* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download What is the primary objective of medicinal chemistry ? Which
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Discovery and development of cephalosporins wikipedia , lookup
NK1 receptor antagonist wikipedia , lookup
Psychopharmacology wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Discovery and development of ACE inhibitors wikipedia , lookup
Drug design wikipedia , lookup
Neuropharmacology wikipedia , lookup
Discovery and development of angiotensin receptor blockers wikipedia , lookup
Drug interaction wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Pharmacognosy wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
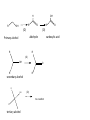
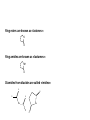
What is the primary objective of medicinal chemistry ? Which disciplines are involved ? The primary objective of medicinal chemistry is the design and discovery of new compounds that are suitable for use as drugs. This process involves a team of workers from a wide range of disciplines such as pharmacy, medicine and other related areas mainly: • analytical chemistry, organic chemistry, pharmaceutical chemistry, • biochemistry, biology, pharmacology, drug metabolism and toxicology, • mathematics, and computing, In our lectures, we will concentrate on the following main titles: • • • • • • • • • • • Research areas of medicinal chemistry Factors affecting biological activity of drugs Drug action Functional groups (their nomenclature, properties, reactions) Heterocyclics (their nomenclature and properties) Stability of organic compounds Metabolism of organic compounds (aims, good and bad aspects, metabolic phases, in vitro and in vivo experiments, metabonates) Pro-drugs Structure-activity relationships Clinical drug development processes Terminology of medicinal chemistry will be given in all of the lectures Main research areas of medicinal chemistry • • • • • Drug synthesis Structure identification Drug metabolism studies Chemical basis of drug bioactivation Pharmacological activity screening Sources of drugs Originally drugs and leads were derived from natural sources. These natural sources are still important sources of lead compounds and new drugs, however the majority of lead compounds are now discovered in the laboratory using a variety of sources, such as local folk remedies (ethnopharmacology), investigations into the biochemistry of the pathology of disease states and highthroughput screening of compound collections, databases and other literature sources of organic compounds. FACTORS ACTING ON THE BIOLOGICAL ACTIVITY OF A DRUG MOLECULE I- PHYSICO-CHEMICAL PROPERTIES OF DRUGS -Solubility -Acid–Base Properties, Ionization, -pKa, pH, logP (oil/water partition coefficient) II-CHEMICAL PROPERTIES OF DRUGS -Chemical bonds -Functional Groups -Heterocyclic rings -Stereoisomerism Solubility A degree of lipid solubility is required to pass through membranes but if it is too high, it is trapped in the membrane and become ineffective. For transportation water solubility is required. Acid base properties Functional groups determine and confer acidity and basicity on a molecule. Human body is 7075% water meaning that we are dealing with a dilute solution if we have a drug molecule of 200 M.A. and a dose of 20 mg pH solution 1 – 3.5 5.3 6.4 7.4 gastric juice intestine saliva blood We can determine the ionisation and absorption site. Chemical properties Chemical bonds If covalent bonds occur between drug and receptor, it is strong and produce irreversible longer pharmacological effects. İf ionic bond occur, it is weaker and has reversible shorter pharmacological effects. Properties of functional groups determine activity, solubility etc. These will be shown later in detail. Stereochemistry Three dimensional structures in space may affect the pharmacological activity. The isomers may interact with receptors in different ways. Racemic mixtures (enantiomers) have asymmetric carbon atoms (chiral center), different pharmacological effects, metabolism and toxicity but same chemical properties. Receptor In order for drug molecules to exhibit their pharmacologic activity, they must interact with a biologic target, typically an enzyme, nucleic acid, or excitable membrane or other biopolymer (RECEPTOR). These interactions occur between the functional groups found in the drug molecule and those found within each biologic target. Lead Compound A compound of known pharmacological activity, the lead related compound is called analogue. Pharmacophor group The moiety interacting with receptor and responsible from biological effect. Bioisosters Groups having similar pharmacophores to eliminate side effects. C=S C=O ; SH, OH, NH2 ; ; O S N Some desirable properties of leads and analogues • Bioavailability (a fraction of the dose of a drug that is found in general circulation) • A molecule mass less than 500 • A calculated value of log P less than 5 • less than ten hydrogen bond acceptor groups (eg. –O- and –N- etc.) • less than five hydrogen bond donor groups (e.g. NH and OH, etc.) We will now be revieving functional groups used in medicinal chemistry. Functional groups have rather than C and H in the molecule and they produce a Chemical, pharmacological, or toxicological effect in the drug formula. ALCOHOLS Any hydroxyl (OH) moiety linked to the saturated C atom forms alcohols. These are generally polar groups. Alcohols can be used as solvents and as mostly synthetic intermediates (SIM). Primary alcohols have 2, secondary alcohols 1 and tertiary alcohols have no H atoms. Alcohols are named with the name of total hydrocarbone name plus -ole ending or alkyl name plus alcohol ending: CH3-OH methanol or methyl alcohol ; C2H5-OH ethanol or ethyl alcohol R R OH OH R OH R R primary secondary R tertiary Primary alcohols can be oxidized to the corresponding aldehydes and ketones but tertiary alcohols not. OH H R R OH R O (O) (O) aldehyde Primary alcohol R carboxylic acid R (O) OH O R R secondary alcohol R OH (O) no reaction R R tertiary alcohol O Esterification of alcohol with carboxylic acid is also important and produces an ester functional group: R-OH + R-COOH -> R-COOR + H2O PHENOLS Phenols have OH groups linked to aromatic ring. OH OH N ETHERS The oxygene bridge has two carbon moiety in ethers (symmetrical or asymmetrical) R-O-R or R-O-R1 Ether is a very inert and non-polar functional group. Ethers can be used as solvents. Their anesthetic property is known. Ring ethers are known as epoxydes and these are generally toxic chemicals. Ethylene oxide O Tetrahydrofurane (THF) O Ethers are named as follows: Groups linked to –O- are alphebetically indicated: CH3-O-C2H5 C2H5-O-C2H5 C6H5-O-C2H5 ethylmethylether diethylether ethylphenylether ALDEHYDES and KETONES Carbonyl gropu at the end of carbon system is aldehyde, if the carbonyl group is in the middle, it is ketone: R-CHO (aldehyde) R-CO-R (ketone) Aldehydes are more reactive than ketones. Their oxidation produces carboxylic acids (look at former slides) Ketones can not be oxidized so they can be somehow inert and used as solvents (ie. acetone) One of the most important common reactions of carbonyl group is imine formation which is rather toxic group: C=O + H2N-R -> C=N-R + H2O (This is a reversible reaction and it goes easily back to the corresponding compounds by hydrolysis) Ketones can be named by indicating first the group linked to carbonyl group alphebetically: CH3-CO-CH2-CH3 Ethylmethylketone C2H5-CO-C6H5 ethylphenylketone CH3-CO-CH3 Dimethylketone (ACETONE) For naming aldehydes, we should know the names of carboxylic acids (please refer to carboxylic acids later). Aldehydes can not be preferred as sovents because of their lability against oxidation and other reactions. Both aldehydes and ketones are used as SIM Ring ketones are indicated by -one ending Their substitent name is oxo O cyclohexanone O 2-oxopentane CARBOXYLIC ACIDS and THEIR DERIVATIVES -COOH (carboxyl group = carbonyl + hydroxyl) This group forms a carboxylic acid moiety and it is a polar and reactive group: H-COOH metanoic acid (formic acid) CH3-COOH etanoic acid (acetic acid) CH3-CH2-COOH propanoic acid (propionic acid) CH3-CH2-CH2-COOH butananoic acid (butyric acid) CH3-CH2-CH2-CH2-COOH pentanoic acid (valeric acid) C6H5-COOH (benzoic acid) O OH N (pyridine-3-carboxylic acid or 3-carboxypyridine) Corresponding aldehydes are named by eliminating ic acid and inserting aldehyde suffixes: H-CHO (formaldehyde) CH3-CHO (acetaldehyde) CH3-CH2-CHO (propionaldehyde) CH3-CH2-CH2-CHO (butyaldehyde) CH3-CH2-CH2-CH2-CHO (valeraldehyde) C6H5-CHO (benzaldehyde) O H N (pyridine-3-carbaldehyde or 3-formylpyridine) Important reactions of carboxylic acids: (from acetic acid) NaOH CH3-COOH 1 , acid, R OH NH3 (R) CH3-COONa (sodium acetate) - a salt CH3-COOR1 (R1 acetate) - an ester CH3-CONH2 (R) (acetamide) - an amide Why are we preparing carboxylic acid derivatives: • To purify carboxylic acids (salts) • To increase and/or decrease the polarity (salts, amides, esters) • To prepare prodrugs (salts, amides, esters) (Acid group is generally not suitable for oral route) Ring esters are known as «lactones»: O O Ring amides are known as «lactames»: NH O Diamides from diacides are called «imides»: O R O O NH NH R O Important diacids*: Oxalic acid Malonic acid Succinic acid Glutaric acid Adipic acid Pimelic acid (n = 0) (n = 1) (n = 2) (n = 3) (n = 4) (n = 5) COOH CH2 COOH *these names will be required for the important drug molecules later in pharmaceutical chemistry To memorize them bear in mind the following words: oh my such good apple pie