Chapter 15a
... Acid/base problems may fall into 4 categories: strong acid/base, weak acid/base, buffers and hydrolysis. We will go through examples of each of these types of problems one at a time. Strong Acids and Strong Bases The strength of the acid is determined by how far the equilibrium lies to the right. Qu ...
... Acid/base problems may fall into 4 categories: strong acid/base, weak acid/base, buffers and hydrolysis. We will go through examples of each of these types of problems one at a time. Strong Acids and Strong Bases The strength of the acid is determined by how far the equilibrium lies to the right. Qu ...
Extractible Synthesis and Characterization of Cu (II) Complex with
... is inserted in a capillary. The capillary then undergoes mentee in temperature of 1°C.min-1. The melting temperature is determined by visual observation. Some of these compounds have the feature to go through plastic or gels called phases. Others have temperature melting and very close degradation. ...
... is inserted in a capillary. The capillary then undergoes mentee in temperature of 1°C.min-1. The melting temperature is determined by visual observation. Some of these compounds have the feature to go through plastic or gels called phases. Others have temperature melting and very close degradation. ...
Chapter 12 Carboxylic Acids
... Dissociation of either an acid or an alcohol involves breaking an O-H bond, but dissociation of a carboxylic acid gives a carboxylate ion with the negative charge spread out equally over two oxygen atoms, compared with just one oxygen atom in an alkoxide ion. The delocalized charge makes the carboxy ...
... Dissociation of either an acid or an alcohol involves breaking an O-H bond, but dissociation of a carboxylic acid gives a carboxylate ion with the negative charge spread out equally over two oxygen atoms, compared with just one oxygen atom in an alkoxide ion. The delocalized charge makes the carboxy ...
Unit 12 Packet
... As mentioned earlier, water is amphiprotic (amphoteric) - acts as either an acid or a base. But what's most interesting about water is that it undergoes autoionization as follows H2O(l) ...
... As mentioned earlier, water is amphiprotic (amphoteric) - acts as either an acid or a base. But what's most interesting about water is that it undergoes autoionization as follows H2O(l) ...
CHAPTER 15 ACIDS AND BASES
... If Ka1 >> Ka2, we can assume that the equilibrium concentration of hydrogen ion results only from the first stage of ionization. In the second stage this always leads to an expression of the type: (c + y )( y ) = K a2 (c − y ) where c represents the equilibrium hydrogen ion concentration found in th ...
... If Ka1 >> Ka2, we can assume that the equilibrium concentration of hydrogen ion results only from the first stage of ionization. In the second stage this always leads to an expression of the type: (c + y )( y ) = K a2 (c − y ) where c represents the equilibrium hydrogen ion concentration found in th ...
CHEMISTRY
... Mandelic acid can be found in certain skin care products. For example, it is used to treat acne. 100 cm3 of an aqueous solution of mandelic acid contains 1.59 g of mandelic acid. The pKa of mandelic acid is 3.85 at 25.0 oC. ...
... Mandelic acid can be found in certain skin care products. For example, it is used to treat acne. 100 cm3 of an aqueous solution of mandelic acid contains 1.59 g of mandelic acid. The pKa of mandelic acid is 3.85 at 25.0 oC. ...
Full Text
... cystine structures are shown in Figure 1. Elemental sulfur was also mixed with the sucrose in order to determine whether or not the bond energies between the sulfur and the hydrogen and carbon in cystine have an effect on the total heat of combustion via modified side reactions involving sulfuric ac ...
... cystine structures are shown in Figure 1. Elemental sulfur was also mixed with the sucrose in order to determine whether or not the bond energies between the sulfur and the hydrogen and carbon in cystine have an effect on the total heat of combustion via modified side reactions involving sulfuric ac ...
Carboxylic acids, esters, and other acid derivatives
... portion” is important for naming esters using the IUPAC system: 1. The name for the alcohol portion comes first: name the alkyl part of the alcohol (e.g., for the ester shown below, the first part of the ester’s name is methyl (alcohol part comes from methanol). Present the alkyl name separate from ...
... portion” is important for naming esters using the IUPAC system: 1. The name for the alcohol portion comes first: name the alkyl part of the alcohol (e.g., for the ester shown below, the first part of the ester’s name is methyl (alcohol part comes from methanol). Present the alkyl name separate from ...
Analytical Chemistry
... Equilibrium exists between the weak acid, water, H:O', and the anion of the weak acid. The equilibrium lies to the left hand side ofthe equation, indicating that not much H3O* is being produced. The fact that very little H3O* is being produced is the ...
... Equilibrium exists between the weak acid, water, H:O', and the anion of the weak acid. The equilibrium lies to the left hand side ofthe equation, indicating that not much H3O* is being produced. The fact that very little H3O* is being produced is the ...
Rate and Equilibrium
... Arrhenius postulated that acids produce H+ ions in solutions and bases produce OHions. A more general theory of acids and bases was suggested by Bronsted and Lowry which says that acid is a proton donor, and base is a proton acceptor. A conjugate base is everything that remains of the acid molecule ...
... Arrhenius postulated that acids produce H+ ions in solutions and bases produce OHions. A more general theory of acids and bases was suggested by Bronsted and Lowry which says that acid is a proton donor, and base is a proton acceptor. A conjugate base is everything that remains of the acid molecule ...
Identification and determination of pI of amino acids
... carboxylic (-COOH) and one basic amino group (-NH2). Biogenic amino acids bind this two functional groups exclusively on α-carbone. Some of biogenic amino acids can bind another dissociable groups on subordinate chain (-R). Acid-base equilibrium in water solutions depends on pH: ...
... carboxylic (-COOH) and one basic amino group (-NH2). Biogenic amino acids bind this two functional groups exclusively on α-carbone. Some of biogenic amino acids can bind another dissociable groups on subordinate chain (-R). Acid-base equilibrium in water solutions depends on pH: ...
2. 2-Isopropyl-5-methylcyclohexanol on carbon skeletal
... 2. with bases; 3. with electrophilic substrates; 4. recovery; 86. Dissolve copper hydroxide (II) to form a blue complex alcohols: 1. ethanol; 2. benzyl alcohol; 3. ethylene glycol; 4. 2-isopropyl-5-methylphenol; 87. Dissolution of copper hydroxide precipitate (II) to form a blue complex salt solutio ...
... 2. with bases; 3. with electrophilic substrates; 4. recovery; 86. Dissolve copper hydroxide (II) to form a blue complex alcohols: 1. ethanol; 2. benzyl alcohol; 3. ethylene glycol; 4. 2-isopropyl-5-methylphenol; 87. Dissolution of copper hydroxide precipitate (II) to form a blue complex salt solutio ...
conjugate base - DarringtonScience
... An indicator is a colored substance that itself can exist in either an acid or a base form ...
... An indicator is a colored substance that itself can exist in either an acid or a base form ...
Lesson Plan
... CO2 + H2O → H2CO3 → H+ + HCO32. Acid rain is defined as rainfall that is more acidic than pH 5.6. 3. Acid rain is caused by sulfur oxides and nitrogen oxides. Primarily SO2, SO3, NO and NO2. They are released by natural ...
... CO2 + H2O → H2CO3 → H+ + HCO32. Acid rain is defined as rainfall that is more acidic than pH 5.6. 3. Acid rain is caused by sulfur oxides and nitrogen oxides. Primarily SO2, SO3, NO and NO2. They are released by natural ...
Structural Studies on Sulfated Glycopeptides from the Carbohydrate
... with 15 ml of water (a flow-through fraction, not shown) and then eluted by a linear gradient with 50 ml each of water and 1.0 M NaC1. Two-ml fractions were collected and monitored by the carbazole reaction. The separated fractions (D-1-D-5) were desalted on a column (1.2 X 82 cm) of Sephadex G-25 ( ...
... with 15 ml of water (a flow-through fraction, not shown) and then eluted by a linear gradient with 50 ml each of water and 1.0 M NaC1. Two-ml fractions were collected and monitored by the carbazole reaction. The separated fractions (D-1-D-5) were desalted on a column (1.2 X 82 cm) of Sephadex G-25 ( ...
am 06 chemistry - University of Malta
... (6 marks) F is an ester formed by reaction of the alcohol D and the carboxylic acid A. (1) These two compounds are heated together in the presence of conc sulfuric acid as catalyst to form F. (Alternatively, the acid is first converted into the acid chloride (PCl5) and then this reacts more complete ...
... (6 marks) F is an ester formed by reaction of the alcohol D and the carboxylic acid A. (1) These two compounds are heated together in the presence of conc sulfuric acid as catalyst to form F. (Alternatively, the acid is first converted into the acid chloride (PCl5) and then this reacts more complete ...
Acid + Base Class # 1
... 20. Strong acids & strong bases have lots of ions in solution. Strong acids & strong bases are good electrolytes, they will conduct electricity well because of all the loose ions they have in solution. 21. All acids and all bases are electrolytes. 22. Their electrolyte strengths (their ability to c ...
... 20. Strong acids & strong bases have lots of ions in solution. Strong acids & strong bases are good electrolytes, they will conduct electricity well because of all the loose ions they have in solution. 21. All acids and all bases are electrolytes. 22. Their electrolyte strengths (their ability to c ...
Chapter 4
... I. The Chemical Composition of Aqueous Solutions A. Water is the most plentiful solvent on Earth and serves as the major medium for chemical analyses. B. Electrolytes – solutes that form ions when dissolved in water producing a solution with electrical conductivity. -Strong electrolytes ionized comp ...
... I. The Chemical Composition of Aqueous Solutions A. Water is the most plentiful solvent on Earth and serves as the major medium for chemical analyses. B. Electrolytes – solutes that form ions when dissolved in water producing a solution with electrical conductivity. -Strong electrolytes ionized comp ...
Carboxylic acids, esters, and other acid derivatives
... • Recall that constitutional isomers are molecules that share the same formula but differ in their atom-to-atom connectivities. • Three kinds of constitutional isomers (in the order we encountered them): – positional isomers – skeletal isomers – functional group isomers ...
... • Recall that constitutional isomers are molecules that share the same formula but differ in their atom-to-atom connectivities. • Three kinds of constitutional isomers (in the order we encountered them): – positional isomers – skeletal isomers – functional group isomers ...
Chapter 5. ACIDITY AND BASICITY OF ORGANIC COMPOUNDS
... more loosely than a smaller atom with tightly held electrons. For example, iodine whose electrons are far from the nucleus is much more polarizable than fluorine whose electrons are close to the nucleus. Thus, polarizability of halogens increases in the following order: F < Cl < Br < I. Stability of ...
... more loosely than a smaller atom with tightly held electrons. For example, iodine whose electrons are far from the nucleus is much more polarizable than fluorine whose electrons are close to the nucleus. Thus, polarizability of halogens increases in the following order: F < Cl < Br < I. Stability of ...
hydroxy- and oxoacids. heterofunctional compounds of benzene
... interacts with sodium as alcohol, and attaches bromine as unsaturated compounds. These properties can be explained if ester is considered as ester of hydroxycrotonic acids: O CH3 C CH2 C O C 2 H5 OH ...
... interacts with sodium as alcohol, and attaches bromine as unsaturated compounds. These properties can be explained if ester is considered as ester of hydroxycrotonic acids: O CH3 C CH2 C O C 2 H5 OH ...
Chapter Ten
... ► The pH of body fluids is maintained by three major buffer systems. The carbonic acid–bicarbonate system, the dihydrogen phosphate–hydrogen phosphate system, and a third system that depends on the ability of proteins to act as either proton acceptors or proton donors at different pH values. ► The c ...
... ► The pH of body fluids is maintained by three major buffer systems. The carbonic acid–bicarbonate system, the dihydrogen phosphate–hydrogen phosphate system, and a third system that depends on the ability of proteins to act as either proton acceptors or proton donors at different pH values. ► The c ...
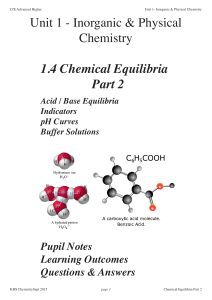
2015 Unit1 Part3.2 Equilibrium2
... A buffer solution is one in which the pH of the solution remains approximately constant when small amounts of acid or base are added or the solution is diluted with water. An acidic buffer consists of a solution of a weak acid and one of its salts with a strong alkali. A basic buffer consists of a s ...
... A buffer solution is one in which the pH of the solution remains approximately constant when small amounts of acid or base are added or the solution is diluted with water. An acidic buffer consists of a solution of a weak acid and one of its salts with a strong alkali. A basic buffer consists of a s ...
Chem. 31 * 9/15 Lecture
... • A Second Way to Make Buffers (not on Exam 1) – To make a traditional buffer, we need both an acid and its conjugate base, but this also can be “made” through other combinations such as: – A weak acid and a strong base or a weak base and a strong acid – This is not covered in the text for buffers, ...
... • A Second Way to Make Buffers (not on Exam 1) – To make a traditional buffer, we need both an acid and its conjugate base, but this also can be “made” through other combinations such as: – A weak acid and a strong base or a weak base and a strong acid – This is not covered in the text for buffers, ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.