General Sciences Sample First Exercise Propanoic Acid Solution
... 2. Justify whether each of the following propositions is true or false: a) pH2 > pH1 b) pH1 = - log C1 c) pH2 = pH1 + 2 d) pH2 < 7 at 25°C. III- Reaction between Propanoic Acid and Sodium Hydroxide Solution The reaction between an aqueous solution of propanoic acid and an aqueous solution of sodium ...
... 2. Justify whether each of the following propositions is true or false: a) pH2 > pH1 b) pH1 = - log C1 c) pH2 = pH1 + 2 d) pH2 < 7 at 25°C. III- Reaction between Propanoic Acid and Sodium Hydroxide Solution The reaction between an aqueous solution of propanoic acid and an aqueous solution of sodium ...
The aim of this exercise is to study the acid... prepare a buffer solution. General Sciences Sample
... 2. Justify whether each of the following propositions is true or false: a) pH2 > pH1 b) pH1 = - log C1 c) pH2 = pH1 + 2 d) pH2 < 7 at 25°C. III- Reaction between Propanoic Acid and Sodium Hydroxide Solution The reaction between an aqueous solution of propanoic acid and an aqueous solution of sodium ...
... 2. Justify whether each of the following propositions is true or false: a) pH2 > pH1 b) pH1 = - log C1 c) pH2 = pH1 + 2 d) pH2 < 7 at 25°C. III- Reaction between Propanoic Acid and Sodium Hydroxide Solution The reaction between an aqueous solution of propanoic acid and an aqueous solution of sodium ...
Acidic Environment
... Then the hydrogen ion reacts with water, ie this reaction occurs: H+ + H2O ...
... Then the hydrogen ion reacts with water, ie this reaction occurs: H+ + H2O ...
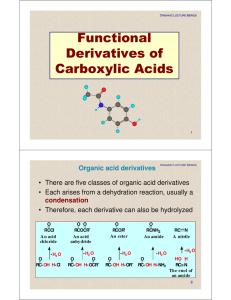
Functional Derivatives of Carboxylic Acids
... Reaction with H2O - Esters – Step 1: attack of hydroxide ion (a nucleophile) on the carbonyl carbon (an electrophile) – Step 2: collapse of the TCAI – Step 3: proton transfer to the alkoxide ion; this step is irreversible and drives saponification to completion O ...
... Reaction with H2O - Esters – Step 1: attack of hydroxide ion (a nucleophile) on the carbonyl carbon (an electrophile) – Step 2: collapse of the TCAI – Step 3: proton transfer to the alkoxide ion; this step is irreversible and drives saponification to completion O ...
PPT CH 8
... • In Step 1, H2SO4 behaves as a strong acid – dissociating completely • In Step 2, HSO4-( behaves as a weak acid – reversibly dissociating, note the double arrow ...
... • In Step 1, H2SO4 behaves as a strong acid – dissociating completely • In Step 2, HSO4-( behaves as a weak acid – reversibly dissociating, note the double arrow ...
+ :O
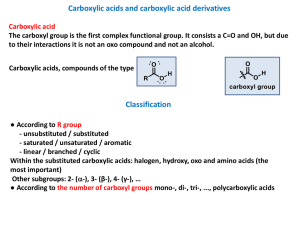
... Carboxylic acids, in neutral solvents, have solubility properties similar to those of alcohols. The first members of the aliphatic series (formic acid through butanoic acid) are miscible with water. Water solubility decreases with increasing chain length, with hexanoic acid being marginally soluble. ...
... Carboxylic acids, in neutral solvents, have solubility properties similar to those of alcohols. The first members of the aliphatic series (formic acid through butanoic acid) are miscible with water. Water solubility decreases with increasing chain length, with hexanoic acid being marginally soluble. ...
Dendrimer-Encapsulated Pd Nanoparticles as Aqueous, Room
... conditions in water, which results in extended catalyst life and reduced likelihood of byproduct formation. Third, the reaction occurs with very good yields in most cases. Several unique properties of DENs1-3 make them attractive for catalytic applications. First, solubility is controlled principall ...
... conditions in water, which results in extended catalyst life and reduced likelihood of byproduct formation. Third, the reaction occurs with very good yields in most cases. Several unique properties of DENs1-3 make them attractive for catalytic applications. First, solubility is controlled principall ...
carbonyl compound group
... (vi) Oxime: Oximes are a class of organic compounds having the general formula RR′CNOH, where R is an organic side chain and R′ is either hydrogen or an organic side chain. If R′ is H, then it is known as aldoxime and if R′ is an organic side chain, it is known as ketoxime. ...
... (vi) Oxime: Oximes are a class of organic compounds having the general formula RR′CNOH, where R is an organic side chain and R′ is either hydrogen or an organic side chain. If R′ is H, then it is known as aldoxime and if R′ is an organic side chain, it is known as ketoxime. ...
08.Carboxylic acids. Functional derivates of carboxylic acids
... carboxylic acids exist as hydrogen-bonded dimers even in the gas phase. In the pure liquid a mixture of hydrogen-bonded dimers and higher aggregates is present. In aqueous solution intermolecular association between carboxylic acid molecules is replaced by hydrogen bonding to water. The solubility p ...
... carboxylic acids exist as hydrogen-bonded dimers even in the gas phase. In the pure liquid a mixture of hydrogen-bonded dimers and higher aggregates is present. In aqueous solution intermolecular association between carboxylic acid molecules is replaced by hydrogen bonding to water. The solubility p ...
The Uptake of Methyl Vinyl Ketone
... volumes (Vliq ) 0.05-2 mL) of water or sulfuric acid solutions were placed in the inner cylinder and rotated (1-5 rpm) in order to maintain a thin film on the wall (∼ 0.02 cm thick for 2 mL of solution). Mixing and saturation of the solutions and wetting of the wall were facilitated by the presence ...
... volumes (Vliq ) 0.05-2 mL) of water or sulfuric acid solutions were placed in the inner cylinder and rotated (1-5 rpm) in order to maintain a thin film on the wall (∼ 0.02 cm thick for 2 mL of solution). Mixing and saturation of the solutions and wetting of the wall were facilitated by the presence ...
Document
... with water to give two molecules of carboxylic acid • higher-molecular-weight acid anhydrides also react with water, but less readily O O CH3 COCCH3 + H2 O ...
... with water to give two molecules of carboxylic acid • higher-molecular-weight acid anhydrides also react with water, but less readily O O CH3 COCCH3 + H2 O ...
physical chemistry notes
... With the bases, the greater the [OH-] then the higher the pH and the greater the conductivity. These experimental results confirm that there are many more hydrogen ions present in a solution of a strong acid than in a weak acid of the same concentration. A further experiment showing this would be ad ...
... With the bases, the greater the [OH-] then the higher the pH and the greater the conductivity. These experimental results confirm that there are many more hydrogen ions present in a solution of a strong acid than in a weak acid of the same concentration. A further experiment showing this would be ad ...
Acidic Environment by Ahmad Shah Idil
... There are many other factors that can affect equilibrium, but these are the most common; simply remember that the system will OPPOSE any change, as stated by Le Chatelier’s principle of chemical equilibrium. ...
... There are many other factors that can affect equilibrium, but these are the most common; simply remember that the system will OPPOSE any change, as stated by Le Chatelier’s principle of chemical equilibrium. ...
Researches of the system of neutralization process control in the
... system has significant drawbacks, because, firstly, it is not clearly understood what the “lower” point of ammonium nitrate solution overflow is; secondly, due to the electrolytic bridge two meters long the displacement of signals arises between the main platinum electrode and the auxiliary silverch ...
... system has significant drawbacks, because, firstly, it is not clearly understood what the “lower” point of ammonium nitrate solution overflow is; secondly, due to the electrolytic bridge two meters long the displacement of signals arises between the main platinum electrode and the auxiliary silverch ...
C3 – Chemicals in Our Lives Revision
... Dioxide SiO2 • This is commonly known as Quartz or Silica ...
... Dioxide SiO2 • This is commonly known as Quartz or Silica ...
carboxylic acid
... Special case - usually irreversible process. Conditions: ● no acidic H (O-H, N-H, S-H) ● sufficiently electrophilic C=O carbon Derivatives that may be relevant : acid halides, acid anhydrides, esters! 1. Reaction with Grignard reagent (you know these ) ...
... Special case - usually irreversible process. Conditions: ● no acidic H (O-H, N-H, S-H) ● sufficiently electrophilic C=O carbon Derivatives that may be relevant : acid halides, acid anhydrides, esters! 1. Reaction with Grignard reagent (you know these ) ...
Chapter 2- Acids and Bases
... D) CH3CH2OH can be a Bronsted-Lowry acid, and CH3CH2NH2 can be a Bronsted-Lowry base. E) CH3CH2OH can be a Bronsted-Lowry acid, CH3CH2NH2 can be a Bronsted-Lowry base, and CH3CH2OH can be a Lewis base. Ans: E 31. Which of the following statements is true? A) CH3CH3 can be a Lewis base. B) BBr3 can b ...
... D) CH3CH2OH can be a Bronsted-Lowry acid, and CH3CH2NH2 can be a Bronsted-Lowry base. E) CH3CH2OH can be a Bronsted-Lowry acid, CH3CH2NH2 can be a Bronsted-Lowry base, and CH3CH2OH can be a Lewis base. Ans: E 31. Which of the following statements is true? A) CH3CH3 can be a Lewis base. B) BBr3 can b ...
Acid - Net Texts
... and amines can be Brønsted-Lowry acids as mentioned above, they can also function as Lewis bases due to the lone pairs of electrons on their oxygen and nitrogen atoms. ...
... and amines can be Brønsted-Lowry acids as mentioned above, they can also function as Lewis bases due to the lone pairs of electrons on their oxygen and nitrogen atoms. ...
FREE Sample Here
... Ans: Ethanol can form hydrogen bonds with water molecules, but ethane cannot. When ethanol dissolves, the decrease in the system's entropy that results from formation of ordered arrays of water around the CH3CH2– group is partly compensated by the favorable interactions (hydrogen bonds) of the hydro ...
... Ans: Ethanol can form hydrogen bonds with water molecules, but ethane cannot. When ethanol dissolves, the decrease in the system's entropy that results from formation of ordered arrays of water around the CH3CH2– group is partly compensated by the favorable interactions (hydrogen bonds) of the hydro ...
Writing and Predicting Chemical Reactions
... about the reaction. In part (i), coefficients should be in terms of lowest whole numbers. Assume that solutions are aqueous unless otherwise indicated. Represent substance in solutions as ions if the substances are extensively ionized. Omit formulas for any ions or molecules that are unchanged by th ...
... about the reaction. In part (i), coefficients should be in terms of lowest whole numbers. Assume that solutions are aqueous unless otherwise indicated. Represent substance in solutions as ions if the substances are extensively ionized. Omit formulas for any ions or molecules that are unchanged by th ...
Kinetics and Equilibrium of the Reversible Formic Acid
... decarbonylation is reversible and that carbon monoxide can be directly converted into formic acid in hot water [5c]. We also made a theoretical analysis on the Gibbs energies of the species (HCOOH, CO, CO2, H2, and H2O) involved in reactions (3) and (4) [5d]. The reversibility and the coupling of th ...
... decarbonylation is reversible and that carbon monoxide can be directly converted into formic acid in hot water [5c]. We also made a theoretical analysis on the Gibbs energies of the species (HCOOH, CO, CO2, H2, and H2O) involved in reactions (3) and (4) [5d]. The reversibility and the coupling of th ...
CHEM 113 GENERAL CHEMISTRY LABORATORY
... * Observe the changes in the substance carefully and write down the type of the changes. 2) Put a small amount of substance (spatula-tip) is into a clean and DRY test tube and fill half of the test tube with pure water. Stir with glass stick and observe. Is the solution homogeneous, heterogeneous, i ...
... * Observe the changes in the substance carefully and write down the type of the changes. 2) Put a small amount of substance (spatula-tip) is into a clean and DRY test tube and fill half of the test tube with pure water. Stir with glass stick and observe. Is the solution homogeneous, heterogeneous, i ...
Chemistry 2100 - Bonham Chemistry
... – Both IUPAC and common names of esters are derived from the names of the parent carboxylic acids. – Name the alkyl or aryl group bonded to oxygen first, followed by the name of the acid; replace the suffix -ic acid by -ate. – A cyclic ester is called a lactone. ...
... – Both IUPAC and common names of esters are derived from the names of the parent carboxylic acids. – Name the alkyl or aryl group bonded to oxygen first, followed by the name of the acid; replace the suffix -ic acid by -ate. – A cyclic ester is called a lactone. ...
Unit 11 acids and bases part 1
... HCl- hydrochloric- stomach acid also known as muriatic acid H2SO4- sulfuric acid - car batteries HNO3 – nitric acid - explosives HC2H3O2- acetic acid - vinegar H2CO3-carbonic acid – sodas H3PO4- phosphoric acid –flavorings HCOOH - formic acid- ant stings ...
... HCl- hydrochloric- stomach acid also known as muriatic acid H2SO4- sulfuric acid - car batteries HNO3 – nitric acid - explosives HC2H3O2- acetic acid - vinegar H2CO3-carbonic acid – sodas H3PO4- phosphoric acid –flavorings HCOOH - formic acid- ant stings ...
Document
... • The acid dissociation constant Ka describes the extent of dissociation of a weak acid (HA H+ and A-) Ka = ...
... • The acid dissociation constant Ka describes the extent of dissociation of a weak acid (HA H+ and A-) Ka = ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.