure water, the pH does not change. Discuss why the pH remains constant on dilution. (c) A ...
The Advanced Placement Examination in Chemistry Acid–Base
... 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1.00 litre with pure water, the pH does not change. Discuss why the pH remains constant on dilution. (c) A ...
... 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1.00 litre with pure water, the pH does not change. Discuss why the pH remains constant on dilution. (c) A ...
Unit 6 Review Answers
... Students should disagree. A solution with a pH of 4 has [H3O+] = 0.0001 mol/L, whereas a solution with a pH of 3 has [H3O+] = 0.001 mol/L. The pH of pure (i.e., distilled) water is 7, because the concentration of [H3O+] = [OH−] = 1.0 × 10−7 mol/L, assuming a temperature of 25°C. (a) Describing acids ...
... Students should disagree. A solution with a pH of 4 has [H3O+] = 0.0001 mol/L, whereas a solution with a pH of 3 has [H3O+] = 0.001 mol/L. The pH of pure (i.e., distilled) water is 7, because the concentration of [H3O+] = [OH−] = 1.0 × 10−7 mol/L, assuming a temperature of 25°C. (a) Describing acids ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.